- Introduction
- X - ray Production
- Energy Changes in an X - Ray Tube
- Properties of X - Rays
- Hard X-rays
- Soft X-rays
- Dangers of X-rays and the Precautions
- Uses of X-rays

Introduction
- X-rays were discovered by a German scientist named Roentgen in 1985.
- They can pass through most substances including soft tissues of the body but not through bones and most metals.
- They were named X-rays meaning 'unknown rays'.

X-ray Production
- They are produced by modified discharge tubes called X-ray tubes. The cathode is in the form of a filament which emits electrons on heating.
- The anode is made of solid copper molybdenum and is called the target.
- A high potential difference between the anode and the cathode is maintained (10,000 v to 1,000,000 or more) by an external source.
- The filament is made up of tungsten and coiled to provide high resistance to the current.
- The electrons produced are changed into x-rays on hitting the anode and getting stopped. Only 0.2% of the energy is converted into x-rays.
- Cooling oil is led in and out of the hollow of the anode to maintain low temperature.
- The lead shield absorbs stray x-rays.
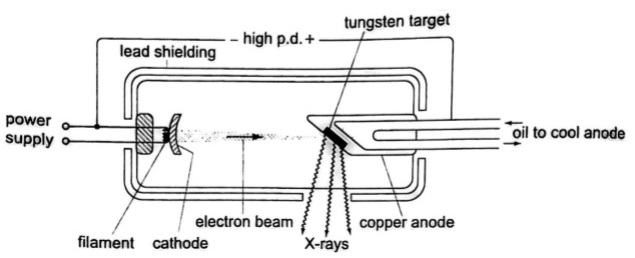

Energy Changes in an X-ray Tube
- When the cathode is heated electrons are emitted by thermionic emission.
- They acquire electrical energy which can be expressed as E = eV. Once in motion the electrical energy is converted to kinetic energy, that is eV = ½ mev2 .
- The energy of an electromagnetic wave can be calculated using the following equation
Energy = hf, where h - Planck’s f –frequency constant, of the wave . - The highest frequency of the X-rays released after an electron hits the target is when the greatest kinetic energy is lost, that is
hfmax = eV - Lower frequencies are released when the electrons make multiple collisions losing energy in stages, the minimum wavelength, λmin, of the emitted X-rays is given by;
(hc)/λmin = eV - These expressions can be used to calculate the energy, frequencies and wavelengths of X-rays.
Examples
- Determine the energy possessed by X-rays whose frequency is 4 × 1017 Hz.
Solution
E = hf
= 6.63 × 10-34 ×4 × 1017
= 2.652 × 10-16 J. - An X-ray tube operates at 60 kV and the current through it is 4.0 mA. Calculate the,
- Number of electrons striking the target per second.
- Speed of the electrons when they hit the target.
Solution
a) Current through the tube is given by I = ne , where n - number of electrons striking target per second and e- electronic charge (e = 1.6 × 10-19 coulombs)
So, n = 1/e = (4.0 × 10-3)/1.6 × 10-19 = 2.5 × 1016 electrons.
b) Kinetic energy = electrical energy
½ mev2 = eV, then v = 2.13 × 108 m/s
- An 18 kV accelerating voltage is applied across an X-ray tube. Calculate;
- The velocity of the fastest electron striking the target
- The minimum wavelength in the continuous spectrum of X-rays produced. (mass of electron - 9 × 10-31 kg, charge on an electron - 1.6 × 10-19 C, h - 6.6 × 10-34 J/s, c - 3 × 108 m/s)
Solution
a) V = 18 × 103 V
me = 9 × 10-31 kg
e = 1.6 × 10-19 C
h = 6.6 × 10-34 J/s
c = 3 × 108 m/s
½ mev2 = ev; therefore v = 8 × 107 m/s
b) (hc)/λmin = eV; λmin = hc/eV
λ min = (6.6 × 10-34 × 3 × 108 )/(1.6 × 10-19 × 18 × 103) = 6.9 × 10-11 m.

Properties of X-rays
- They travel in straight lines
- They undergo reflection and diffraction
- They are not affected by electric or magnetic fields since they are not charged particles.
- They ionize gases causing them to conduct electricity
- They affect photographic films
- They are highly penetrating, able to pass easily through thin sheets of paper, metal foils and body tissues
- They cause fluorescence in certain substances for example barium platinocynide.

Hard X-rays
- These are x-rays on the lower end of their range (10 - 11 – 10 - 8 m) and have more penetrating power than normal X - rays.
- They are capable of penetrating flesh but are absorbed by bones.

Soft X-rays
- They are on the upper end of the range and are less penetrative.
- They can only penetrate soft flesh and can be used toshow malignant growth in tissues.

Dangers of X-rays and the Precautions
- They can destroy or damage living cells when over exposed.
- Excessive exposure of living cells can lead to genetic mutation.
- As a precautionary measure X-ray tubes are shielded by lead shields.

Uses of X-rays
- Medicine – X-ray photos called radiographs are used as diagnostic tools for various diseases. They are also used to treat cancer in radiotherapy.
- Industry – they are used to photograph and reveal hidden flaws i.e. cracks in metal casting and welded joints.
- Science –since the spacing of atomic arrangement causes diffraction of x-rays then their structure can be studied through a process called X-ray crystallography.
- Security –used in military and airport installations to detect dangerous metallic objects i.e. guns, explosives, grenades etc.
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download X - RAYS - Form 4 Physics Notes.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

