Titration
Titration is determining the end point of the burette contents that react with fixed (usually 25.0cm3 from a pipette) conical flask contents.
As evidence of a titration actually done examining body require the candidate to record their burette readings before and after the titration.
It is important in this practical to use appropriate apparatus to make and record a range of volume measurements accurately.
For KCSE candidates burette readings must be recorded in a titration table in the format provided by the Kenya National Examination Council.
As evidence of all titration actually done Kenya National Examination Council require the candidate to record their burette readings before and after the titration to complete the titration table in the format provided.
Glossary of terms
- Titration - A quantitative procedure in which two solutions react in a known ratio, so if the concentration of one solution is known and the volumes of both are measured, the concentration of the other solution can be determined.
- Indicator - A substance that has different colours, depending upon the pH of the solution it is in.
- Quantitative - Results that can be expressed using numerical values.
- Acid - Corrosive substance which has a pH lower than 7. Acidity is caused by a high concentration of hydrogen ions.
- Alkali - A base which is soluble in water.
- Neutralises - To be made neutral by removing any acidic or alkaline nature.
- Valid - A measurement, test or observation that can be repeated or reproduced to get similar data to support a conclusion.
- precise - A measurement of the spread of numerical results. Data is precise if values are clustered closely together.
- Accurate - When the value of a measurement is close to the true value.
- Pipette - A piece of apparatus used to measure accurate and repeatable volumes of liquid. Also called a volumetric pipette.
- End-point - In a titration, the point at which the indicator first permanently changes colour.
- Repeatable - When a measurement gives a very similar result if it is taken many times over in the same experiment.
- Titre - Volume of one reactant needed to react completely with the other reactant in a titration.
- Meniscus - The curve in the upper surface of a liquid contained within a glass container. Where the container is narrow, like a burette, the meniscus is most noticeable.
Aims
- To carry out an accurate titration using dilute hydrochloric acid, dilute sodium hydroxide solution, and phenolphthalein indicator.
- Titration is a practical technique used to determine the amount or concentration of a substance in a sample. It is an example of quantitative analysis. An acid-alkali titration can be used to find out what volume of acid (or alkali) of known concentration exactly neutralises a known volume of alkali (or acid) of unknown concentration. This concentration can then be calculated.
- To obtain valid results, it is important that measurements are precise and accurate. This can be achieved by using a standard procedure for carrying out a titration.
There are a number of ways that you could carry out a titration in Chemistry. This is an outline of the required steps to undertake one of these methods.
Practical activity 1 - Sample Titration table format
| Final burette reading (cm3) | 24.0 | 24.0 | 24.0 |
| Initial burette reading (cm3) | 0.0 | 0.0 | 0.0 |
| Volume of solution used (cm3) | 24.0 | 24.0 | 24.0 |
Calculate the average volume of solution used
24.0 + 24.0 + 24.0 = 24.0 cm3
3
Practical activity 2 - carrying out a titration
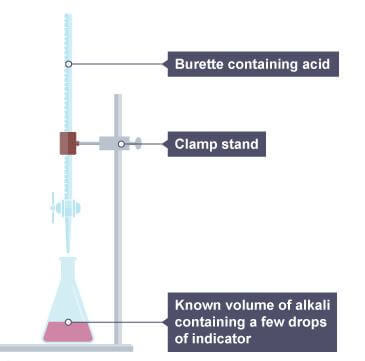
- Use a pipette and pipette filler to add 25 cm3 of alkali solution to a clean conical flask.
- Add a few drops of a suitable indicator and put the conical flask on a white tile.
- Fill the burette with dilute acid. Flush the tap through to remove any air bubbles. Ensure the burette is vertical.
- Slowly add the acid from the burette to the conical flask, swirling to mix. (The mixture may at first change colour, and then back again when swirled.)
- Stop adding the acid when the end-point is reached (when the colour first permanently changes). Note the final volume reading.
- Repeat steps 1 to 5 until three results are repeatable (in close agreement). Ideally these should lie within 0.10 cm3 of each other.
Results
Record the results in a suitable table. The one here also shows some sample readings.
| Run | Rough | 1 | 2 | 3 | 4 |
| End reading (cm3) | 25.45 | 24.80 | 47.90 | 23.70 | 23.90 |
| Start reading (cm3) | 0.00 | 1.00 | 23.80 | 0.00 | 0.00 |
| Titre (cm3) | 25.45 | 23.80 | 24.10 | 23.70 | 23.90 |
The titre is the volume added (the difference between the end and start readings). The burette is marked in 0.10 cm3 graduations. It is possible to record results to the nearest 0.05 cm3 by noting if the meniscus lies between markings.
Analysis
Example
Calculate the mean titre. In the calculation, ignore the rough run and any results that are not in close agreement (24.10 cm3 in the table above).
Ignoring the rough run, and run 2 (because it is not concordant):
mean titre=23.80+23.70+23.90
3
= 23.80 cm3
Evaluation
Questions
- Explain why a pipette is used to measure the acid, rather than a measuring cylinder.
- Describe two steps needed to obtain accurate results.
- Explain the importance of a suitable indicator in obtaining accurate results.
Answers
- A pipette is more precise than a measuring cylinder. Adding slightly different volumes of alkali to the flask will result in a systematic error. The pipette allows the same volume of acid to be added each time, helping to make the results repeatable.
- Take the readings from the bottom of the meniscus. Near to the end-point, rinse the inside of the flask with distilled water and add the acid drop by drop.
- The indicator must change colour sharply when the solution in the flask is neutralised. This means the volume of acid measured is very close to the true value. The white tile makes it easier to see the colour change. Misjudging the colour change could result in a random error.
Hazards, risks and precautions
It is important in this practical activity to use appropriate apparatus and methods. This includes the safe use and careful handling of substances.
Evaluate the hazards and the precautions needed to reduce the risk of harm. For example:
| Hazard | Possible harm | Possible precaution |
|---|---|---|
| Dilute sodium hydroxide solution | Causes skin and serious eye irritation | Wear gloves and eye protection, and use a pipette filler |
| Spilling hydrochloric acid while filling the burette | Causes eye irritation | Fill the burette slowly below eye level, using a |

Note:
- As evidence of understanding the degree of accuracy of burettes, all readings must be recorded to a decimal point.
- As evidence of accuracy in carrying the out the titration, candidates value should be within 0.2 of the school value.
- The school value is the teachers readings presented to the examining body/council based on the concentrations of the solutions s/he presented to her/his candidates.
- Bonus mark is awarded for averaged reading within 0.1 school value as Final accuracy.
- Calculations involved after the titration require candidates thorough practice mastery on the:
- relationship among the mole, molar mass, mole ratios, concentration, molarity.
- mathematical application of 1st principles.
- Very useful information which candidates forget appear usually in the beginning of the paper as:
“You are provided with…” - All calculation must be to the 4th decimal point unless they divide fully to a lesser decimal point.
- Never round off answers.
Download Titration/volumetric analysis - KCSE Chemistry Paper 3 - Practicals.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

