INSTRUCTIONS TO CANDIDATES
- Answer all the questions in the spaces provided .
- KNEC Mathematical tables and silent electronic calculators may be used.
- All working must be clearly shown where necessary.
- Candidates should answer the questions in English.

Questions
-
- Give the name of one reagent which when reacted with concentrated hydrochloric acid produces chlorine gas. (1mk)
- A student set out to prepare iron (lll) chloride using apparatus shown in the diagram below
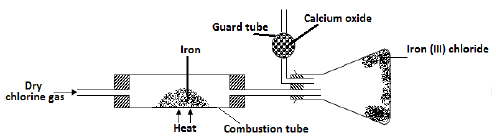
- Explain why it is necessary to pass chlorine gas through the apparatus before heating begins? (1mk)
- Why is calcium oxide most preferred to calcium (II) chloride. (1mk)
- What property of iron (III) chloride makes it possible to be collected as shown in the diagram (1mk)
- The total mass of iron (III) chloride formed was found to be 0.5g. Calculate the volume of chlorine gas that reacted with iron. (Fe = 56, Cl = 35.5 and molar gas volume at r.t.p is 24,000 cm3) (3mks)
- When hydrogen sulphide gas passed through a solution of iron (III) chloride the following observation was made;
The colour of the solution changed from reddish brown to green and yellow solid was deposited. Explain these observations (1mk) - State and explain the observations that would be made if a moist blue- litmus paper was placed in a gas jar full of chlorine gas (2mks)
- Study the energy level diagram below and answer the questions that follow.
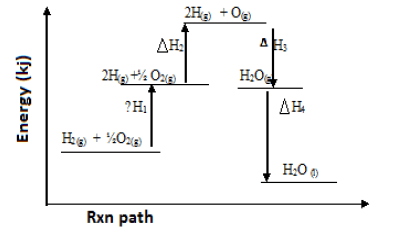
-
- Which ΔH values have a positive sign. 1mk)
- Which ΔH values have a negative sign (1mk)
- What chemical changes is being represented by (2mks)
ΔH1 -
ΔH4 -
- The hydration energy of Al3+ and Cl- are -4690 and -364kJmol-1 respectively. The heat of solution of alluminium chloride is -332kJ mol-1. Calculate the lattice energy of alluminium chloride (2mks)
- When one mole of butanol is burnt. 2676kJ are liberated .
- Calculate the heating value of butanol.(C=12,H=1,O=16) (2mks)
- Write a chemical reaction for combustion of butanol. (1mk)
- Considering the following heats of combustion
HθC (Graphite) = -393kJ mol-1
HθC (H2) (g) = -286kJ mol-1
HθC (Butanol) = -2676kJ mol-1
Using an energy cycle diagram, calculate the heat of formation of butanol. (3mks)
-
-
- The table below shows properties of some elements represented by symbols W,X,Y and Z. Study the information in the table and answer the questions that follows
Element No. Of protons Atomic radius(nm) Boiling point ºC W 2 0.93 -269 X 10 1.31 -246 Y 18 1.54 -186 Z 36 1.89 -152 - Write down the electron arrangement for elements W and X . (1mk)
- In which group of the periodic table are the elements in the table above?Give the chemical family name of the group. (2 mks)
- Explain why the atomic radius of W is smaller than that of X. (1mks)
- State one use of element X. (1mk)
- The section below represents part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbol of the elements.
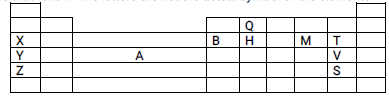
- Select the most reactive non-metal. (1mk)
- Give the name of the elements occupying region A (1mk)
- Explain why the atomic radius of T is smaller than that of M. (2mks)
- Compare the electrical conductivity of elements X and B. (2mks)
- The table below shows properties of some elements represented by symbols W,X,Y and Z. Study the information in the table and answer the questions that follows
-
-
- Write the chemical name for rust. (1 mk)
- State any two ways of preventing rusting. (2 mks)
- Give a reason why vehicles based in Mombasa rust faster than those based in Limuru. (1 mk)
- Oxygen to obtained by fractional distillation of liquid air. Name two other gases which are obtained during the distillation. (1 mk)
- In an experiment to determine the solubility of sodium chloride, 5cm3 of a saturated solution of sodium chloride of mass 5.35g were placed in a volumetric flask and diluted to a total of 250 cm3. 25 cm3 of the dilute solution reacted completely with 24cm3 of 0.1 moldm3 silver nitrate solution. Calculate:
- Moles of silver nitrate in 24cm3 of solution. (1 mk)
- Moles of sodium chloride in 25cm3 of solution. (2 mks)
- Moles of sodium chloride in 250 cm3 of solution. (1 mk)
- Mass of sodium chloride in 5 cm3 of the original saturated sodium chloride solution (1 mk)
- Solubility of sodium chloride. (1 mk)
- The apparatus below was used to investigate the effect of dry hydrogen gas on hot lead (II) oxide.
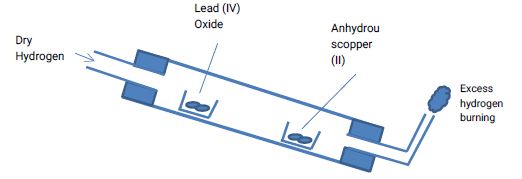
- What is observed in the combustion tube at the end of the experiment? (1 mk)
- Why should the tube be slanting? (1 mk)
- State any precaution to be observed when doing the above experiment. (1 mk)
-
-
- Read the following passage and answer the questions that follow.
A salt K was heated with slaked lime (calcium hydroxide). A colourless gas L with a characteristic choking smell and turns red litmus paper blue was evolved. A large quantity of this gas was passed through an inverted filter funnel into Copper(II)sulphate solution, and a deep blue solution M was obtained.- Identify gas L (1 mk)
- What is K most likely to be? (1 mk)
- Write an equation for the reaction between K and slaked lime (1 mk)
- Write an ionic equation for the reaction with copper(II)sulphate forming the deep blue solution (1 mk)
- Study the flow chart below and answer questions that follow:
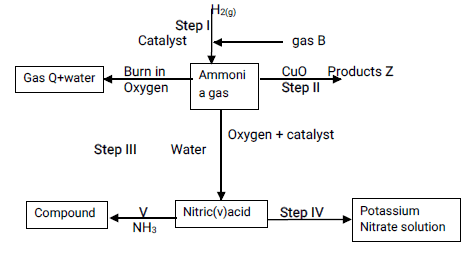
- State one source of gas B (1 mk)
- Name the catalysts used in; (2 mks)
- Step I
- Step III
- Write chemical equations for reactions in; (3 mks)
- Step I
- Step II
- Step V
- Identify any other gas that can be used instead of Ammonia in step II (1 mk)
- State one use of gas Q (1mk)
- Read the following passage and answer the questions that follow.
- An experiment was carried out using magnesium ribbon and dilute hydrochloric acid of different concentrations. The time needed to produce 50cm3 of the gas for every experiment was recorded in a table.
Concentration of HCl (moles per litre) 2.0 1.75 1.50 1.25 1.00 0.75 0.50 0.25 Time (seconds) 8.8 10.0 11.7 14.0 17.5 18.7 35.0 70.0 1/time ( Sec-1) - Complete the table above for 1/time. (4mks)
- Plot a graph of 1/time against concentration

- From your graph determine the concentration needed to produce 50cm3 of hydrogen gas when time is 15.0 seconds (2mks)
- From your graph state the relationship between the rate of reaction and concentration. Give a reason. (1mk)
- The flow diagram below shows a reaction scheme starting from propene.
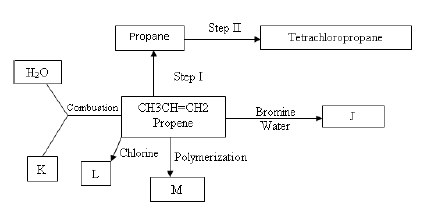
- Name the process in step 1 (1 Mark)
- State the reagent and condition necessary for step1 to occur
Reagent:- (1 Mark)
Condition: - (1 Mark) - Give the names of the following substances. J-
K –
L –
M- (2 Marks) - Write the equation for the reaction that produces substance L. (1 Mark)
- State the type of reaction that occurs in step I. - (1 Mark)
- Write the equation for complete combustion of propene. (1 Mark)
-
- Give one use of substances M. (1 Mark)
- State the environmental effects of the continued use of plastics to the environment
Explain. (2 Marks)











Marking Scheme
-
- Give the name of one reagent which when reacted with concentrated hydrochloric acid produces chlorine gas (1mk)
- potassium manganate (VII)
- Lead (IV) oxide
- Manganese (IV) oxide
- Calcium chlorate (CaOCl2)
- A student set out to prepare iron (lll) chloride using apparatus shown in the diagram below
- Explain why it is necessary to pass chlorine gas through the apparatus before heating begins? (1mk)
- To remove all the oxygen which would react with iron to form iron oxide instead of iron (III) chloride.
- Why is calcium oxide most preferred to calcium (II) chloride. (1mk)
- Absorbs both water and chlorine gas
- What property of iron (III) chloride makes it possible to be collected as shown in the diagram (1mk)
- Iron (III) chloride sublimes
- The total mass of iron (III) chloride formed was found to be 0.5g. Calculate the volume of chlorine gas that reacted with iron. (Fe = 56, Cl = 35.5 and molar gas volume at r.t.p is 24,000 cm3) (3mks)
Fe :Cl2 :FeCl3 2: 3 : 2
RMM FeCl3 = 162.5
Moles of FeCl3 0.5 = 0.003
162.5
3 x.0.003 = 3 x 0.003 = 0.0045
2 ½
Vol of Cl2= 0.0045 x 24000 = 110.8cm3
- Explain why it is necessary to pass chlorine gas through the apparatus before heating begins? (1mk)
- When hydrogen sulphide gas passed through a solution of iron (III) chloride the following observation was made;
The colour of the solution changed from reddish brown to green and yellow solid was deposited. Explain these observations (2mks)
- Fe3+ is reduced to Fe2+; H2S is oxidized to sulphur
- State and explain the observations that would be made if a moist blue- litmus paper was placed in a gas jar full of chlorine gas (2mks)
- Turns, red then white because chlorine is acidic and a bleaching agent in presence of water.
- Give the name of one reagent which when reacted with concentrated hydrochloric acid produces chlorine gas (1mk)
- Study the energy level diagram below and answer the questions that follow.
-
- Which ΔH values have a positive sign. 1mk)
- ΔH1 & ΔH2
- Which ΔH values have a negative sign (1mk)
- ΔH3 & ΔH4
- What chemical changes is being represented by (2mks)
- H1 – Atomisation
- H4 - Condensation
- Which ΔH values have a positive sign. 1mk)
- The hydration energy of Al3+ and Cl- are -4690 and -364kJmol-1 respectively. The heat of solution of alluminium chloride is −332kJ mol-1.
Calculate the lattice energy of alluminium chloride (2mks)
ΔH latt + −4690 + (3x −364) = −332
ΔH latt − 5782 = −332
ΔH latt = +5450kJmol-1 - When one mole of butanol is burnt. 2676kJ are liberated .
- Calculate the heating value of butanol.(C=12,H=1,O=16) (2mks)
C4H9OH =4x12+10x1+16=74
Heating value= 2676 kJ/mole =36.16Kj/g
74g/mole - Write a chemical reaction for combustion of butanol. (1mk)
- 2C4H9OH + 12 O2 (g) → 8 CO2(g) +10 H2O(l)
- Considering the following heats of combustion
HθC (Graphite) = -393kJ mol-1
HθC (H2) (g) = -286kJ mol-1
HθC (Butanol) = -2676kJ mol-1
Using an energy level diagram,calculate the enthalpy of formation of butanol. (3mks)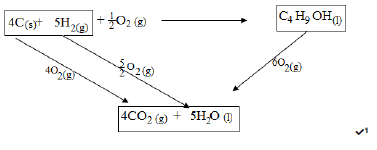
ΔHf + -2676 = (4 x -393) + (5 x -286)
ΔHf = -1572 + -1430 + 2676 = -326kJ mol-1
- Calculate the heating value of butanol.(C=12,H=1,O=16) (2mks)
-
-
- The table below shows properties of some elements represented by symbols W,X,Y and Z. Study the information in the table and answer the questions that follows
Element No. Of protons Atomic radius(nm) Boiling point ºC W 2 0.93 -269 X 10 1.31 -246 Y 18 1.54 -186 Z 36 1.89 -152 - Write down the electron arrangement for elements W and X . (1mk)
- W - 2
- X - 2 .8
- In which group of the periodic table are the elements in the table above?Give the name of the group. (2 mks)
- Group VIII Noble gases
- Explain why the atomic radius of W is smaller than that of X. (1mks)
- X has more/2 energy levels than W (1 energy levels).
- State one use of element X. (1mk)
- Use in making neon advertising coloured signs
- Used to make high voltage indicators
- Neon and helium are used in making gas lasers
- Liquid helium is an economical refrigerant
- Write down the electron arrangement for elements W and X . (1mk)
- The section below represents part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbol of the elements.
- Select the most reactive non-metal. (1mk)
- T
- Give the name of the elements occupying region A (1mk)
- Transition elements
- Explain why the atomic radius of T is smaller than that of M. (2mks)
- T has more protons(18) hence higher nuclear charge than M (17 protons) attracting outermost electrons closer to the nucleaus reducing the atomic radius
- Compare the electrical conductivity of element X and B. (2mks)
- B has higher conductivity,it has 3 delocalised electrons X has 1 delocalised electrons
- Select the most reactive non-metal. (1mk)
- The table below shows properties of some elements represented by symbols W,X,Y and Z. Study the information in the table and answer the questions that follows
-
-
- Write the chemical name for rust. (1 mk)
- Hydrated iron (III)oxide
- State any two ways of preventing rusting. (2 mks)
- Electroplating,Alloying, Galvanizin,Painting,Oiling and greasing
- Give a reason why vehicles based in Mombasa rust faster than those based in Limuru. (1 mk)
- Salty water in Mombasa unlike in Limuru
- Oxygen to obtained by fractional distillation of liquid air. Name two other gases which are obtained during the distillation. (1 mk)
- Nitrogen,Argon
- Write the chemical name for rust. (1 mk)
- In an experiment to determine the solubility of sodium chloride, 5cm3 of a saturated solution of sodium chloride of mass 5.35g were placed in a volumetric flask and diluted to a total of 250 cm3. 25 cm3 of the dilute solution reacted completely with 24cm3 of 0.1 moldm3 silver nitrate solution. Calculate:
- Moles of silver nitrate in 24cm3 of solution. (1 mk)
24 x 0.1 =0.0024 moles
1000 - Moles of sodium chloride to 25cm3 of solution. (2mks)
NaCl : AgNO3
1 : 1
0.0024 moles of NaCl - Moles of sodium chloride in 250 cm3 of solution. (1 mk)
0.0024 x 250 =0.024 moles
25 - Mass of sodium chloride in 5 cm3 of the original saturated sodium chloride solution (1 mk)
NaCl =23+35.5 =58.5g
Mass =58.5 x 0.024 =1.404g - Solubility of sodium chloride. (1 mk)
Mass of water =5.35-1.404 =3.946g
Solubility = 1.404 x 100 =35.58g/100g of water
3.946- The apparatus below was used to investigate the effect of dry hydrogen gas on hot lead (II) oxide.
- What is observed in the combustion tube at the end of the experiment? (1mk)
- A grey solid
- White anhydrous copper(II) sulphate turns blue
- Why should the tube be slanting? (1 mk)
To prevent any liquid collecting in the cooler parts of the combustion tube coming back and cracking the hot part of the tube - State any precaution to be observed when doing this experiment. (1 mk)
- Pass hydrogen through the tube to drive out any air to prevent explosion
- Hot lead metal must be cooled in a stream of hydrogen to prevent re- oxidation of the hot metal.
- What is observed in the combustion tube at the end of the experiment? (1mk)
- The apparatus below was used to investigate the effect of dry hydrogen gas on hot lead (II) oxide.
- Moles of silver nitrate in 24cm3 of solution. (1 mk)
-
-
- Read the following passage and answer the questions.
A salt K was heated with slaked lime (calcium hydroxide). A colourless gas L with a characteristic smell and turns red litmus paper blue was evolved. A large quantity of this gas was passed through an inverted filter funnel into Copper(II)sulphate solution, and a deep blue solution M was obtained.- Identify gas L (1 mk)
- Ammonia
- What is K most likely to be? (1 mk)
- Ammonium chloride
- Write an equation for the reaction between K and slaked lime (1 mk)
- Ca(OH)2(s)+NH4Cl(s)→CaCl2(aq)+H2O(l)+2NH3(g)
- Write an ionic equation for the reaction with copper(II)sulphate forming the deep blue solution (1 mks)
- Cu(OH)2(s) + 4NH3(aq) → [Cu(NH3)4]2+(aq)+2OH–(aq)
- Identify gas L (1 mk)
- Study the flow chart below and answer questions that follow:
- State one source of gas B (1 mk)
- Fractional distillation of liquid air
- Name the catalysts used in; (2 mks)
- Step I
- Finely divided Iron
- Step III
- Platinum or platinum rhodium
- Step I
- Write chemical equations for reactions in; (3 mks)
- Step I
- N2(g) + 3H2(g) → 2NH3(g)
- Step II
- 2NH3(g) + 3CuO(s) → N2(g) + 3H2O(I) + 3Cu(s)
- Step V
- NH3(aq) + HNO3(aq)→NH4NO3(aq)
- Step I
- Identify any other gas that can be used instead of Ammonia in step II (1 mk)
- Hydrogen/Carbon(II)oxide
- State one use of gas Q (1mk)
- Used in the Haber process in the manufacture of ammonia. ¹
- Due to its inert nature, it is mixed with argon to fill electric bulbs (to avoid soot formation).
- In liquid state it is used as an inert refrigerant e.g. storage of semen for artificial insemination.
- Due to its inert nature, it is used in food preservation particularly for canned products i.e. it prevents combination of oxygen and oil which tends to enhance rusting.
- It is used in oil field operation called enhanced oil recovery where it helps to force oil from subterranean deposits.
- State one source of gas B (1 mk)
- Read the following passage and answer the questions.
- An experiment was carried out using magnesium ribbon and dilute hydrochloric acid of different concentrations. The time needed to produce 50cm3 of the gas for every experiment was recorded in a table.
Concentration of HCl (moles per litre) 2.0 1.75 1.50 1.25 1.00 0.75 0.50 0.25 Time (seconds) 8.8 10.0 11.7 14.0 17.5 18.7 35.0 70.0 1/time ( Sec-1) 11.4 10.0 8.54 7.14 5.71 5.34 2.86 1.43 - Complete the table above for 1/time mark each calculation (4mks)
- Plot a graph of rate i.e 1/time against concentration. (3mks)
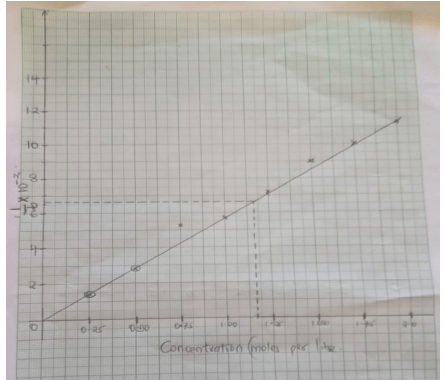
- From your graph determine the concentration needed to produce 50cm3 of hydrogen gas when time is 15.0 seconds (2mks)
T=15
1/15 = 0.0667
= 1.1625 - From your graph state the relationship between the rate of reaction and concentration. Give a reason.
- Rate of reaction increases with increase in concentration (1mks)
- The flow diagram below shows a reaction scheme starting from propene.
- Name the process in step 1 (1 Mark)
- Hydrogenation
- State the reagent and condition necessary for step1 to occur
- Reagent: Hydrogen gas. (1 Mark)
- Condition: - Nickel catalyst/platinum (1 Mark)
180-200ºC Reject range
- Give the names of the following substances.
- J - 2 – bromopropane
- K - Carbon (IV) oxide
- L - 1,2-dichloropropane
- M - Polypropene (2 Marks)
- Write the equation for the reaction that produces substance L. (1 Mark)
- CH₃- CH= CH₂ +Cl₂ CH₃ –CH Cl –CH₂ Cl
- State the type of reaction that occurs in step I. - (1 Mark)
- Addition
- Write the equation for complete combustion of propene. (1 Mark)
- 2C3H6(g) + 9 O2(g) → 6CO2(g) +6H2O(l)
-
- Give one use of substances M. (1 Mark)
- Making buckets
- Making plastic chairs and tables.
- State the environmental effects of the continued use of plastics to the environment
Explain. (2 Marks)- They lead to environmental when they are burned causing pollution.¹
- They are non- biodegradable since bacteria cannot decompose them hence they persist in the environment
- Give one use of substances M. (1 Mark)
- Name the process in step 1 (1 Mark)
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - KCSE 2022 Prediction.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students
