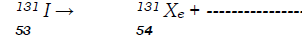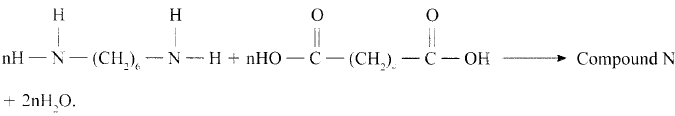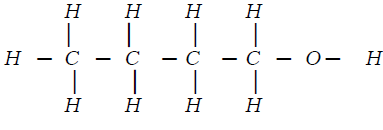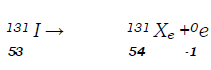- A student investigated the effect of an electric current by passing it through some substances. The student used inert electrodes, and connected a bulb to the circuit. The table below shows the substances used and their states.
Experiment Substance State 1 2 3 4 - In which experiment did the bulb not light? (1 mark)
- Explain your answer in (a) above. (2 marks)
- An alkanol has the following composition by mass: hydrogen 13.5%, oxygen 21.6% and carbon 64.9%
- Determine the empirical formula of the alcohol(C=12.0; H=1.0’ 0=16.0). (2mks)
- Given that empirical formula and the molecular formula of the alkanol are the same,draw the structure of the alkonol
- The figure below shows an energy cycle.
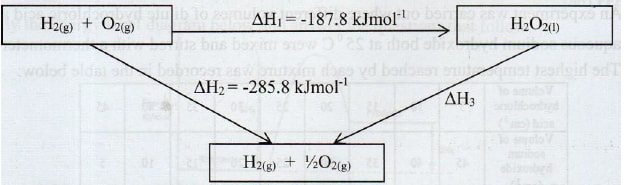
- Give the name of the enthalpy change ΔH1. (1 mark)
- Determine the value of ΔH3.(2 marks)
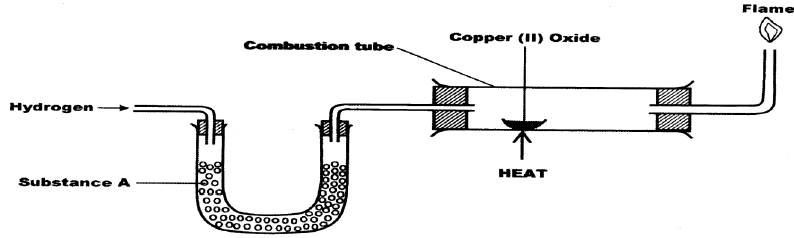
- The set up below was used to investigate the reaction between dry hydrogen gas and copper
- Name substance A.(1 mark)
- State the observation made in the combustion tube. (1 mark)
- Explain the observation stated in (b) above. (1 mark)
- Starting with sodium metal, describe how a sample of crystals of sodium hydrogen carbonate may be prepared. (3 marks)
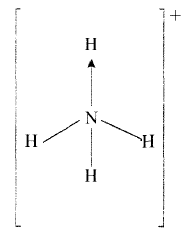
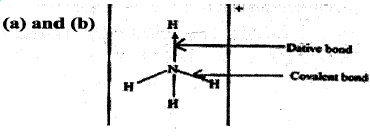
- Ammonium ion has the following structure Label on the structure:
- covalent bond;
- coordinate (dative) bond.
- When 8.53g of sodium nitrate were heated in an open test-tube, the mass of oxygen gas produced was 0.83 g . Given the equation of the reaction as 2NaNO3 (s) -> 2NaNO2 (s) + O2 (g)
Calculate the percentage of sodium nitrate that was converted to sodium nitrite. (Na = 23.0, N = 14.0, O = 16.0) (3 marks) - Aluminium is both malleable and ductile.
- What is meant by?
- Malleable: (1 mark).
- Ductile (1 mark)
- State One use of aluminium based on:
- malleability (½ marks)
- ductility (½ marks)
- What is meant by?
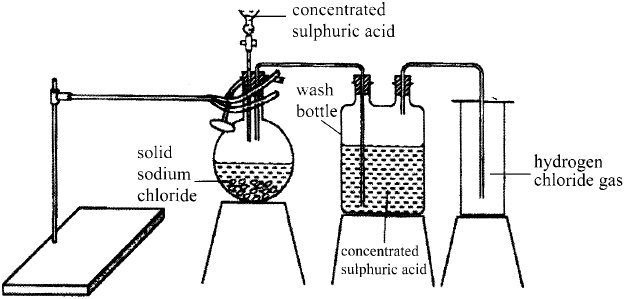
- The diagram below represents the set-up that was used to prepare and collect hydrogen chloride gas in the laboratory.
- State the purpose of concentrated sulphuric (VI) acid in the wash bottle. (1 mark)
- Write an equation for the reaction between dry hydrogen chloride gas and heated iron. (1 mark)
- Iron (III) oxide was found to be contaminated with copper (II)sulphate. Describe how a pure sample of iron (III) oxide can be obtained. (3 marks)
-
- Complete the nuclear equation below: (1 mark)
- The half-life of I is 8 days. Determine the mass of 131I remaining if 50 grammes decayed for 40 days (1 mark)
- Give one harmful effect of radioisotopes. (1 mark)
- Complete the nuclear equation below: (1 mark)
- During an experiment, chlorine gas was bubbled into a solution of potassium iodide.
- State the observations made. (1 mark)
- Using an ionic equation, explain why the reaction is redox. (2 marks)
-
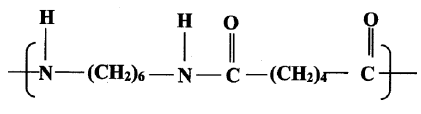
- Draw the structure of compound N formed in the following reaction.
- Give one use of compound N. (1mk)
- Draw the structure of compound N formed in the following reaction.
- When fuels burn in the internal combustion engine at high temperature, one of the products formed is nitrogen (II) oxide.
- Write the equation for the formation of nitrogen (II) oxide. (1 mark)
- Give a reason why nitrogen (II) oxide is not formed at room temperature. (1 mark)
- Describe how formation of nitrogen (II) oxide in the internal combustion engine leads to gaseous pollution. (2 mark)
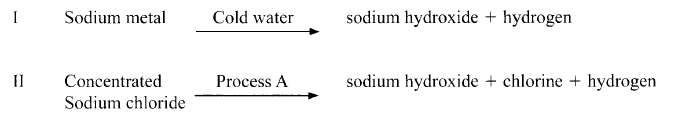
- Sodium hydroxide can be prepared by the following methods; I and II.
- Name one precaution that needs to be taken in method I. (1 mark)
- Give the name of process A. (1 mark)
- Give one use of sodium hydroxide. (1 mark)
- The atomic number of sulphur is 16. Write the electron arrangement of sulphur in the following: (2 marks)
- H2S
- S032-
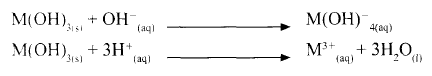
- A compound whose general formula is M(OH)3 reacts as shown by the equation below.
- What name is given to compounds which behave like M(OH) 3 in the two reactions. (1 mark)
- Name two elements whose hydroxides behave like that of M. (2 marks)
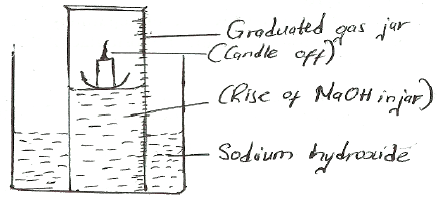
- A water trough, aqueous sodium hydroxide, burning candle, watch class and a graduated gas jar were used in an experimental set up to determine the percentage of active part of air. Draw a labeled diagram of the set up at the end of the experiment.
- In an experiment on rates of reaction, potassium carbonate was reacted with dilute sulphuric (VI) acid.
- What would be the effect of an increase in the concentration of the acid on the rate of the reaction? (1 mark
- Explain why the rate of reaction is found to increase with temperature. (2 mark
- 60 cm3 of oxygen gas diffused through a porous partition in 50 seconds. How long would it take 60cm3 of sulphur (IV) oxide gas to diffuse through the same partition under the same conditions? (S = 32.0, 0 = 16.0) (3 marks)
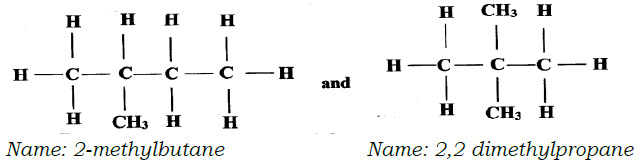
- Draw and name the isomers of pentane.(3 marks)
- The set-up below was used to collect a dry sample of a gas. Give two reasons why the set-up cannot be used to collect carbon (IV) oxide gas. (2 marks)
- Given the following substances: wood ash, lemon juice and sodium chloride.
- Name one commercial indicator that can be used to show whether wood ash, lemon juice and sodium chloride are acidic, basic or neutral. (1 mark)
- Classify the substances in 15(a) above as acids, bases or neutral. (2 marks
- Describe how a solid sample of potassium sulphate can be prepared starting with 200cm3of 2M potassium hydroxide. (3 marks)
- Charcoal is a fuel that is commonly used for cooking. When it burns it forms two oxides.
- Name the two oxides. (2 marks)
- State one use of the two oxides. (1 mark)
- Hydrogen sulphide is a highly toxic and flammable gas. It is normally prepared in a fume chamber.
- Name two reagents that can be used to prepare hydrogen sulphide in the laboratory.(1 mark)
- One of the uses of hydrogen sulphide is to produce sulphur as shown in the following equation: 2H2S(g) + S02 (g) -> 3S(s) + 2H20(l)
Identify the reducing agent in this reaction and give a reason for your answer. (1 mark) - Other than production of sulphuric (VI) acid, state one commercial use of sulphur.(1 mark)
- Describe an experimental procedure that can be used to extract oil from nut seeds. (2 marks)
- A mixture contains ammonium chloride, copper (II) oxide and sodium chloride. Describe how each of the substances can be obtained form the mixture. (3 marks)
- When a student was stung by a nettle plant, a teacher applied an aqueous solution of ammonia to the affected area of the skin and the student was relieved of pain. Explain. (2 marks)
MARKING SCHEME
- A student investigated the effect of an electric current by passing it through some substances. The student used inert electrodes, and connected a bulb to the circuit. The table below shows the substances used and their states.
Experiment Substance State 1 Potassium Carbonate Solid 2 Copper(II)sulphate Solution 3 Sugar Solution 4 Lead(II)iodide Molten - In which experiment did the bulb not light? (1 mark)
- 1 and 3
- Explain your answer in (a) above. (2 marks)
- In 1 ions K2CO3 are held rigidly within the crystal cannot move (no mobile ions) In 3 sugar exist as molecule hence no mobile ions
- In which experiment did the bulb not light? (1 mark)
- An alkanol has the following composition by mass: hydrogen 13.5%, oxygen 21.6% and carbon 64.9%
- Determine the empirical formula of the alcohol(C=12.0; H=1.0’ 0=16.0). (2mks)
- [E.F.= C4H9OH]
- Given that empirical formula and the molecular formula of the alkanol are the same,draw the structure of the alkonol
- Determine the empirical formula of the alcohol(C=12.0; H=1.0’ 0=16.0). (2mks)
- The figure below shows an energy cycle.

- Give the name of the enthalpy change ΔH1. (1 mark)
- Enthalpy of formation of hydrogen peroxide or enthalpy of formation
- Determine the value of ΔH3.(2 marks)
- ΔH1 + ΔH3 = ΔH2
ΔH3 = ΔH2 - ΔH1
= - 285.8 – (-187.8) = 187.8 – 285.8 = - 98 kJmol-1
- ΔH1 + ΔH3 = ΔH2
- Give the name of the enthalpy change ΔH1. (1 mark)
- The set up below was used to investigate the reaction between dry hydrogen gas and copper
- Name substance A.(1 mark)
- Fused anhydrous calcium chloride
- CaO: fused CaCl2
- State the observation made in the combustion tube. (1 mark)
- Black CuO changes to brown Cu metal
- Formation of colourless liquid on the cooler parts of the combustion tube.
- Explain the observation stated in (b) above. (1 mark)
- Copper (II) oxide is reduced by hydrogen to copper metal while hydrogen is oxidized to water/CuO reduced to Cu/H2 Oxidized to H2O
- Name substance A.(1 mark)
- Starting with sodium metal, describe how a sample of crystals of sodium hydrogen carbonate may be prepared. (3 marks)
- React sodium with water to get sodium hydroxide. Bubble into this solution excess carbon (IV) oxide to get sodium hydrogen carbonate
- Ammonium ion has the following structure Label on the structure:
- covalent bond;
- coordinate (dative) bond.
- When 8.53g of sodium nitrate were heated in an open test-tube, the mass of oxygen gas produced was 0.83 g . Given the equation of the reaction as 2NaNO3 (s) -> 2NaNO2 (s) + O2 (g)
Calculate the percentage of sodium nitrate that was converted to sodium nitrite. (Na = 23.0, N = 14.0, O = 16.0) (3 marks) - Aluminium is both malleable and ductile.
- What is meant by?
- Malleable: (1 mark)
- Can be hammered into sheets.
- Ductile (1 mark)
- Can be drawn into wires.
- Malleable: (1 mark)
- State One use of aluminium based on:
- malleability (½ marks)
- Making of sufurias/ motor vehicle parts/ aeroplane parts,window / door flames, cups, plates, packaging materials, pans, making sheets/ roof.
- ductility (½ marks)
- electricity cables/ wires.
- malleability (½ marks)
- What is meant by?
- The diagram below represents the set-up that was used to prepare and collect hydrogen chloride gas in the laboratory.
- State the purpose of concentrated sulphuric (VI) acid in the wash bottle. (1 mark)
- It is a drying agent.
- Write an equation for the reaction between dry hydrogen chloride gas and heated iron. (1 mark)
- Fe(s) + 2HCl(g) → FeCl2(s) +H2(g)
- State the purpose of concentrated sulphuric (VI) acid in the wash bottle. (1 mark)
- Iron (III) oxide was found to be contaminated with copper (II)sulphate. Describe how a pure sample of iron (III) oxide can be obtained. (3 marks)
- Add water to dissolve CUSO4, Fe2O3 doesn’t dissolve
- Filter out the undissolved Fe2O3
- Wash the residue with plenty of water ro remove traces of the filtrate.
- Dry the residue between the filter papers
-
- Complete the nuclear equation below: (1 mark)
- The half-life of I is 8 days. Determine the mass of 131I remaining if 50 grammes decayed for 40 days (1 mark)
- 50g 25g 12.5g 6.25g 3.125g 1.5625g
Or NW = 0.4(½ )n
NW = 50 x (½ )5 = 1.5625g
- 50g 25g 12.5g 6.25g 3.125g 1.5625g
- Give one harmful effect of radioisotopes. (1 mark)
- Instant / cause death
- Cause cancer
- Cause gene mutation
- Complete the nuclear equation below: (1 mark)
- During an experiment, chlorine gas was bubbled into a solution of potassium iodide.
- State the observations made. (1 mark)
- Solution turned from colourless to dark brown
- Greenish yellow / pale green colour of Cl2 disappears
- Brown solution / black solid is deposited
- Using an ionic equation, explain why the reaction is redox. (2 marks)
- Cl2 (aq) +2I– (aq) → I2 (aq)+2Cl- (aq)
- Explanation; Iodine oxidation state changes from -1 to 0 hence oxidation while Cl2 0.5 changes from 0 to -1 hence reduction / increase is ON and decrease is ON or movement of electrons Cl2 gains e’s where lose
- State the observations made. (1 mark)
-
- Draw the structure of compound N formed in the following reaction.
Answer - Give one use of compound N. (1mk)
- Making synthetic fibres such as for: - Ropes
- Blouses
- Stockings
- Undergarments Trouser
- Draw the structure of compound N formed in the following reaction.
- When fuels burn in the internal combustion engine at high temperature, one of the products formed is nitrogen (II) oxide.
- Write the equation for the formation of nitrogen (II) oxide. (1 mark)
- N2(g)+ 02(g)→ 2NO(g)
- Give a reason why nitrogen (II) oxide is not formed at room temperature. (1 mark)
- Nitrogen atoms in the molecule are joined by strong triple covalent bond that requires a lot of energy to break than provided at room temperature
- Describe how formation of nitrogen (II) oxide in the internal combustion engine leads to gaseous pollution. (2 mark)
- Nitrogen (II) oxide reacts with oxygen in air to form nitrogen (IV) oxide that dissolves in water vapour causing acid rain.
- Write the equation for the formation of nitrogen (II) oxide. (1 mark)
- Sodium hydroxide can be prepared by the following methods; I and II.
- Name one precaution that needs to be taken in method I. (1 mark)
- Small piece of sodium metal (pea size) with a lot of water
- Perform the experiment wearing goggles.
- Give the name of process A. (1 mark)
- Electrolysis
- Give one use of sodium hydroxide. (1 mark)
- Manufacture of paper (soften), soaps and detergents
- Fractional distillation of liquid air
- Extraction of aluminium metal
- Manufacture of bleaching agents eg NaOCl paper, textiles, oil refinery
- Making herbicides on weed killers
- Textile industry to soften
- Name one precaution that needs to be taken in method I. (1 mark)
- The atomic number of sulphur is 16. Write the electron arrangement of sulphur in the following: (2 marks)
- H2S
- 2.8.8
- S032-
- 2.8.2
- H2S
- A compound whose general formula is M(OH)3 reacts as shown by the equation below.
- What name is given to compounds which behave like M(OH) 3 in the two reactions. (1 mark)
- Amphoteric
- Name two elements whose hydroxides behave like that of M. (2 marks)
- Lead, Zinc and Aluminium
- What name is given to compounds which behave like M(OH) 3 in the two reactions. (1 mark)
- A water trough, aqueous sodium hydroxide, burning candle, watch class and a graduated gas jar were used in an experimental set up to determine the percentage of active part of air. Draw a labeled diagram of the set up at the end of the experiment.
- In an experiment on rates of reaction, potassium carbonate was reacted with dilute sulphuric (VI) acid.
- What would be the effect of an increase in the concentration of the acid on the rate of the reaction? (1 mark
- The rate of reaction increases. This is because when the concentration is high: the number of collisions between particles is also high hence reacts faster,
- Explain why the rate of reaction is found to increase with temperature. (2 mark
- Increase in temperature results in increase in the kinetic energy of the particles. This makes particles move faster and collide frequently leading to faster rate of reaction.
- What would be the effect of an increase in the concentration of the acid on the rate of the reaction? (1 mark
- 60 cm3 of oxygen gas diffused through a porous partition in 50 seconds. How long would it take 60cm3 of sulphur (IV) oxide gas to diffuse through the same partition under the same conditions? (S = 32.0, 0 = 16.0) (3 marks)
- Draw and name the isomers of pentane.(3 marks)
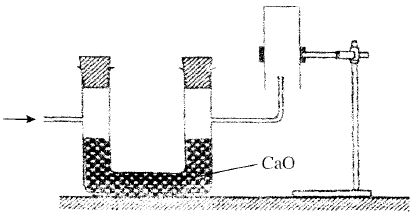
- The set-up below was used to collect a dry sample of a gas. Give two reasons why the set-up cannot be used to collect carbon (IV) oxide gas. (2 marks)
- It is denser than air
- It will react with calcium oxide since CO2 is acidic and CaO is basic
- Given the following substances: wood ash, lemon juice and sodium chloride.
- Name one commercial indicator that can be used to show whether wood ash, lemon juice and sodium chloride are acidic, basic or neutral. (1 mark)
- Classify the substances in 15(a) above as acids, bases or neutral. (2 marks
- Describe how a solid sample of potassium sulphate can be prepared starting with 200cm3of 2M potassium hydroxide. (3 marks)
- Vol of 2KOH = 100cm3 (or mols = 0.4/2 = 0.2
- Mix the KOH(aq) and H2SO4 acid
- Concentrate the mixture/ heat the mixture
- Crystalise the solution ( or heating the solution to dryness)
- Dry crystals
- Charcoal is a fuel that is commonly used for cooking. When it burns it forms two oxides.
- Name the two oxides. (2 marks)
- Carbon (IV) oxide /CO2/ carbon dioxide
- Carbon (II) oxide/ CO/ carbon monoxide
- State one use of the two oxides. (1 mark)
- Fire extinguisher/ photosynthesis
- Refrigeration
- Solvay process
- Fizzy drinks
- Food preservation
- Extraction of metals
- Manufacture of methanol
- Manufacture of fuel (water, gas)
- Name the two oxides. (2 marks)
- Hydrogen sulphide is a highly toxic and flammable gas. It is normally prepared in a fume chamber.
- Name two reagents that can be used to prepare hydrogen sulphide in the laboratory.(1 mark)
- Iron (II) sulphide or conc sulphide / copper sulphide (Accp formula: Fes/ HCl)
- Hydrochloric acid or lead (II) sulphide/ HNO3
- One of the uses of hydrogen sulphide is to produce sulphur as shown in the following equation: 2H2S(g) + S02 (g) -> 3S(s) + 2H20(l)
Identify the reducing agent in this reaction and give a reason for your answer. (1 mark)- Hydrogen sulphide
- The sulphur changes from -2 to zero/ (it reduces SO2 to S) i.e. +4 to 0 / sulphur lost e’s in the H2S to form sulphur
- Other than production of sulphuric (VI) acid, state one commercial use of sulphur.(1 mark)
- Vulcanization of rubber
- Manufacture of sulphur drugs
- Manufacture of gun powder/ match sticks / explosives/ fungicides
- Name two reagents that can be used to prepare hydrogen sulphide in the laboratory.(1 mark)
- Describe an experimental procedure that can be used to extract oil from nut seeds. (2 marks)
- Crush grind using a pestle and mortar, add suitable solvent of propanone ethanol alcohol and stir to dissolve oil. Filter the mixture to obtain a solution of the oil. Leave the solution in the sun for propanode to evaporate leaving the oil.
- A mixture contains ammonium chloride, copper (II) oxide and sodium chloride. Describe how each of the substances can be obtained form the mixture. (3 marks)
- Heat the mixture to sublime the NH4Cl. Add water to dissolve the NaCl. Copper (II) oxide does not dissolve. Filter and evaporate the filtrate to obtain sodium chloride.
- When a student was stung by a nettle plant, a teacher applied an aqueous solution of ammonia to the affected area of the skin and the student was relieved of pain. Explain. (2 marks)
- The product from nettle plant is acidic; aqueous ammonia solution being basic neutralize the acidic product
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2016 Chemistry Paper 1 Questions with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students