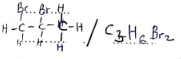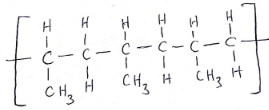INSTRUCTIONS TO CANDIDATES
- Answer ALL questions in the spaces provided.
- Mathematical tables and silent electronic calculators may be used.
- All workings MUST be clearly shown where necessary.
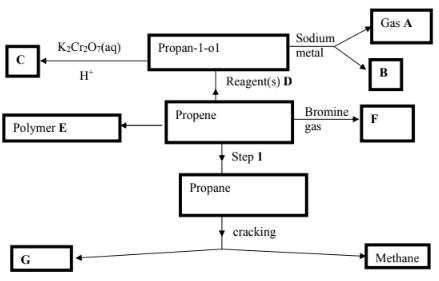
- The scheme below shows a series of reactions and compounds. Study it and use it to answer the questions that follow.
- Identify the following compounds and products (6marks)
A……………………………………………………
B……………………………………………………
C……………………………………………………
E……………………………………………………
F……………………………………………………
G…………………………………………………… - State 2 conditions for step 1 to occur. (1 mark)
- Write an equation for the formation compound F. (1 mark)
- Identify reagent(s) D. (1 mark)
- State one industrial use of methane. (1 mark)
- Name the following organic compounds. (3 marks)
- C3H4
- CH3CH2 CH2 CH CH2 CH3
│
OH - CH2 = C – CH3
│
CH3
- Draw the structure of a section of polymer E showing three repeat units. (1 mark).
- Identify the following compounds and products (6marks)
- The table below shows the elements in period 3 of the periodic table. Study it and answer the questions that follow.
Element Na Mg Al Si P S Cl Ar - Write the formulae of two oxides for each of the following: (2 marks)
- Sodium: Oxide I ………………… Oxide II ………………
- Sulphur: Oxide I ………………… Oxide II …………………
- The products of the reaction between phosphorus and chlorine depend on the conditions used. Write the equation for the reaction when phosphorus reacts with limited phosphorus. (1 mark)
- Identify the most electronegative element. Give a reason. (2 marks)
- State and explain the differences in the boiling points of:
- Magnesium oxide and sulphur (IV) oxide. (2 marks)
- Sulphur and phosphorus. (2 marks)
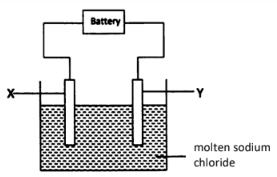
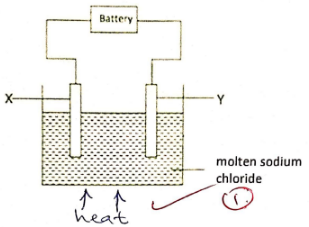
- The diagram below shows the electrolysis of the chloride of sodium.
- On the diagram, indicate the missing condition. (1 mark)
- During the electrolysis, chlorine gas was formed at electrode Y. Identify the:
- Anode………………………………………………(1 mark)
- Cathode……………………………………………(1 mark)
- Write the half equation for the reaction taking place at the:
- Anode. (1 mark) ……………………………………………
- Cathode. (1 mark) …………………………………………
- Write the formulae of two oxides for each of the following: (2 marks)
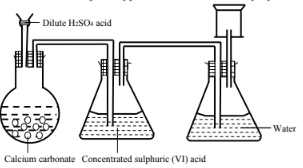
- A student set up the apparatus shown below to prepare and collect dry carbon (IV) oxide gas.
- State a correction for three mistakes in the set up above (3 marks)
- Give two reasons why carbon (IV) oxide is used as a fire extinguisher (1 mark)
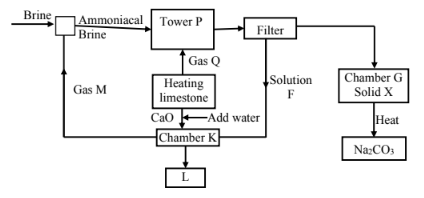
- The flow chart below is for the manufacture of sodium carbonate by the Solvay process. Use it to answer the questions that follow.
- Name:
Gas M …………………… Gas Q ……………………. (1 mark)
Solution F ………………… Solid X ……………………. (1 mark) - Name the product L formed and give one of its uses. (2 marks)
- Write equations of the reactions in: (2 marks)
Tower P (Overall equation)
Chamber K - Name the two raw materials required in the manufacture of sodium carbonate (1 mark)
- Name:
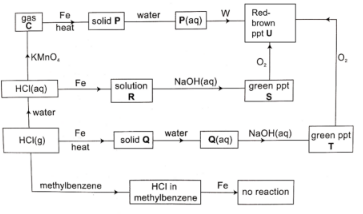
- Study the flow chart below and answer the questions that follow.
- Identify:
- Gas C ………………………………( ½ mark)
- Solid Q ……………………………( ½ mark)
- Solid P ……………………………( ½ mark)
- From the flow chart, give two solutions that contain the same metallic ions. (1 mark)
- Give two precipitates in the flow chart that are the same and name them. (3 marks)
Precipitates: ……………………………………………
Name: ………………………………………………… - Explain the difference between HCl (g) in water and in methylbenzene as shown in the flow chart. (2 marks)
- Name reagent W. ( ½ mark)
- Write the equations for:
- The formation of solid P. (1 mark)
- The formation of solid Q. (1 mark)
- In the preparation of a bleaching agent (sodium hypochlorite), excess chlorine gas was bubbled into 15 litres of cold 2M sodium hydroxide.
- Write the equation for the reaction between chlorine gas and cold dilute sodium hydroxide. (1 mark)
- Calculate the mass in kilograms of sodium hypochlorite produced. (Na = 23.0, Cl = 35.5, O = 16.0) (3 marks)
- Identify:
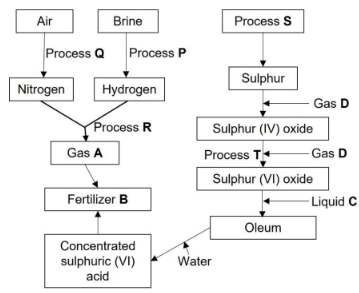
- Study the flow chart below and answer the questions that follow.
- Name the following: (3 marks)
- Process Q …………………
- Process R ………………………
- Process S ……………………
- Process T ………………………
- Gas A ……………………………
- Fertilizer B ………………………
- Why is liquid C used instead of water? (1 mark)
- Write the formula of oleum. (1 mark)
- Write the equation for the formation of:
- fertilizer B. (1 mark)
- gas A. (1 mark)
- Name the catalyst and give the conditions for:
- Process R. (3 marks)
Catalyst …………………………………
Conditions ……………………………… - Process T. (3 marks)
Catalyst ……………………………………
Conditions …………………………………
- Process R. (3 marks)
- Explain how nitrogen is obtained from air using process Q. (3 marks)
- Name the following: (3 marks)
- The following is a procedure that was used to obtain the solubility of a salt Q in water at 25°C. Study it and answer the questions that follow.
Salt Q was dissolved in warm distilled water until no more could dissolve. The mixture was then cooled to 25°C and allowed to settle. A dry evaporating dish and dry watch glass were weighed. Some of the solution was decanted into the dish, covered with the watch glass, and then weighed.
The solution was evaporated to dryness over a small flame. This residue, the dish and the watch glass were weighed. The residue was then heated repeatedly until a constant mass was obtained. The results below were obtained.
Mass of dish + Watch glass = 50.60g
Mass of solution + dish + watch glass = 80.6g
Mass of residue + dish + watch glass = 62.60g- Use the data to answer the questions that follow.
- What is the purpose of the watch glass in such an experiment? (1 mark)
- Why should the heating be continued until a constant mass is obtained? (1 mark)
- Calculate the mass of the solution. (1 mark)
- Calculate the mass of the residue. (1 mark)
- Calculate the mass of the water. (1 mark)
- Calculate the solubility of salt Q in g per 100g of water at 25°C. (2 marks)
- Hard water has both advantages and disadvantages. Give one advantage and one disadvantage of using hard water. (2 marks)
- Using an equation, explain how addition of sodium carbonate is used to remove water hardness. (2 marks)
- Use the data to answer the questions that follow.

MARKING SCHEME
- The scheme below shows a series of reactions and compounds. Study it and use it to answer the questions that follow.
- Identify the following compounds and products (6marks)
A - Hydrogen/H2
B - Sodium propoxide/ CH3CH2CH2O Na
C - Propanoic acid/ CH2CH2COOH
E - Polypropene/
F - 1,2- dibromopropane/
G - ethene/C2H4 - State 2 conditions for step 1 to occur. (1 mark)
- 150°C - 250°C
- Nickel/Palladium catalyst
- Write an equation for the formation compound F. (1 mark)
- C3H6 + Br2 → C3H6Br2
- Identify reagent(s) D. (1 mark)
- Steam (H2O(g)) or concentrated H2SO4 and water
- State one industrial use of methane. (1 mark)
- Used as a fuel
- Name the following organic compounds. (3 marks)
- C3H4 Propyne
- CH3CH2 CH2 CH CH2 CH3
│
OH Hexan-3-ol - CH2 = C – CH3
│
CH3 2-methylprop-2-ene
- Draw the structure of a section of polymer E showing three repeat units. (1 mark).
- Identify the following compounds and products (6marks)
- The table below shows the elements in period 3 of the periodic table. Study it and answer the questions that follow.
Element Na Mg Al Si P S Cl Ar - Write the formulae of two oxides for each of the following: (2 marks)
- Sodium: Oxide I Na2O Oxide II Na2O2
- Sulphur: Oxide I SO2 Oxide II SO3
- The products of the reaction between phosphorus and chlorine depend on the conditions used. Write the equation for the reaction when phosphorus reacts with limited phosphorus. (1 mark)
- 2P(s) + 3Cl2(g) → 2PCl3(s) OR P4(s) + 3Cl2(g) → 4PCl3(s)
- Identify the most electronegative element. Give a reason. (2 marks)
- Cl or chlorine
It most readily gains electrons
- Cl or chlorine
- State and explain the differences in the boiling points of:
- Magnesium oxide and sulphur (IV) oxide. (2 marks)
- Magnesium oxide has higher boiling point than sulphur (IV) oxide
- Magnesium oxide has strong ionic bonds in a giant ionic structure that requires more energy to break than sulphur(IV)oxide which has weak vander waals forces between molecules.
- Sulphur and phosphorus. (2 marks)
- Sulphur has a higher boiling point than phosphorus.
- Sulphur has a larger S8 molecule hence more intermolecular forces of attraction than (P4) Phosphorus molecules. More energy is required to break the bonds between Sulphur molecules than phosphorus molecules.
- Magnesium oxide and sulphur (IV) oxide. (2 marks)
- The diagram below shows the electrolysis of the chloride of sodium.
- On the diagram, indicate the missing condition. (1 mark)
- During the electrolysis, chlorine gas was formed at electrode Y. Identify the:
- Anode Y (1 mark)
- Cathode X (1 mark)
- Write the half equation for the reaction taking place at the:
- Anode. (1 mark) 2Cl−(l) → Cl2(g) + 2e−
- Cathode. (1 mark) 2Na+(l) + 2e− → Na(s)
- On the diagram, indicate the missing condition. (1 mark)
- Write the formulae of two oxides for each of the following: (2 marks)
- A student set up the apparatus shown below to prepare and collect dry carbon (IV) oxide gas.
- State a correction for three mistakes in the set up above (3 marks)
- Dip the thistle funnel ibnto the solution in the flask.
- Remove the conical flask with water.
- Collect the gas using downward delivery or upward displacement.
- Give two reasons why carbon (IV) oxide is used as a fire extinguisher (1 mark)
- It is denser than air hence covers the fire.
- It does not support combustion extinguishing the fire.
- The flow chart below is for the manufacture of sodium carbonate by the Solvay process. Use it to answer the questions that follow.
- Name:
Gas M - Ammonia Gas Q - Carbon(IV) Oxide (1 mark)
Solution F - Ammonium chloride Solid X - Sodium hydrogen carbonate (1 mark) - Name the product L formed and give one of its uses. (2 marks)
Name: Calcium chloride
Use:- Used in road surfacing
- Used ib de-icing of roads
- Used in extraction of sodium from rock salt by electrolysis
- Write equations of the reactions in: (2 marks)
Tower P (Overall equation)
NaCl(aq) + NH3(g) + H2O(l) + CO2(g) → NaHCO3(s) + NH4Cl(aq)
Chamber K
2NaHCO3(s) → Na2CO3(s) + H2O(l) + CO2(g) - Name the two raw materials required in the manufacture of sodium carbonate (1 mark)
- Ammonia
- Brine
- Limestone
- Coke
- Name:
- State a correction for three mistakes in the set up above (3 marks)
- Study the flow chart below and answer the questions that follow.
- Identify:
- Gas C - Chlorine/Cl2 ( ½ mark)
- Solid Q - Iron (II) chloride/ FeCl2 ( ½ mark)
- Solid P - Iron (III) chloride/ FeCl3 ( ½ mark)
- From the flow chart, give two solutions that contain the same metallic ions. (1 mark)
- Solution Q
- Solution R
- Give two precipitates in the flow chart that are the same and name them. (3 marks)
Precipitates:Green ppt S and green ppt T
Name: Iron (II) hydroxide - Explain the difference between HCl (g) in water and in methylbenzene as shown in the flow chart. (2 marks)
- When HCl(g) dissolves in water it ionises to H+ and Cl− where H+ exhibit acidic properties such as reacting with iron. However, when HCl (g) dissolves in methylbenzene, it does not ionise and does not exibit acidic properties hence no reaction with iron metal.
- Name reagent W. ( ½ mark)
- Sodium hydroxide soltion/ Ammonia solution
- Write the equations for:
- The formation of solid P. (1 mark)
- 2Fe(s) + 3Cl2(g) → 2FeCl3(g)
- The formation of solid Q. (1 mark)
- Fe(s) + 2HCl(g) → FeCl2(g) + H2(g)
- The formation of solid P. (1 mark)
- In the preparation of a bleaching agent (sodium hypochlorite), excess chlorine gas was bubbled into 15 litres of cold 2M sodium hydroxide.
- Write the equation for the reaction between chlorine gas and cold dilute sodium hydroxide. (1 mark)
- 2NaOH(aq) + Cl2(g) → NaOCl(aq) + NaCl(aq) + H2O(l)
- Calculate the mass in kilograms of sodium hypochlorite produced. (Na = 23.0, Cl = 35.5, O = 16.0) (3 marks)
Moles of NaOH used
2 moles → 1000cm3
? → 15000cm3
15000 × 2 = 30 moles
1000
M.R NaOH : NaOCl
2 : 1
30 moles : ?
30 × 1 = 15 moles of NaOCl
2
R.F.M = 23 + 16 + 35.5 = 74.5
Mass = 15 × 74.5 = 1117.5g
1117.5 = 1.1175kg (−½ if no units or wrong units)
1000
- Write the equation for the reaction between chlorine gas and cold dilute sodium hydroxide. (1 mark)
- Identify:
- Study the flow chart below and answer the questions that follow.
- Name the following: (3 marks)
- Process Q - Fractional distillation
- Process R - Haber process
- Process S - Frasch process
- Process T - Contact process
- Gas A - Ammonia
- Fertilizer B - Ammonium sulphate
- Why is liquid C used instead of water? (1 mark)
- If water is used SO3 will dissolve producing a lot of heat causing the acid to boil/ forming acid fumes that are harmful to factory workers
- Write the formula of oleum. (1 mark)
- H2S2O7
- Write the equation for the formation of:
- fertilizer B. (1 mark)
- 2NH3(g) + N2SO4(l) → (NH4)2SO4(s)
- gas A. (1 mark)
Fe catalyst
- N2(g) + 3H2(g)
2NH3(g)
- N2(g) + 3H2(g)
- fertilizer B. (1 mark)
- Name the catalyst and give the conditions for:
- Process R. (3 marks)
Catalyst - Finely divided iron
Conditions - 400°C - 500°C and 200 - 500 atmospheres - Process T. (3 marks)
Catalyst - Vanadium (V) Oxide / Platinum catalyst
Conditions - 450°C and 2 - 3 atmospheres
- Process R. (3 marks)
- Explain how nitrogen is obtained from air using process Q. (3 marks)
- Air is passed through electrostatic filters to remove dust. Dust free air is passed through concentrated sodium hydroxide to remove carbon (IV) oxide. The air is then cooled to -25°C to remove water vapour. The remaining part of air is repeatedly compressed and allowed to expand rapidly to cool it to -200°C. The liquid air is passed through a fractional distillation tower where nitrogen is collected as the first fraction at -196°C
- Name the following: (3 marks)
- The following is a procedure that was used to obtain the solubility of a salt Q in water at 25°C. Study it and answer the questions that follow.
Salt Q was dissolved in warm distilled water until no more could dissolve. The mixture was then cooled to 25°C and allowed to settle. A dry evaporating dish and dry watch glass were weighed. Some of the solution was decanted into the dish, covered with the watch glass, and then weighed.
The solution was evaporated to dryness over a small flame. This residue, the dish and the watch glass were weighed. The residue was then heated repeatedly until a constant mass was obtained. The results below were obtained.
Mass of dish + Watch glass = 50.60g
Mass of solution + dish + watch glass = 80.6g
Mass of residue + dish + watch glass = 62.60g- Use the data to answer the questions that follow.
- What is the purpose of the watch glass in such an experiment? (1 mark)
- To prevent loss of solid / spitting out of solid
- Why should the heating be continued until a constant mass is obtained? (1 mark)
- To ensure all watrer is removed
- Calculate the mass of the solution. (1 mark)
- 80.6 − 50.6 = 30g
- Calculate the mass of the residue. (1 mark)
- 62.6 − 50.6 = 12g
- Calculate the mass of the water. (1 mark)
30 − 12 = 18g OR 80.6 − 62.6 = 18g - Calculate the solubility of salt Q in g per 100g of water at 25°C. (2 marks)
Solubility = 12/18 × 100 = 66.67g/100g of water
- What is the purpose of the watch glass in such an experiment? (1 mark)
- Hard water has both advantages and disadvantages. Give one advantage and one disadvantage of using hard water. (2 marks)
- Advantage
- Provides calcium ions essential for strenghthening bones and teeth.
- Good for beer brewing
- Hard water does not dissolve in lead pipes.
- Disadvantage
- Wastes soap
- Stains white clothes
- Deposition of fur in kettles, pipes and boilers
- Advantage
- Using an equation, explain how addition of sodium carbonate is used to remove water hardness. (2 marks)
- When sodium carbonate is added, it precipitates out Ca2+ and Mg2+
Ca2+(aq) + CO32-(aq) → CaCO3(s)
OR
Mg2+(aq) + Co32-(aq) → MgCO3(s)
- When sodium carbonate is added, it precipitates out Ca2+ and Mg2+
- Use the data to answer the questions that follow.
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - Joint Pre-Mock Exams 2021/2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students