- The diagram below shows a set up for laboratory preparation of chlorine gas. Study it and answer the questions that follow.
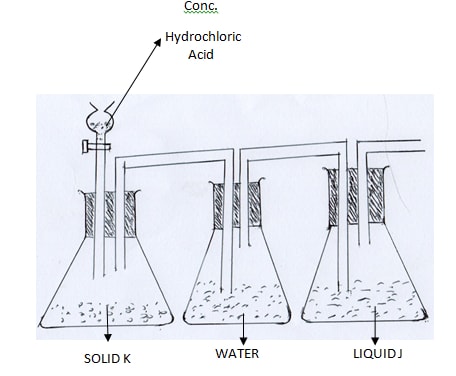
- Complete the set up to show how dry chlorine may be collected (1 mk)
- Name Soild K (2 mks)
Liquid J
State and explain the observation made when chlorine gas is bubbled through a solution of potassium iodide (2 mks) - Write an ionic equation for the reaction that occurs in iii) above (1 mk)
- Study the flow chart below and answer the questions that follow
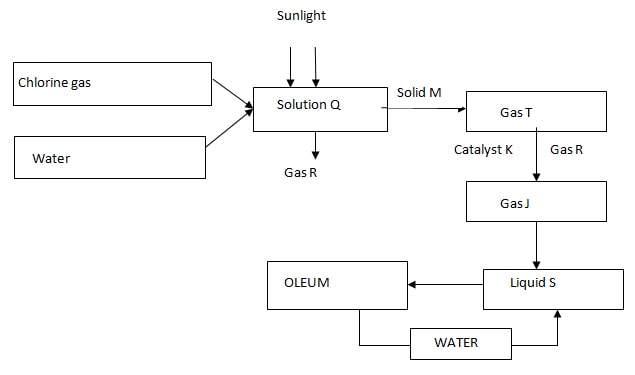
- Identify the following; (2 mks)
Gas R
Solid M
Gas J
Liquid S - Write a chemical equation for the formation of gas T (1 mk)
- Name catalyst K (1 mk)
- State two uses of liquid S (2mks)
- Identify the following; (2 mks)
- The diagram below shows a set up for laboratory preparation of chlorine gas. Study it and answer the questions that follow.
- The figure below represents a section of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of elements.
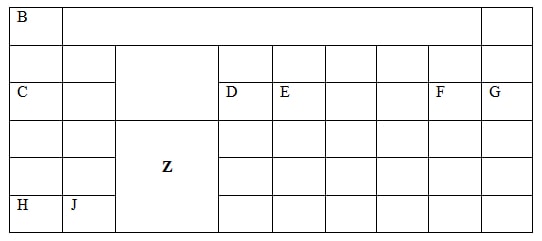
- What chemical family does element J belong to? (1 mk)
- Compare the reactivity of element C and H. Explain. (2 mks)
- Write the chemical formula of the chloride of element D (1 mk)
- Name the type of structure of the chloride in b(i) above. (1 mk)
- State and explain the difference in atomic radius and ionic radius of element F (2 mks)
- Using dots (.) and crosses (x) show how bonding occurs when element E and F react. (1mk)
- Study the diagram below and answer the questions that follow
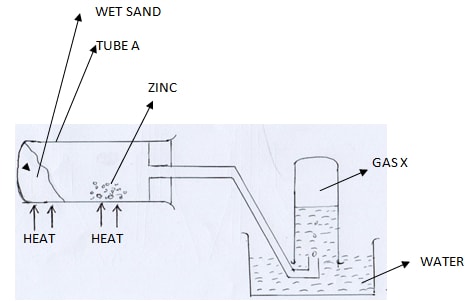
- State two observations made in the tube A (2 mks)
- Explain why the sand is heated first before heating the zinc metal (1 mk)
- Give the chemical test of gas X. (1 mk)
- Name the heat change represented by DH in the process represented below.
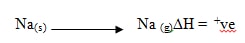
- The table below gives some bond energies of some bonds.
H 2(g) + Cl2(g) = 2 HCl(g) (2mks)Bond
Bond energy (kJ mol )
H – H
435
Cl – Cl
243
H – Cl
431
Calculate the enthalpy changes for the reaction. - Define enthalpy of formation of a substance. (1mk)
- Given that
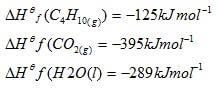
Calculate the molar heat of combustion of butane ( C4H10) (2mks)
- Study the following exothermic reactions involving enthalpy of neutralization
- NaOH(s) + HCl(aq) = NaCl(aq) + H2O(l)DH1
- NaOH(aq) + HCl(aq ) = NaCl(aq) + H2O(l)DH2
- Identify the reaction with a higher DH value. Giving a reason for your answer. (2mks)
- The atomic numbers of Li and K are 3 and 19 respectively.
Li+(g) + (aq) = Li+(aq)DH3 = -519
K+(g) + (aq) = K+(aq) DH4 = -322
Study the equations above and explain why DH3 is larger than DH4. (2mks)
- Name the heat change represented by DH in the process represented below.
- Use the standard electrode potentials given below to answer the questions that follow.
Ag+(aq) + e- = Ag(s) + 0.80v……….(i)
Cu2+(aq) + 2e- = Cu(s) + 0.34v ….…..(ii)
Pb2+(aq) + 2e = Pb(s) -0.13v…….…(iii)
Zn2+(aq) + 2e- = Zn(s) -0.76v………(iv)- Select two half - cells which when combined will give the lowest e.m.f (1mk)
- Can a solution of silver nitrate be stored in a container of Zinc? (2mks)
- An iron jug was electroplated using Chromium. The chromium electrode and iron jug were thoroughly cleaned and weighed before being dipped into the electrolyte. Why was cleaning necessary? (1mk)
- A current of 0.75 Amperes was passed through the solution for one hour and four minutes. The mass of chromium deposited on the jug was 0.52g
(1Faraday = 96500C Cr = 52)- Calculate the quantity of electricity passed. (2mks)
- How many moles in Chromium were deposited? (1mk)
- Calculate the quantity of electricity in coulombs required to deposit one mole of Chromium. (2mks)
- Deduce the charge of the Chromium ion. (2mks)
- An organic compound G whose empirical formula is HCO2 has molar mass of 90. It reacts with ethanoic in the presence of a few drops of concentrated Sulphuric (VI) acid forming another compound J with a pleasant smell.
- Determine the molecular formula of compound G (2marks)
- To which class of homologous series do compound G and J belong? (2marks)
- What is vulcanization of rubber? (1mk)
State two properties of vulcanized rubber. (2mks) - The scheme below shows various reactions and process for some organic compounds.
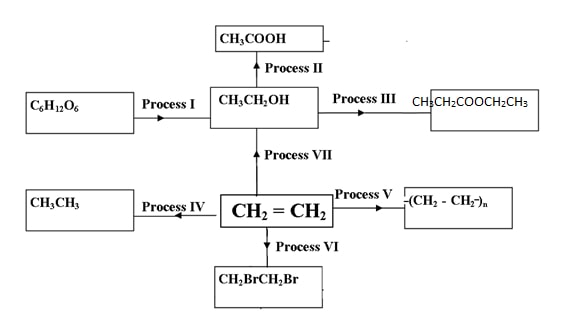
Study it and answer the questions that follow.- Name process I and name any other product formed in the process.
- Name the reagent and condition required in process II.
- Name the catalyst and reagent required in step IV.
- Give any one use of the product formed in process (V) (1mk)
- Name process (VI) (1mk)
- An organic compound G whose empirical formula is HCO2 has molar mass of 90. It reacts with ethanoic in the presence of a few drops of concentrated Sulphuric (VI) acid forming another compound J with a pleasant smell.
- The flow chart below outlines some of the processes involved in extraction of copper from copper pyrites. Study it and answer the questions that follow.
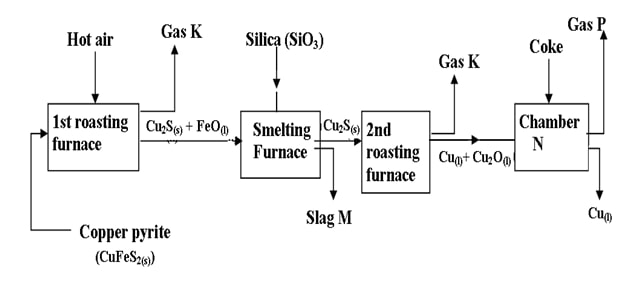
- Name gas K (1mk)
- Write an equation for the reaction that takes place in the first roasting furnace.(1mk)
- Write the formula for the cation present the slag M. (1mk
- Identify gas P. (1mk)
- What name is given to the reaction that takes place in chamber N?
Give a reason for your answer. (2mks) - State two uses of copper metal (2mks)
- Below is a simplified diagram of the Downs cell in which sodium metal is manufactured.
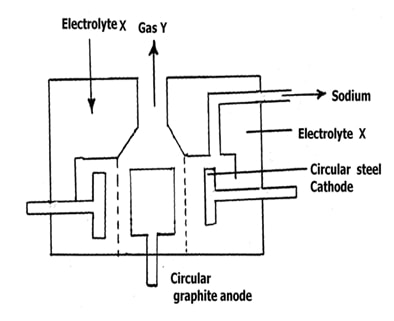
- Identify electrolyte X and gas Y. (2mks)
Electrolyte X ………………………………….…Gas Y……………………………… - Give two properties of sodium that make it possible to collect as shown above. (2mks)
- Why is graphite preferred to steel as the anode? (1mk)
- Explain how sodium hydroxide pellets would be obtained from sodium amalgam? (1mk)
- Identify electrolyte X and gas Y. (2mks)
- Study the reaction scheme below and answer the questions that follow.
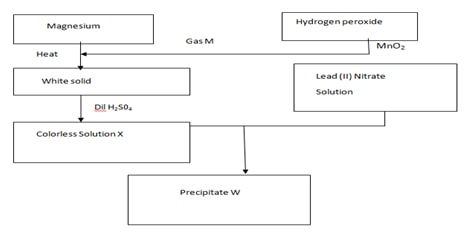
- Name; Gas M…………………………………………………………….. (1 mk)
Precipitate W……………………………………………………... (1 mk) - Write an equation for the formation of ;
The white solid (1 mk)
Colorless solution X (1 mk) - What name is given to the process that produce precipitate W (1 mk)
- State two commercial uses of gas M (2 mks)
- If 100cm3 of 0.05M hydrogen peroxide were used to produce gas M. calculate the volume of gas M produced if the hydrogen peroxide decomposed completely.
(MGV= 22400cm3) ( 2mks)
- Name; Gas M…………………………………………………………….. (1 mk)

MARKING SCHEME
- Downward delivery
- K; potassium manganate vii
J;Concentrated sulphuricvi acid - Brown solution; chlorine oxidises iodide ions to iodine.
- Cl2 (g) + 2I- (aq) → 2Cl-(aq) + I2 (aq)
- R; Oxygen
M; Sodium sulphite * Accept correct formulas
J;Sulphur iv oxide
S; Concsulphuricvi acid - Na2SO3 (s) + 2HCl (aq) → 2NaCl (aq) + SO2 (g)+ H2O (l)
- Vanadium v oxide/ platinum
- - manufacture of fertilizers
-manufacture of detergents
- Manufacture of plastics * any two
-in lead acid accumulators
- In making dyes and paints
- processing metal ores
- R; Oxygen
- Alkaline earth metals
- H is more reactive than C; H has lower ionization energy/ H has more ease of losing an electron
- DCl3/ D2Cl6 // AlCl3/ Al2Cl6
- Molecular structure
- The atomic radius is lower than the ionic radius; This is due to repulsion between existing electrons and the gained electrons;
- ( correct electron distribution)
- – zinc glows
- Yellow solid when hot (and white when cool) - To drive out air initially present in the tube
- Extinguishes/ puts off a burning splint with a pop sound
- – zinc glows
- Enthalpy of atomization of Na(s)ü1\
- DH reaction = ( H – H + Cl – Cl) – ( 2 x H –Cl)
= ( 435 + 243) – ( 2 x 431)ü
= - 184 kJ / Molü - Is the heat change which occur when a compound is formed from its constituent elements in their standard stateü.
- 4C(s) + 5H2(g)Hf(C4H10 = C4H10
5/2O2
4O2 DHf(H2O) 13/2O2
DHf(CO2) DHc(C4H10)
4CO2(g) + 5H2O(l)
DHf(C4H10) + DHc(C4H10) = 4 x Hf ( CO2) + 5 x DHf(H2O)
-125 + DHc (C4H10) = (4 x -395) + ( 5 x – 289)ü
DHc(C4H10) = -3025 + 125 ü ½
= + 2900kJ / molü ½
- DH1 ü has higher value since it involves heat of solution of NaoH(s) in addition to heat of neutralization of NaOH(aq) but DH2 does not involve heat of solution since NaOH (aq) is already in solution ü 1.
- Li+ smaller in size ü hence has a stronger effective nuclear charge ü attraction which leads to formation of stronger bonds with water releasing more heat.
- ii and iii ü
- EMF = 0.80 – -76 = ±1.76 = +1.56vüvü
- The reaction can occur hence cannot be stored ü - To remove any oxide layer. ü
- Q = 0.75 x 64 x 60 ü
= 2880Cü - Moles of Chromium deposited = 52/52ü 1/2
= 0.01 moles ü ½ - 0.01 moles 2880 C
\1 mole 2880/0.01x 1ü
= 288000cü - No of faradays = 288000/96500ü
= 2.984ü ½
Charge = +3 ü ½
- Q = 0.75 x 64 x 60 ü
- (HCO2)n = 90
(1+12+32)n=90
45n=90
n=2
(HCO2)2 = H2C204 - G- Alkanoic acids
J-Esters
- (HCO2)n = 90
- Process of adding sulphur to rubber then heating
- - It’s tougher
- Its less flexible
- It’s harder
- process I –Fermentation
Product- carbon IV oxide - Reagent- Acidified potassium manganate vii/ acidified potassium dichromate VI
Condition- Warming - Catalyst- Nickel
Reagent- Hydrogen - Used in making packaging materials ( carrier bags,bottles,jerry cans,crates etc.)
- Addition halogenation/ Addition Bromination
- process I –Fermentation
- Suphuric (IV) Oxide ü ½
- 2CuFeS2(s) + 4O2(g) = 3SO2(g) + 2FeO(s) + Cu2S(s)ü 1
- Fe2+ü ½
- Carbon (II) Oxide / CO(g)ü ½
- Reduction ü
Copper (I) Oxide is reduced by coke to copper metal ü - - To make electrical appliances
To make coins
In roofing and making gutters
To make alloy such as brass and bronze
- X – Molten sodium chloride ü ½
gas Y - Chlorine ü ½ - Less dense than molten sodium Chloride ü ½
Has low melting point ü ½
Crystallises faster than calcium - At high temperature, steel would react with chlorine formed, but graphite is inert even at high temperatureü
- Add water then evaporate to form sodium hydroxide (NaOH) ü
2NaHg(l) + 2H2O(l) // 2NaOH(ag) + 2Hg(s) + H2(g)
- X – Molten sodium chloride ü ½
- Gas M- Oxygen
W- Lead ii sulphate - 2Mg (s) + O2 (g) → 2MgO (s)
MgO (s) + H2SO4 (aq) → MgSO4 (aq) + H2O (l) - Precipitation/double decomposition/ ion exchange
– Used by mountain climbers and deep sea divers
- To burn rocket fuel
- In hospitals by patients with breathing difficulties/ In incubators
- Used in welding to cut metals
- To remove impurities during steel making
- As a reactant in fuel cells - 2H2O2 → 2H2O + O2
Moles of H2O2= 0.05X 100/1000
=0.005
Mols of O2 = 0.005/2 = 0.0025
Volume of O2 = 0.0025X 22400
=56cm3
- Gas M- Oxygen
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download CHEMISTRY PAPER 2 - 2019 LAINAKU JOINT MOCK EVALUATION EXAMINATION.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

