- Introduction
- The Alkali Metals
- Electron Arrangement of the First Three Alkali Metals
- Diagram: Part of Periodic Table Showing the Alkali Metals
- Gradation in Properties of Alkali Metals
- Physical Properties of Alkali Metals
- Chemical Properties of Alkali Metals
- The General Trend in Reactivity of Alkali Metals.
- Similarity of Ions and Formulae of Some Compounds of Alkali Metals.
- Uses of Alkali Metals and their Compounds.
- The Alkaline Earth Metals
- Electron Arrangement of the First Three alkaline Earth Metals.
- Diagram: Part of Periodic Table Showing the Alkaline Earth Metals
- Dot and Cross Diagram for the First Three Alkaline Earth Metals
- Gradation in Properties of Alkaline Earth Metals.
- Physical Properties of Alkaline Earth Metals.
- Chemical Properties of Alkaline Earth Metals.
- Trend in Reactivity in Alkaline Earth Metals.
- Similarity of Ions and Formulae of Some Compounds of Alkaline Earth Metals.
- Uses of Some Alkaline Earth Metals and their Compounds
- The Halogens
- The Noble Gases.

Introduction
- Elements are classified and hence positioned in the periodic table based on the number of valence electrons and the number of energy levels.
- The number of valence electrons is equal to the group to which the element belongs; while the number of energy levels is equal to the period to which the element belongs.
- Elements in the same group are said to belong to the same chemical family.
- Thus a chemical family refers to a group of elements with the same number of valence electrons and hence in the same group of the periodic table.
Characteristics of a Chemical Family
- have same number of valence electrons;
- Show a uniform gradation in physical properties;
- have similar chemical properties;
Main Chemical Families
- Four main chemical families will be studied in this section.
- The Alkali metals
- The Alkaline earth metals
- The halogens
- The noble gases

The Alkali Metals
- Are the elements with one valence electron and hence in group I of the periodic table.
- All are metallic in nature.
- The members of the family in order down the group is as follows:
- Lithium
- Sodium
- Potassium
- Rubidium
- Caesium
- Francium
Electron Arrangement of the First Three Alkali Metals
| Elements | Electron arrangement |
| Lithium | 2.1 |
| Sodium | 2.8.1 |
| Potassium | 2.8.8.1 |
Part of Periodic Table Showing the Alkali Metals
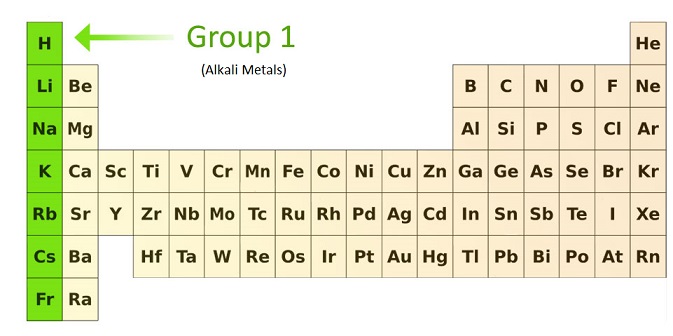
Gradation in Properties of Alkali Metals
1) Atomic and ionic radius
Atomic radius:
- Refers to the distance between the centre of the nucleus of an atom and the outermost energy level occupied by electron(s)
Ionic radius
- Refers to the distance between the centre of the nucleus of an ion and the outermost energy level occupied by electron(s)
Trend
- The ionic radius and the atomic radius of alkali metals increase down the group.
Reason:
- There is an increase in the number of energy levels down the group from lithium to Francium.
Illustration:
- Lithium (2.1) has only 2 energy levels; sodium (2.8.1) has 3 energy levels while potassium (2.8.8.1) has 4 energy levels.
- Thus the outermost electron in potassium is further from the nucleus than the outermost electron in sodium and lithium.
2) Atomic and ionic radius of the same element
- For the same alkali metals the atomic radius is larger than the ionic radius.
Reason:
- Alkali metals form ions by losing the valence electron, leading to the loss of an entire outermost energy level. Thus the atoms have more energy levels than the corresponding ion hence a larger radius in the atom than in the ion.
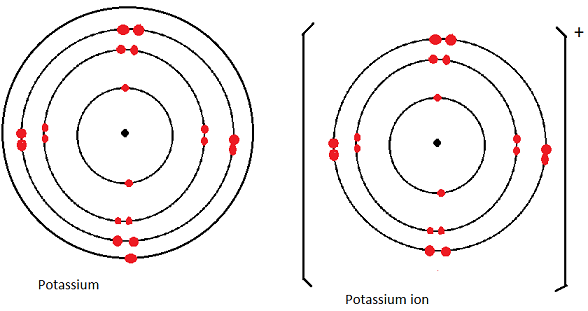
Illustration
- Potassium atom has electron arrangement of 2.8.8.1 hence 4 energy levels.
- During ion formation potassium reacts by losing the single valence electron to acquire a new electron arrangement of 2.8.8 hence 3 energy levels.
- Thus the ion has a smaller radius than the atom.
Diagrammatically: Potassium atom and potassium ion
Summary: changes in atomic and ionic radius among alkali metals
| Element | Symbol | Atomic number | Atomic radius (nm) | Ionic radius (nm) |
| Lithium | Li | 3 | 0.133 | 0.060 |
| Sodium | Na | 11 | 0.157 | 0.095 |
| Potassium | K | 19 | 0.203 | 0.133 |
Physical Properties of Alkali Metals
-
Appearance
- Alkali metals have metallic luster when freshly cut. This refers to a shiny appearance on the cut surface.
- This surface however tarnishes due to reaction on exposure to air.
-
Ease of cutting
- They are soft and easy to cut.
- The softness and ease of cutting increase down the group.
Reason: - Alkali metals have giant metallic structures held together by metallic bonds.
- Metallic bond is due to attraction between the positively charged nucleus of one atom and the electrons in the outermost energy level of the next atom.
- Thus the force of attraction is stronger is smaller atoms than in larger atoms.
- The increase in atomic radius down the group implies that the strength of metallic bonds also decrease down the group (hence ease of cutting and softness).
-
They have relatively low melting and boiling points (in comparison to other metals).
- Reason : they have relatively weaker metallic bonds.
- The melting and boiling points decrease down the group.
Reason: - The size of the atoms increase down the group due to increasing number of energy levels hence decrease in the strength of the metallic bonds (down the group).
-
Electrical conductivity.
- Alkali metals are good conductors of heat and electricity.
Reason : - they have delocalized electrons in the outermost energy level.
- The electrical conductivity is similar for all alkali metals.
Reason : - all alkali metals have the same number of delocalized electron (a single electron) in the outermost energy level.
Note: - In metals the electrons in the outermost energy level do not remain in one fixed position. They move randomly throughout the metallic structure and are thus said to be delocalised.
- Alkali metals are good conductors of heat and electricity.
-
Ionization energy.
- Ionization energy is the minimum energy required to remove an electron from the outermost energy level of an atom in its gaseous state.
- The number of ionization energies an element may have is equivalent to the number of valence electrons.
- The first ionization energy is the minimum energy required to remove the first electron from the outermost energy level of an atom in its gaseous state.
- The first ionization among alkali metals decreases down the group.
Reason: - The effective force of attraction of on the outermost electron by the positive nucleus decreases with increasing atomic size and distance from the nucleus.
- Note that the atomic radius increases down the group due to increase in the number of energy levels.
Summary on physical properties of Alkali metals
| Element | Appearance | Ease of cutting | Melting point ( oC) | Boiling point ( oC) | Electrical conductivity | Atomic radius (nm) | Ionic radius (nm) |
| Lithium | Silver white | Slightly hard | 180 | 1330 | Good | 0.133 | 520 |
| Sodium | Shiny white | Soft | 98 | 890 | Good | 0.157 | 496 |
| Potassium | Shiny grey | Soft | 64 | 774 | Good | 0.203 | 419 |
Chemical Properties of Alkali Metals
Reaction with air
- When exposed to air alkali metals react with atmospheric moisture to form the corresponding metal hydroxide and hydrogen gas.
General equation:
Metal + Water → Metal hydroxide + hydrogen gas. - The metal hydroxide further reacts with atmospheric carbon (IV) oxide to form hydrated metal carbonate.
General equation:
Metal hydroxide + carbon (IV) oxide → Hydrated metal carbonate.
Examples:
Lithium
With moisture : 2Li(s) + 2H2O(l) → 2LiOH(aq) + H2(g) ;
Then with carbon (IV) oxide :2LiOH(aq) + CO2(g) → Li2CO3.H2O(s) ;
Sodium
With moisture : 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) ;
Then with carbon (IV) oxide : 2NaOH(aq) + CO2(g) → Na2CO3.H2O(s) ;
Potassium
With moisture : 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) ;
Then with carbon (IV) oxide : 2KOH(aq) + CO2(g) → K2CO3.H2O(s) ;
Burning in air
- Alkali metals burn in air with characteristic flame colours to form corresponding metal oxides.
Examples:
Lithium
- Burns in air to form lithium oxide as the only product.
- Equation:
4Li(s) + O2(g) → 2Li2O(s)
Sodium
- Burns in air to with a yellow flame to form sodium oxide as the only product.
- Equation:
4Na(s) + O2(g) → 2Na2O(s) - Note:When burned in air enriched with oxygen or pure oxygen sodium burns with a yellow flame to form sodium peroxide (instead of sodium oxide).
- Equation: 2Na(s) + O2(g) → Na2O2(s)
Potassium
- Burns in air with a lilac flame to form potassium oxide as the only product.
- Equation:
4K(s) + O2(g) → 2K2O(s)
Reaction with water
- Alkali metals react with water to form the corresponding hydroxides and hydrogen gas.
Examples:
Potassium.
Procedure:
- A small piece of potassium metal is cut and dropped into a trough containing water;
- The resultant solution is tested with litmus paper;
Diagram of apparatus:
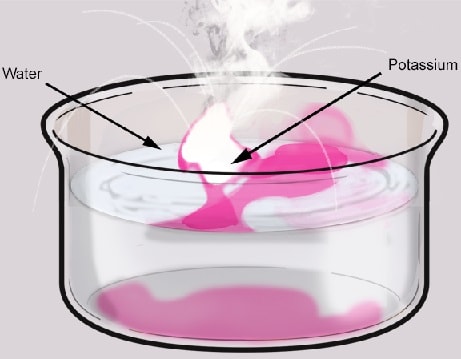
Observations and explanations:
- The metal floats on the water surface; because it is less dense than water;
- A hissing sound is produced; due to production of hydrogen gas;
- It explosively melts into a silvery ball then disappears because reaction between water and potassium is exothermic (produces heat). The resultant heat melts the potassium due to its low melting point.
- It darts on the surface; due to propulsion by hydrogen;
- The metal bursts into a lilac flame; because hydrogen explodes into a flame which then burns the small quantities potassium vapour produced during the reaction;
- The resultant solution turns red litmus paper blue; because potassium hydroxide solution formed is a strong base;
Reaction equations
Equation I
2K(s) + 2H2O(l) → 2KOH(aq) + H2(g) ;
Equation II
4K(s) + O2(g) → 2K2O(s) ;
Equation III:
K2O(s) + H2O(l) → 2KOH(aq)
Effect of resultant solution on litmus paper;
- Litmus paper turns blue; sodium hydroxide formed is highly soluble in water; releasing a large number of hydroxyl ions which result into alkaline conditions // high pH;
Sodium
Procedure:
- A small piece of sodium metal is cut and dropped into a trough containing water;
- The resultant solution is tested with litmus paper;
Diagram of apparatus:

Observations and explanations:
- The metal floats on the water surface; because it is less dense than water;
- A hissing sound is produced; due to production of hydrogen gas;
- It vigorously melts into a silvery ball then disappears because reaction between water and sodium is exothermic (produces heat). The resultant heat melts the sodium due to its low melting point.
- It darts on the surface; due to propulsion by hydrogen;
- The metal may burst into a golden yellow flame; because hydrogen may explode into a flame which then burns the sodium;
- The resultant solution turns blue; because sodium hydroxide solution formed is a strong base;
Reaction equations
Equation I
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) ;
Equation II
4Na(s) + O2(g) → 2Na2O(s) ;
Equation III:
Na2O(s) + H2O(l) → 2NaOH(aq)
Effect of resultant solution on litmus paper;
- Litmus paper turns blue; sodium hydroxide formed is highly soluble in water; releasing a large number of hydroxyl ions which result into alkaline conditions // high pH;
Lithium
Procedure:
- A small piece of lithium metal is cut and dropped into a trough containing water;
- The resultant solution is tested with litmus paper;
Observations and explanations:
- The metal floats on the water surface; because it is less dense than water;
- A hissing sound is produced; due to production of hydrogen gas;
- It reacts less vigorously than sodium and does not melt since the melting point of lithium is relatively higher.
- It darts on the surface; due to propulsion by hydrogen;
- The gas does not ignite spontaneously;
- The resultant solution turns blue; because lithium hydroxide solution formed is a strong base;
Reaction equations.
Equation I
2Li(s) + 2H2O(l) → 2LiOH(aq) + H2(g) ;
Equation II
4Li(s) + O2(g) → 2Li2O(s) ;
Equation III:
Li2O (s) + H2O(l) → 2LiOH(aq)
Effect of resultant solution on litmus paper;
- Litmus paper turns blue; sodium hydroxide formed is highly soluble in water; releasing a large number of hydroxyl ions which result into alkaline conditions // high pH;
Summary on reaction rate.
| Metal | Reaction rate |
| Lithium | Vigorous |
| Sodium | More vigorous |
| Potassium | Explosive |
Reaction with chlorine
- All alkali metals react with chlorine to form corresponding metal chlorides.
General procedure:
- A small piece of the alkali metal is cut and placed in a deflagrating spoon;
- It is then warmed and quickly lowered into a gas jar containing chlorine.
Precautions
- The experiment should be done in a working fume chamber or in the open because Chlorine gas is poisonous.
- This experiment should not be attempted in the laboratory with potassium because the reaction is too explosive and very dangerous.
Observations:
Sodium:
- The metal bursts into a yellow flame.
- White fumes of sodium chloride are formed.
Equation : 2Na(s) + Cl2(g) → 2NaCl(s)
Lithium:
- The metal reacts less vigorously than sodium without bursting into a flame.
- White fumes of lithium chloride are formed.
Equation : 2Li(s) + Cl2(g) → 2LiCl(s)
Potassium:
- The metal bursts into a lilac flame.
- White fumes of potassium chloride are formed.
Equation : 2K(s) + Cl2(g) → 2KCl(s)
The General Trend in Reactivity of Alkali Metals.
- The reactivity of alkali metals increase down the group.
Reason: - Alkali metals react by losing the single valence electron. The ease of loss of the valence electron increases down the group due to decrease in attraction of the valence electron towards the positive nucleus. This in turn is due to the increase in atomic radius down the group as a result of increase in number of energy levels.
Similarity of Ions and Formulae of Some Compounds of Alkali Metals.
- Alkali metals have a similar charge on their ions since they all have a single valence electron.
- Thus they have the same general formula in their ions and compounds.
Examples
| Alkali metal ion | Hydroxide (OH-) | Oxides (O2-) | Chloride (Cl-) | Sulphates (SO42-) |
| Li+ | LiOH | Li2O | LCl | Li2SO4 |
| Na+ | NaOH | Na2O | NaCl | Na2SO4 |
| K+ | KOH | K2O | KCl | K2SO4 |
Note:
- Alkali metals are usually not found as free elements; but rather in their combined states in the earth's crust.
Reason:
- They are highly reactive
Uses of Alkali Metals and their Compounds.
- Sodium is used in the manufacture of sodium cyanide for gold extraction.
- Lithium is used in the manufacture of special high strength glasses and ceramics.
- Lithium compounds are used in the manufacture of dry cells for use in mobile phones, laptops, stopwatches, and zero emission electric vehicles.
- A molten mixture of sodium and potassium is used as a coolant in nuclear reactors.
- Sodium vapour is used to produce the yellow glow in street and advertisement lights.
- Molten sodium is used as a reducing agent in the extraction of titanium.
Equation : Na(l) + TiCl4(g) → Ti(s) + 4NaCl(l) - Sodium chloride is used as a food additive.
- A mixture of sodium hydroxide (caustic soda) and carbon disulphide is used in the manufacture of artificial silk called rayon.

The Alkaline Earth Metals
- Are the elements with two valence electrons and hence in group II of the periodic table.
- All are metallic in nature.
- The members of the family in order down the group is as follows:
- Beryllium
- Magnesium
- Calcium
- Strontium
- Barium
- Radium
Electron Arrangement of the First Three Alkaline Earth Metals.
| Elements | Electron arrangement |
| Beryllium | 2.2 |
| Magnesium | 2.8.2 |
| Calcium | 2.8.8.2 |
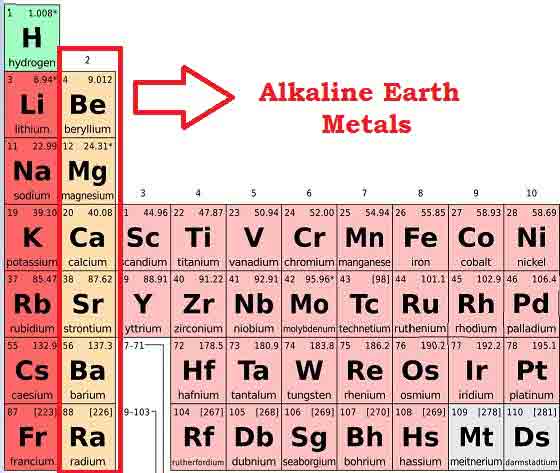
Diagram: Part of Periodic Table Showing the Alkaline Earth Metals
Dot and Cross Diagrams for the First Three Alkaline Earth Metals
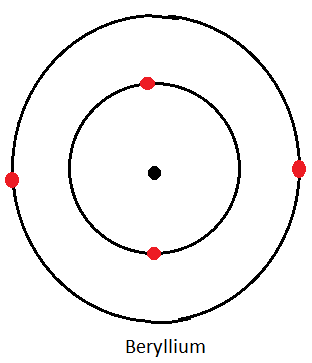
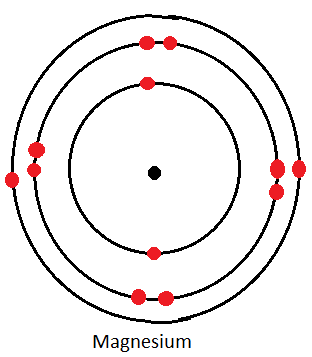
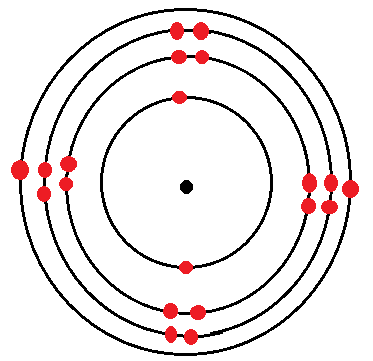 Calcium
Calcium
Gradation in Properties of Alkaline Earth Metals.
Atomic and ionic radius.
Atomic radius:
- Refers to the distance between the centre of the nucleus of an atom and the outermost energy level occupied by electron(s)
Ionic radius:
- Refers to the distance between the centre of the nucleus of an ion and the outermost energy level occupied by electron(s)
Trend:
- The ionic radius and the atomic radius of alkaline earth metals increase down the group.
Reason:
- There is an increase in the number of energy levels down the group from Beryllium to radium.
Illustration:
- Beryllium (2.2) has only 2 energy levels; Magnesium (2.8.2) has 3 energy levels while calcium (2.8.8.2) has 4 energy levels.
- Thus the outermost electron in calcium is further from the nucleus than the outermost electron in magnesium and beryllium.
Atomic and ionic radius of the same element.
- For the same alkaline earth metal the atomic radius is larger than the ionic radius.
Reason:
- Alkaline earth metals form ions by losing the valence electrons, leading to the loss of an entire outermost energy level. Thus the atoms have more energy levels than the corresponding ion hence a larger radius in the atom than in the ion.
Illustration
- Calcium atom has electron arrangement of 2.8.8.2 hence 4 energy levels.
- During ion formation it reacts by losing the 2 valence electrons to acquire a new electron arrangement of 2.8.8 hence 3 energy levels.
- Thus the ion has a smaller radius than the atom.
Diagrammatically: calcium atom and calcium ion
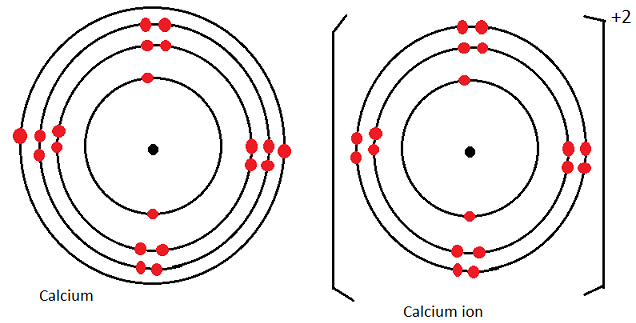
Summary: changes in atomic and ionic radius among alkaline earth metals.
| Element | Symbol | Atomic number | Electron arrangement | Atomic radius (nm) | Ionic radius (nm) |
| Beryllium | Be | 4 | 2.2 | 0.089 | 0.031 |
| Magnesium | Mg | 12 | 2.8.2 | 0.136 | 0.065 |
| Calcium | Ca | 20 | 2.8.8.2 | 0.174 | 0.099 |
Physical Properties of Alkaline Earth Metals.
Appearance.
- Alkaline earth metals acquire a metallic luster when polished. This refers to a shiny appearance on the cut surface.
- They however lose this metallic luster when exposed to air due to oxidation.
Note:
- The purpose of polishing alkaline earth metals before using them in experiment is to remove the oxide coating that slows down and prevents them from reacting.
Ease of cutting.
- Magnesium is hard to cut with a knife; but is however malleable and ductile.
- Calcium cannot also be cut with a knife because it is brittle.
Note:
Malleability: Refers to the ability of a material to be hammered into sheets. Example : Iron sheets are possible to be made because iron metal is malleable.
Ductility: The ability of a material to be rolled into wires. Example: electric cables are made of aluminium because aluminium metal is ductile.
Brittle: Refers to a substance which is hard and likely to break.
They have relatively high melting and boiling points in comparison to alkali metals.
Reason : they have relatively stronger metallic bonds (than alkali metals).
The melting and boiling points decrease down the group.
Reason: The size of the atoms increase down the group due to increasing number of energy levels. As the atomic radius increase the force of attraction between the positive nucleus and the delocalized electrons decrease. This leads to a decrease in the strength of the metallic bonds (down the group).
Electrical conductivity.
- Alkaline earth metals are good conductors of heat and electricity this is because they have delocalized electrons in the outermost energy level.
- The electrical conductivity is similar for all the alkaline earth metals since all alkaline earth metals have the same number of delocalized electron (two valence electrons) in the outermost energy level.
Ionization energy.
- Ionization energy is the minimum energy required to remove an electron from the outermost energy level of an atom in its gaseous state.
- The number of ionization energies an element may have is equivalent to the number of valence electrons.
- Thus alkaline earth metals have two ionization energies.
- The first ionization energy is the minimum energy required to remove the first electron from the outermost energy level of an atom in its gaseous state.
- The second ionization energy is the minimum energy required to remove the second electron from the outermost energy level of an atom in its gaseous state.
- The first and second ionization energies among alkaline earth metals decreases down the group because the effective force of attraction of on the outermost electron by the positive nucleus decreases with increasing atomic size and distance from the nucleus.
- Note that the atomic radius increases down the group due to increase in the number of energy levels.
Variation between 1st and 2nd Ionization energies.
- The first ionization energy is always lower than the second ionization energy for the same element because after losing the first electron from an atom, the overall positive charge holds the remaining electrons more firmly. Thus removing a second electron from the ion requires more energy than the first electron.
Note:
- The third ionization energy will also be higher than the second ionization energy for the same reason.
Example:
Magnesium.
- First ionization energy: Mg(g) → Mg+(g) + e- (1st I.E = 736 kJ per mole)
- Second ionization energy: Mg+(g) → Mg2+(g) + e- (2nd I.E = 1450 kJ per mole)
Summary on physical properties of alkaline earth metals.
| Element |
Atomic number |
Melting point (oC) | Boiling point (oC) | Atomic radius (nm) |
1st I.E (kJmol-1) |
2nd I.E (kJmol-1) |
| Beryllium | 4 | 1280 | 2450 | 0.089 | 900 | 1800 |
| Magnesium | 12 | 650 | 1110 | 0.136 | 736 | 1450 |
| Calcium | 20 | 850 | 1140 | 0.174 | 590 | 1150 |
| Strontium | 38 | 789 | 1330 | 0.210 | 550 | 1060 |
| Barium | 56 | 725 | 1140 | 0.220 | 503 | 970 |
Chemical Properties of Alkaline Earth Metals.
Burning alkaline earth metals in air.
- Alkaline earth metals react burn in air to form corresponding oxides.
- More reactive alkaline earth metals may also react with atmospheric nitrogen to form corresponding nitrides.
Examples:
Magnesium.
- Burns in air with a blinding brilliant flame forming a white solid.
- The white solid is a mixture of magnesium oxide ad magnesium nitride.
Equations:
Reacting with oxygen : 2Mg(s) + O2(g) → 2MgO(s)
Reacting with nitrogen : 3Mg(s) + N2(g) → Mg3N2(s)
Calcium.
- Burns in air with a faint orange flame forming a white solid.
- The white solid is a mixture of calcium oxide ad calcium nitride.
Equations:
Reacting with oxygen : 2Ca(s) + O2(g) → 2CaO(s)
Reacting with nitrogen : 3Ca(s) + N2(g) → Ca3N2(s)
Note:
- The trend in the reactivity of alkaline earth metals when burning in air is not clear; due to the oxide coating on the calcium that tends to slow down the reaction of calcium in air.
- For this reason it is important to polish the surfaces of alkaline earth metals before using them in experiments.
Reaction of alkaline earth metals with cold water.
- Alkaline earth metals react slowly with cold water to form corresponding hydroxides and hydrogen gas.
Examples
Magnesium:
- Reacts slowly with water to form magnesium hydroxide and hydrogen gas.
- The reaction is very slow and the amount of hydrogen gas evolved is very low hence the hydrogen gas bubbles stick on the surface of the metal.
- The magnesium hydroxide formed dissolves slightly in water to form magnesium hydroxide.
- Thus the resultant solution is slightly alkaline.
Equation:
Mg(s) + 2H2O(l) → Mg(OH)2(aq) + H2(g)
Calcium:
Diagram of apparatus:
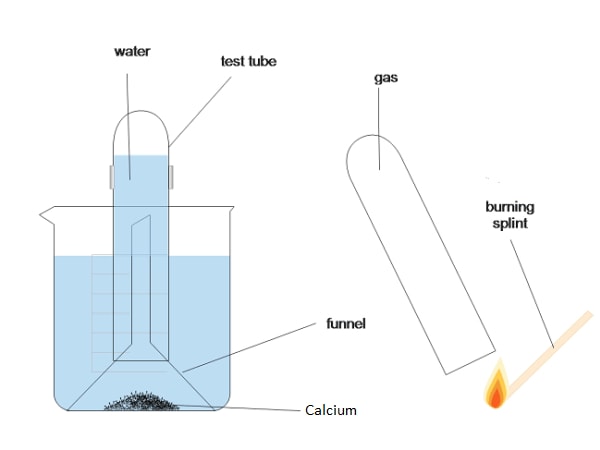
Observations and explanations:
- Calcium sinks to the bottom of the beaker; because it is denser than water;
- Slow effervescence of a colourless gas; due to slow evolution of hydrogen gas;
- Soapy solution formed; due to formation of alkaline calcium hydroxide;
- A white suspension is formed; because calcium hydroxide is slightly soluble in water;
Reaction equation:
Ca(s) + H2O(l) → Ca(OH)2(aq) + H2(g) ;
Effect of resultant solution on litmus paper;
- Litmus paper slowly turns blue; calcium hydroxide formed is slightly soluble in water; releasing a small number of hydroxyl ions which result into alkaline conditions // high pH;
Reaction with steam.
- Alkaline earth metals react with steam to produce corresponding metal oxide and hydrogen gas.
- The reactivity with stem is faster and more vigorous for each alkaline earth metals as compared to reaction with cold water.
Examples:
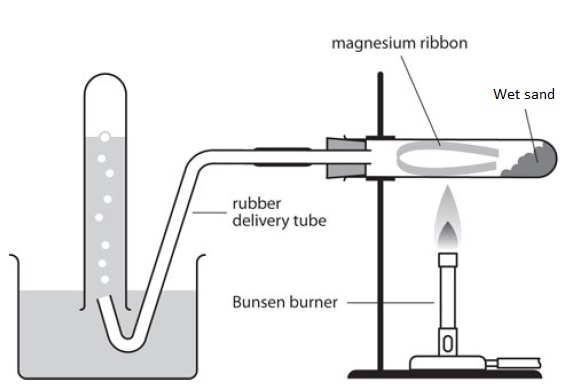
Magnesium
Procedure:
- A small amount of wet sand is put at the bottom of a boiling tube;
- A small piece of magnesium ribbon is cleaned and put in the middle of the combustion tube;
- The wet sand is heated gently first to generate steam that drives out the air that would otherwise react with the magnesium (preventing reaction with steam) then the magnesium ribbon is heated strongly until it glows
- The delivery tube is removed from the water before heating stops to prevent sucking back (of the gas) as the apparatus cools.
- The gas produced is tested using a burning splint.
Diagram of apparatus:
Observations and explanations.
- Magnesium burns with a white blinding flame;
- Grey solid (magnesium) forms a white solid; due to formation of magnesium oxide;
- Evolution of a colourless gas that produces a pop sound when exposed to a burning splint; confirming it is hydrogen;
Reaction equation.
Magnesium + Steam → Magnesium oxide + Hydrogen gas;
Mg(s) + H2O(g) → MgO(s) + H2(g) ;
Calcium
- Reaction between calcium and steam would produce calcium oxide and hydrogen gas.
- However the reaction is too explosive to be done under laboratory conditions.
Reaction with chlorine.
- Alkaline earth metals react with chlorine to form corresponding chlorides as the only products.
- Condition: presence of heat, hence the metal must be heated first.
- Precaution : reaction should be done in a working fume chamber because chlorine gas is poisonous).
Examples:
Magnesium.
Procedure:
- A piece of burning magnesium is lowered into a gas jar containing chlorine.
Observations:
- The metal continues to burn with a brilliant white flame.
- They grey solid forms a white powder.
Explanation.
- Reaction between magnesium and chlorine is exothermic.
- The heat produced keeps the metal burning; thus facilitates the reaction between magnesium and chlorine to form magnesium chloride, which is the white powder.
Equation:
Mg(s) + Cl2(g) → MgCl2(s)
Magnesium
Procedure:
- A piece of burning calcium is lowered into a gas jar containing chlorine.
Observations and explanations.
- The metal burns shortly with an orange flame but soon smolders off.
- There is no steady reaction between calcium and chlorine.
Reason:
- When calcium is heated a coating of the metal oxide is formed first which prevents furtheer reaction between the metal and chlorine.
- However under suitable conditions calcium reacts with chlorine to form a white powder of calcium chloride.
Equation:
Ca(s) + Cl2(g) → CaCl2(s)
Reaction of alkaline earth metals and dilute acids
- Generally alkaline earth metals react with dilute acids to form salts and hydrogen gas.
Examples:
With hydrochloric acid.
Beryllium:
- When a piece of beryllium is dropped into a beaker containing hydrochloric acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of beryllium chloride remains in the test tube // boiling tube.
Equation : Be(s) + 2HCl(aq) → BeCl2(aq) + H2(g)
Magnesium:
- When a piece of magnesium is dropped into a beaker containing hydrochloric acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of magnesium chloride remains in the test tube // boiling tube.
Equation : Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g)
Calcium:
- When a piece of calcium is dropped into a beaker containing hydrochloric acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of calcium chloride remains in the test tube // boiling tube.
Equation : Ca(s) + 2HCl(aq) → CaCl2(aq) + H2(g)
With sulphuric (VI) acid.
Beryllium:
- When a piece of beryllium is dropped into a beaker containing sulphuric (VI) acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of beryllium sulphate remains in the test tube // boiling tube.
Equation : Be(s) + H2SO4(aq) → BeSO4(aq) + H2(g)
Magnesium:
- When a piece of magnesium is dropped into a beaker containing sulphuric (VI) acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of magnesium sulphate remains in the test tube // boiling tube.
Equation : Mg(s) + H2SO4(aq) → MgSO4(aq) + H2(g)
Calcium:
- When a piece of calcium is dropped into a beaker containing sulphuric (VI) acid, there is slight effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- The reaction however quickly stops and there is formation of a white precipitate in the test tube or boiling tube because when calcium reacts with dilute sulphuric (VI) cid there is formation of calcium sulphate which is insoluble hence the formation of a white precipitate. The insoluble calcium sulphate coats the surface of the (calcium) metal stopping further reaction.
Equation : Ca(s) + H2SO4(aq) → CaSO4(s) + H2(g)
With nitric (V) acid.
Beryllium:
- When a piece of beryllium is dropped into a beaker containing nitric (V) acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of beryllium nitrate remains in the test tube // boiling tube.
Equation : Be(s) + 2HNO3(aq) → Be(NO3)2(aq) + H2(g)
Magnesium:
- When a piece of magnesium is dropped into a beaker containing nitric (V) acid, there is effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of magnesium nitrate remains in the test tube // boiling tube.
- Equation : Mg (s) + 2HNO 3(aq) → Mg(NO 3 ) 2(aq) + H 2(g)
Calcium:
- When a piece of calcium is dropped into a beaker containing nitric (V) acid, there is slight effervescence of a colourless gas.
- The colourless gas produces a pop sound when exposed to a burning splint confirming that it is hydrogen gas.
- A colourless solution of calcium nitrate remains in the test tube // boiling tube.
Equation : Ca(s) + 2HNO3(aq) → Ca(NO3)2(aq) + H2(g)
Trend in Reactivity in Alkaline Earth Metals.
- The reactivity of the alkaline earth metals increases down the group because the atomic radius increases down the group due to increasing number of energy levels. The attraction of the two valence electrons towards the positive nucleus thus decreases down the group. Since alkaline earth metals react by losing electrons the ease with which the valence electrons are lost
therefore decreases down the group.
Similarity of Ions and Formulae of Some Compounds of Alkaline Earth Metals.
- Alkaline earth metals have the same valency (2) and a similar charge (2+) on their ions since they all have a single valence electron.
- Thus they have the same general formula in their ions and compounds.
Examples.
| Alkali metal ion | Hydroxide (OH-) | Oxides (O2-) | Chloride (Cl-) | Sulphates (SO42-) |
| Be2+ | Be(OH)2 | BeO | BeCl2 | BeSO4 |
| Mg2+ | Mg(OH)2 | MgO | MgCl2 | MgSO4 |
| Ca2+ | Ca(OH)2 | CaO | CaCl2 | CaSO4 |
Uses of Some Alkaline Earth Metals and their Compounds
- Magnesium is used in the manufacture of magnesium hydroxide which is used as an anti-acid medicine because magnesium hydroxide is a non-toxic base.
- A low-density alloy of magnesium and aluminium is used in aeroplanes construction.
- Hydrated calcium sulphate (plaster of Paris) is used in hospitals to set fractured bones.
- Cement is made by heating a mixture of calcium carbonate (limestone), clay and sand.
- Calcium carbonate is used in the extraction of iron.
- Calcium oxide (quicklime) is added tom acidic soils to raise pH for agricultural purposes.
- Calcium nitrate is used as a nitrogenous fertilizer.
- Magnesium oxide is used in the lining of furnaces.
- Barium sulphate is used in the diagnosis of ulcers.
- Barium nitrate is used to produce the green flame in fireworks.
- Calcium carbonate is mixed with oil to make putty.

The Halogens
- Are the elements with seven valence electrons and hence in group VII of the periodic table.
- All are non-metallic in nature.
- The members of the family in order down the group is as follows:
- Fluorine.
- Chlorine
- Bromine
- Iodine
- Astatine
Electron Arrangement of the First Three Halogens.
| Elements | Electron arrangement |
| Fluorine | 2.7 |
| Chlorine | 2.8.7 |
| Bromine | 2.8.18.7 |
Note: Halogen is a derivative of two Greek words: halo and gen.
Diagram: Part of Periodic Table Showing the Halogens
Gradation in Properties of Halogens.
Atomic and ionic radius.
Atomic radius: Refers to the distance between the centre of the nucleus of an atom and the outermost energy level occupied by electron(s)
Ionic radius: Refers to the distance between the centre of the nucleus of an ion and the outermost energy level occupied by electron(s)
Trend:
- The ionic radius and the atomic radius of halogens increase down the group because there is an increase in the number of energy levels down the group from Fluorine to iodine.
Illustration:
- Fluorine (2.7) has only 2 energy levels; Chlorine (2.8.7) has 3 energy levels while bromine (2.8.18.7) has 4 energy levels.
- Thus the outermost electron in bromine is further from the nucleus than the outermost electron in chlorine and fluorine.
Atomic and ionic radius of the same element.
- For the same halogen the ionic radius is larger than the atomic radius because Halogens form ions by gaining (an) electron into the outermost energy level. This increases the electron-electron repulsion in the outermost energy level hence decreasing the nuclear attraction of the outermost electrons towards the nucleus.
Illustration
- Chlorine atom has electron arrangement of 2.8.7 hence 3 energy levels.
- During ion formation it reacts by gaining an electron into the third energy level to acquire a new electron arrangement of 2.8.8.
- In the atom 17 protons are attracting 17 electrons; while in the chloride ion there are 17 protons attracting 18 electrons in the outermost energy level.
- Thus in the ion the effect of the positive nucleus is lower.
- This is due to increased repulsive effect between the existing electrons in the outermost energy level and the incoming electron (electron-electro repulsion)
Diagrammatically: chlorine atom and chloride ion
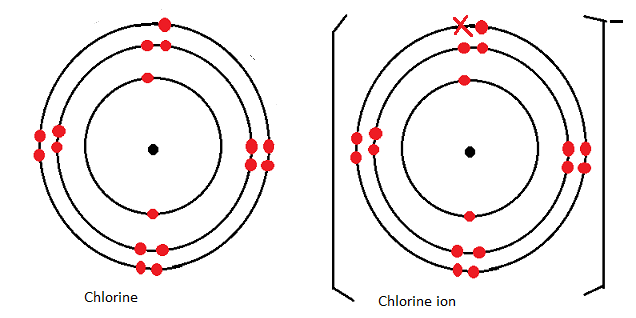
Summary: changes in atomic and ionic radius among alkali metals.
| Element | Symbol | Atomic number | Electron arrangement | Atomic radius (nm) | Ionic radius (nm) |
| Fluorine | F | 9 | 2.7 | 0.064 | 0.136 |
| Chlorine | Cl | 17 | 2.8.7 | 0.099 | 0.181 |
| Bromine | Br | 35 | 2.8.18.7 | 0.114 | 0.195 |
| Iodine | I | 53 | 2.8.18.18.7 | 0.133 | 0.216 |
Laboratory Preparation of Chlorine.
Note: It is usually prepared by oxidation of concentrated hydrochloric acid by removal of hydrogen.
Equation:
2HCl(aq) + [O] → Cl2(g) + H2O(l)
- The [O] is from a substance containing oxygen (an oxidizing agent).
- The main oxidizing agents normally used for preparation of chlorine are:
- Potassium manganate (VII); KMnO4.
- Manganese (IV) oxide; MnO2
Preparation of chlorine from MnO2 and HCl.
Apparatus:
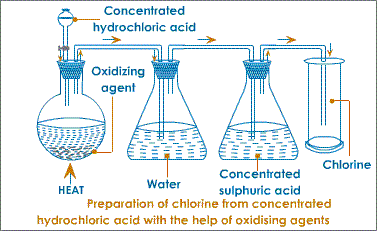
Conditions:
- Heating;
- Presence of an oxidizing agent; in this case it is manganese (IV) oxide.
Procedure:
- Hydrochloric acid is reacted with manganese (IV) oxide (dropwise);
Equation:
MnO2(s) + 4HCl(aq) –Heat→ MnCl2(aq) + 2H2O(l) + Cl2(g)
Explanation:
- Manganese (IV) oxide oxidizes hydrochloric acid by removing hydrogen resulting into chlorine.
- The manganese (IV) oxide is reduced to water and manganese chloride.
- The resultant chlorine gas is passed through a bottle containing water to remove hydrogen chloride fumes (gas) which is very soluble in water.
- Next it is passed through concentrated sulphuric acid or anhydrous calcium chloride; to dry the gas.
Collection:
- Wet chlorine is collected over brine (saturated sodium chloride solution) or hot water because It does not dissolve in brine and is less soluble in water.
- Dry chlorine is collected by downward delivery (upward displacement of air) because it is denser than air (2.5 times).
Note: Chlorine may also be dried by adding calcium chloride to the jar of chlorine. - The first bottle must contain water and the second concentrated sulphuric acid. If the gas is first passed through concentrated sulphuric acid in the first bottle then to the water; it will
be made wet again.
Physical Properties of Halogens.
Physical state and appearance at room temperature.
| Halogen | State and appearance |
| Fluorine | Pale yellow gas |
| Chlorine | Green-yellow gas |
| Bromine | Volatile brown liquid |
| Iodine | Shiny dark grey solid. |
Solubility
In water
Experiment: To investigate solubility of halogens in water.
Procedure:
- A boiling tube containing chlorine gas is inverted into a beaker containing water.
- The experiment is repeated with fluorine, bromine and a few crystals of iodine.
Observations:
Fluorine:
- The level of solution rises in the boiling tube.
- The pale yellow colour of fluorine disappears.
Chlorine:
- The level of solution rises in the boiling tube.
- The green-yellow colour of chlorine disappears.
Bromine
- The level of solution rises in the boiling tube.
- The brown colour of bromine disappears.
Iodine:
- The level of solution remains the same in the boiling tube.
- The shiny dark grey crystals remain in the beaker.
- The rise in water level is higher in fluorine than in chlorine while the rise in chlorine is higher than in bromine.
Explanations:
- Fluorine, chlorine and bromine are all soluble in water, while iodine is insoluble in water.
- When a boiling tube containing the soluble halogens is inverted into a beaker containing water, the halogen dissolves in the water.
- This creates a partial vacuum and the water in the beaker thus rises to occupy the space left by the dissolved gas.
- The halogens dissolve in water to form acidic solutions.
- The more soluble the halogen, the higher the rise in water level in the boiling tube.
- Thus the solubility of halogens decreases down the group from fluorine to bromine.
Reason:
- All halogens have molecular structures with wreak van der waals forces between the molecules.
- During the dissolution the Van der Waals must be broken. The strength of Van der Waals increase as the atomic size and hence the molecular size increases which occurs down the group.
In tetrachloromethane.
- The same procedure (of dissolving halogens in water) is followed with tetrachloromethane.
Observations:
- All halogens are soluble in tetrachloromethane.
- The solubility of each halogen is higher in tetrachloromethane than in water, since Halogens are molecular thus non-polar and thus are more soluble in polar organic solvents like tetrachloromethane than in polar solvents like water.
Effect of heat
- Fluorine and chlorine are gases at room temperature and simply expand and increase in volume when heated.
- Bromine is a brown liquid at room temperature and evolves brown fumes when heated.
- Iodine exists as shiny dark grey solid at room temperature and sublimes to give brown fumes when heated.
Electrical conductivity.
- All halogens are non conductors of heat and electricity because they are molecular and thus have neither delocalized electrons nor free mobile ions for electrical conductivity.
Trend in melting and boiling points.
- The melting and boiling points of halogens increase down the group because Halogens exist as diatomic molecules and thus have molecular structures;
- The molecules are held together by intermolecular forces called the Van der Waals which have to be broken during melting and boiling;
- The strength of the Van der Waals increases as the size of the atoms and hence molecules increases which occurs down the group.
Summary on some physical properties of halogens
| Halogen | Formula | Atomic number | Appearance | Melting point (oC) | Boiling point (oC) |
| Fluorine | F | 9 | Pale yellow gas | -238 | -188 |
| Chlorine | Cl | 17 | Green yellow gas | -101 | -35 |
| Bromine | Br | 35 | Brown liquid | -7 | 59 |
| Iodine | I | 53 | Shiny dark grey solid | 114 | 184 |
Chemical Properties of Halogens.
Note: It is not easy for non-metals like halogens to form cations because the ionization energy (amount of energy required to lose an electron(s) from the outermost energy level of a gaseous atom) is very large.
Thus most non-metals react by forming anions (negatively charged ions) by electron loss.
Ion formation.
- Halogens react by gaining a single electron into the outermost energy level to form a stable electron configuration and corresponding anions.
- During ion formation by electron loss energy is released, and the energy change for this process is called electron affinity.
Note: Definition: Electron Affinity is thus the energy released when an atom in gaseous state gains an electron to form a negative ion. (anion)
Trend in electron affinity of halogens:
- Generally the electron affinity of halogens decreases down the group since the size of the atoms increases (due to increasing number of energy levels) down the group and thus
the force of attraction of the electrons in the outermost energy level towards the nucleus decreases. - Thus down the group the ease with which electrons are gained decreases and the faster the ease of electron gain, the more the energy released hence the more the electron affinity.
Summary:
| Element | Ionization equation | Electron affinity (kJ per mole) |
| Fluorine | F (g) + e → F - (g) | -322 |
| Chlorine | Cl (g) + e → Cl - (g) | -349 |
| Bromine | Br (g) + e → Br - (g) | -325 |
| Iodine | I (g) + e → I - (g) | -295 |
Reaction with metals.
Chlorine.
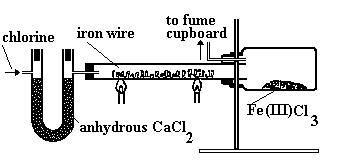
Reaction of chlorine with iron.
Apparatus.
Precaution.
- Experiment should be done in a functional fume cupboard or in the open since Chlorine gas is poisonous and will thus be harmful to the human body.
Procedure:
- A stream of dry chlorine gas is passed over heated iron wool as per the diagram.
Conditions.
- Chlorine gas has to be dry (done by the anhydrous calcium chloride in the U-tube) to prevent hydration hence oxidation of iron (which will then form Fe2O3.5H2O) hence preventing
reaction between iron and chlorine. - Iron metal must be hot ; and this is done by heating to provide activation energy i.e. the minimum kinetic energy which the reactants must have to form products.
- Anhydrous calcium chloride In the U-tube; to dry the chlorine gas. In the thistle funnel; to prevent atmospheric water vapour (moisture) from getting into the
apparatus and hence reacting with iron (III) chloride.
Note : In the guard tube, calcium oxide is preferable to anhydrous calcium chloride because Other than preventing atmospheric water vapour from getting into the flask with iron wool; it also absorbs excess chlorine thus preventing environmental pollution
Observations:
- Iron metal glows red-hot.
- Red brown fumes (FeCl3(g)) are formed in the combustion tube.
- A black solid (FeCl3(s)) is collected in the flask.
- Iron (III) chloride cannot be easily collected in the combustion tube since It sublimes when heated and hence the hotter combustion tube causes it to sublime and its vapour is
collected on the cooler parts of the flask.
Reaction equation.
2Fe(s) + 3Cl2(g) → 2FeCl3(g)
Conclusion.
- Iron (III) chloride sublimes on heating; the black solid changes to red-brown fumes on heating.
Equation:
FeCl3(s)(black) ⇌ FeCl3(g)(Red-brown)
Reaction with aluminium.
- Chlorine reacts with aluminium in a similar manner to iron to from aluminium chloride.
Equation:
2Al(s) + 3Cl2(g) → 2AlCl3(s)
2Al(s) + 3Cl2(g) → Al2Cl6(s)
- Aluminium chloride also sublimes on heating.
Equation:
AlCl3(s) ⇌ AlCl3(g)
(White) (White)
Reaction with burning magnesium.
Procedure:
- Burning magnesium is lowered into a gar jar of chlorine gas.
Observations:
- The magnesium continues to burn with a bright blinding flame ;
- Formation of white fumes (MgCl2); which cools into a white powder.
Equation:
Mg(s) + Cl2(g) → MgCl2(s)
- Generally chlorine reacts with most metals when hot to form corresponding chlorides.
Note:
Where a metal forms two chlorides when it reacts with chlorine, the higher chloride is usually formed because the higher chloride is stable. This explains why reactions of chlorine with iron results into iron (III) chloride and not iron (II) chloride.
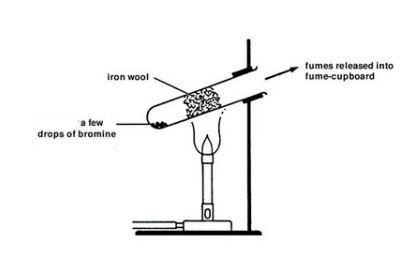
Bromine.
Reaction of bromine with iron
Apparatus
Precaution.
- Experiment should be done in a functional fume cupboard or in the open since Bromine gas is poisonous and will thus be harmful to the human body.
Procedure:
- Bromine liquid is heated to generate bromine vapour (fumes).
- The iron wool is then heated and a stream of the bromine gas is passed over heated iron wool as per the diagram.
Conditions.
- Bromine must be heated to generate fumes before heating the iron, so that bromine vapour will drive out air from the boiling tube to prevent oxidation of iron with oxygen which would otherwise prevent reaction between iron and bromine.
- Iron metal must be hot ; and this is done by heating. To provide activation energy i.e. the minimum kinetic energy which the reactants must have to form products.
Observations:
- Brown fumes of bromine are produced on heating bromine liquid.
- The iron wool glows red-hot upon heating
- The iron wool forms dark red crystals (of iron (III) bromide)
Reaction equation.
Word : Iron + bromine → Iron (III) bromide.
Chemical: 2Fe(s) + 3Br2(g) → 2FeBr3(s)(Dark red crystals.)
Reaction of bromine with zinc
- Using the same set up bromine also reacts with zinc to form zinc bromide.
Equation
Zn(s) + Br2(g) → ZnBr2(s)
Reaction of bromine with magnesium
- Using the same set up bromine also reacts with zinc to form magnesium bromide.
Equation
Mg(s) + Br2(g) → MgBr2(s)
Reaction of bromine with sodium
- Using the same set up bromine also reacts with zinc to form sodium bromide.
Equation
2Na(s) + Br2(g) → 2NaBr(s)
Iodine.
Reaction of iodine with iron
Apparatus
(same as bromine above)
Precaution.
- Experiment should be done in a functional fume cupboard or in the open since Iodine is poisonous and will thus be harmful to the human body.
Procedure:
- Iodine crystals are heated to generate iodine vapour (fumes).
- The iron wool is then heated and a stream of the iodine gas is passed over heated iron wool as per the diagram.
Conditions.
- Iodine must be heated to generate fumes before heating the iron so that iodine vapour will drive out air from the boiling tube to prevent oxidation of iron with oxygen which would otherwise prevent reaction between iron and iodine.
- Iron metal must be hot ; and this is done by heating to provide activation energy i.e. the minimum kinetic energy which the reactants must have to form products.
Observations:
- Purple vapour (fumes) of iodine is produced on heating iodine crystals.
- The iron wool glows red-hot upon heating
- The iron wool forms grey black crystals (of iron (II) iodide)
Reaction equation.
Word : Iron + iodine → Iron (II) iodide.
Chemical: Fe(s) + I2(g) → FeI2(s) Grey-black crystals.
Reaction of Iodine with zinc
- Using the same set up bromine also reacts with zinc to form zinc iodide.
Equation:
Zn(s) + I2(g) → ZnI2(s)
Reaction of Iodine with magnesium
- Using the same set up bromine also reacts with zinc to form magnesium iodide.
Equation:
Mg(s) + I2(g) → MgI2(s)
Reaction of Iodine with sodium
- Using the same set up bromine also reacts with zinc to form sodium iodide.
Equation:
2Na(s) + I2(g) → 2NaI(s)
- The reactivity of chlorine with metals is more vigorous than that of bromine, which is more than that of iodine.
- Thus reactivity of halogens with metals decreases down the group since Halogens react by gaining electrons; the ease of gaining electrons decrease down the group as the atomic size increases which leads to progressive decrease in the force of attraction of electrons in the outermost energy levels by the positively charged nucleus.
Reaction with water.
- Halogens that dissolve in water form a mixture of two acids.
Reaction of chlorine with water
- Chlorine dissolves in water to form chlorine water, which is a mixture of two acids: chloric (I) acid and hydrochloric acid.
Equation:
Cl2(g) + H2O(l) → HOCl(aq) + HCl(aq)
Effects of sunlight on chlorine water.
Procedure:
- Chlorine water is made by dissolving the gas in water.
- A long tube filled with chlorine water is inverted over a beaker containing water.
- It is then exposed to sunlight (bright light).
Observations:
- After sometime a gas collects in the tube and on applying a glowing splint, the splint is rekindles showing that the gas collected is oxygen.
Explanation:
- Chlorine water has two components.
Equation:
Cl2(g) + H2O(l) → HCl(aq) + HOCl(aq)
- The HOCl being unstable will dissolve on exposure to sunlight, giving out oxygen.
Equation:
2HOCl(aq) → 2HCl(aq) + O2(g) (slow reaction)
Overall reaction:
2H2O(l) + 2Cl2(g) → 4HCl(aq) + O2(g)
Effect of chlorine water on litmus papers
Procedure:
- A strip of blue and a strip of red litmus papers are dropped into chlorine water in a beaker.
Observations:
- The blue litmus paper turns red; then both litmus papers are decolourised.
Explanations.
- Chlorine water contains a mixture of chloric (I) acid and hydrochloric acid.
Equation :
Cl2(g) + H2O(l) → HCl(aq) + HOCl(aq)
- The two acids cause the litmus paper to turn red (from blue) while the red litmus paper remained red.
- The litmus papers are then decolourised due to bleaching action of chlorine water, which is due to the activity of chloric (I) acid.
- The chloric (I) acid is unstable and thus decomposes to give hydrochloric acid and oxygen atom.
Equation
HOCl(aq) → HCl(aq) + [O]
- The oxygen atom combines with the chemical of the natural dye in the litmus thus decolourising it.
Equation:
Dye (Coloured) + HOCl(aq) → HCl(aq) + (Dye (Decolourised) + [O])
Note: solutions of bromine and fluorine in water will behave in a similar manner.
Fluorine dissolving in water
F2(g) + H2O(l) → HF(aq) + HOF(aq)
- Hydrofluoric acid Fluoric (I) acid
- The mixture is flourine water
Bromine dissolving in water
Br2(g) + H2O(l) → HBr(aq) + HOBr(aq)
- Hydrobromic acid Bromic (I) acid
- The mixture is bromine water.
Some Uses of Halogens and their Compounds.
- Fluorine is a raw material in the preparation of a synthetic fibre known as polytetrafluoroethane.
- Some compounds of fluorine are added to water and some tooth pastes in small quantities to reduce tooth decay.
- Fluorine is used in the manufacture of hydrogen fluoride used to engrave words or pictures on glass.
- Chlorine is used to make bleaches used in paper and textile industries.
- Chlorine is added to water to kill micro-organisms in water treatment works.
- Chlorine is used in the manufacture of a plastic known as polyvinylchloride (PVC).
- Chlorine is used in large scale manufacture of hydrochloric acid.
- Bromine is used in the manufacture of silver bromide which is used to make the light sensitive photographic paper and films.
- A solution of iodine in alcohol (tincture of iodine) is used as a disinfectant.

The Noble Gases.
- Are the elements with the maximum possible number of valence electrons and hence in group VIII of the periodic table.
- All are gaseous in nature.
- The members of the family in order down the group is as follows:
- Helium
- Neon
- Argon
- Krypton
- Xenon
- radon
- They are found as free atoms in nature and form about 1% of air with argon being the most abundant.
Electron Arrangement of the First Three Noble Gases.
| Elements | Electron arrangement |
| Helium | 2 |
| Neon | 2.8 |
| Argon | 2.8.8 |
- Helium with only two electrons has one occupied energy level; which is completely full and hence is said to have a duplet electron configuration.
- The rest of the noble gases have eight electrons in their outermost occupied energy level hence are said to have the octet electron configuration.
Diagram: Part of Periodic Table Showing the Noble Gases
Dot and Cross Diagrams for the First Three Noble Gases
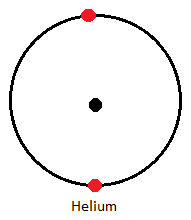
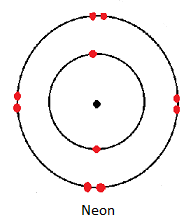
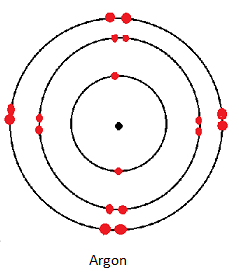
Physical Properties of Noble Gases
Atomic and ionic radius.
- The atomic radius of noble gases increase down the group since there is an increase in the number of energy levels down the group from Helium to radon.
- Helium (2.) has only 1 energy level; Neon (2.8) has 2 energy levels while Argon (2.8.8) has 3 energy levels.
- Thus the outermost electron in Argon is further from the nucleus than the outermost electron in neon and helium.
- Under normal circumstance the noble gases neither lose nor gain electrons and are thus stable hence unreactive.
- Due to this they rarely form ions and hence comparative studies of ionic radii among noble gases are not feasible.
Ionization energies.
- Noble gases have very high ionization energies. They are colourless monoatomic gases with very stable electron arrangements, either dupltet (helium) or octet (the rest).
- The nuclear attraction force of electrons in the outermost energy level towards the nucleus is thus very strong.
- The ionization energy decrease down the noble gases group as the size of the atoms increase down the group (due to increase in the number of energy levels), the force with which the electrons in the outermost energy levels are attracted towards the nucleus decrease hence increase in the ease of electron loss (from the outermost energy level).
Melting and boiling points.
- Generally noble gases have very low melting and boiling points since they exist as monoatomic gases with simple molecular structures with weak van der waals forces that are easy to break.
- Melting and boiling points among the noble gases increase down the group.
- Noble gases exist as monoatomic molecules and thus have simple molecular structures;
- The molecules (atoms) are held together by intermolecular (inter-atomic) forces called the Van der Waals which have to be broken during melting and boiling;
- The strength of the Van der Waals increases as the size of the atoms and hence molecules increases which occurs down the group.
Summary: changes in atomic and ionic radius among alkali metals.
| Element | Symbol | Atomic number | Atomic radius (nm) | 1st Ionization energy (kJmol-1 ) | Melting point (oC) | Boiling point (oC) |
| Helium | He | 2 | 0.128 | 2372 | -270 | -269 |
| Neon | Ne | 10 | 0.160 | 2080 | -249 | -246 |
| Argon | Ar | 18 | 0.192 | 1520 | -189 | -186 |
| Krypton | Kr | 36 | 0.197 | 1350 | -157 | -152 |
| Xenon | Xe | 54 | 0.217 | 1170 | -112 | -108 |
Chemical Properties of Noble Gases.
- Generally the noble gases neither lose nor gain electrons and are thus stable hence unreactive.
- Due to this they rarely form ions and hence have no feasible chemical reactions.
- However xenon and radon with very large atomic radii and smaller ionization energies take part in some reactions and thus display some chemical properties.
Uses of Some Noble Gases.
Note: The application of noble gases is centered on their inert nature.
- Argon is used in light bulbs to provide an inert environment to prevent oxidation.
- Argon is used as an insulator in arch-welding.
- Neon gas is used in streets and advertisement lights.
- Helium mixed with oxygen is used in deep sea diving and mountaineering.
- Helium mixed with oxygen also is used in hospitals for breathing in patients with respiratory problems and those undergoing certain forms of surgery.
- Helium can be used instead of hydrogen in balloons for meteorological research because Hydrogen is explosive in the presence of air and may explode causing serious accidents. Helium is less dense than hydrogen.
- Helium is used in thermometers for measurements of very low temperatures.
- Liquid helium is used to keep certain metal alloys at temperatures low enough for them to become superconductors.
Download CHEMICAL FAMILIES: PATTERNS IN PROPERTIES - Chemistry Notes Form 2.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students