Instructions to candidates
- This paper consists of two sections A and B.
- Answer all the questions in the two sections in the spaces provided after each question
- All working must be clearly shown.
- Electronic calculators and Mathematical tables may be used.
- All numerical answers should be expressed in the decimal notations.
SECTION A (25 MARKS)
Answer all the questions in this section in the spaces provided.
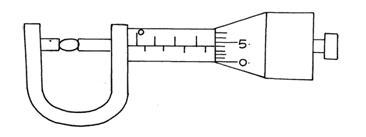
- Figure 1. shows a micrometer screw gauge being used to measure the diameter of a ball bearing.
If the instrument has a negative zero error of 0.01mm, record the actual diameter of the ball bearing. (1mark)
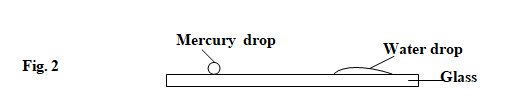
- Figure 2. shows drops of mercury and water on a glass surface,
Explain the difference in the shapes of the drops. (2 marks)
- State why diffusion is faster in gases than in liquids. (1mark)
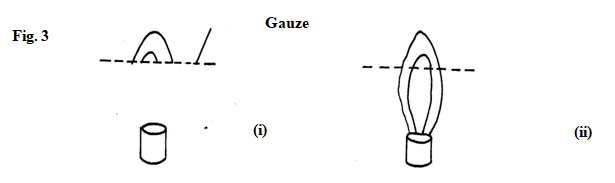
- When a Bunsen burner is lit above a wire gauze, it is observed that the flame initially burns above the gauze shown in figure 3 (i). After sometime, the flame burns below as well as above the gauze as shown in figure 3(ii).
Explain the observation. (2marks)
- In an experiment to demonstrate Brownian motion, smoke was placed in a smoke cell and observed using a microscope. The smoke particles were seen moving randomly in the cell. Explain the observation. (1mark)
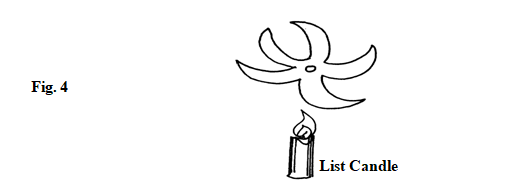
- A paper vane in a horizontal axis was placed above a Bunsen burner as shown in figure 4. When the burner was lit, the paper vane begun to rotate. Explain the observation. (2marks)
- An electric kettle with shiny outer surface is more efficient than one with a dull outer surface, give a reason for this. (1mark)
- What is the reason why trailers carrying heavy loads have many wheels. (1mark)
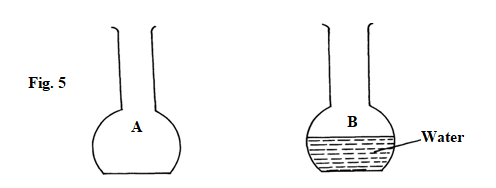
- Two flasks A and B were placed on a horizontal surface as shown in figure 5.
State and explain which flask is more stable. (2marks)
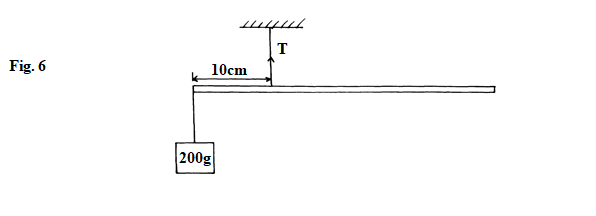
- Figure. 6 below shows a metre rule balancing when a mass of 200g is hung at one end. Determine the tension, T in the string. (3marks)
- State Newton’s second law of motion. (1mark)
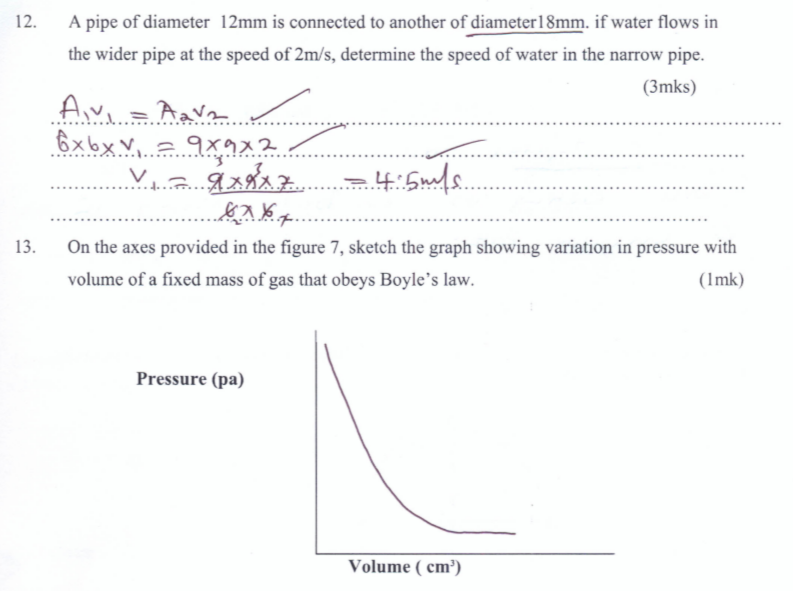
- A pipe of diameter 12mm is connected to another of diameter18mm. if water flows in the wider pipe at the speed of 2m/s, determine the speed of water in the narrow pipe. (3marks)
- On the axes provided in the figure 7, sketch the graph showing variation in pressure with volume of a fixed mass of gas that obeys Boyle’s law. (1mark)
- An oil drop has a volume of 0.01mm3. When it is placed on the surface of water, it spreads out to form a circular patch of area 500cm2.
- Calculate the size of the molecule of the oil. (3marks)
- State one assumption made in (i) above. (1mark)
SECTION B (55 marks)
-
- Define angular velocity (1 mark)
- The figure below shows an object of mass 0.2kg whirled in a vertical circle of radius 0.3m at uniform speed of 4m/s
- Determine the tension of the string at:
- Position A (2 marks)
- Position B (2 marks)
- Suggest the point where the string is likely to snap. (1 mark)
- The figure below shows the motion of a trolley on a ticker tape timer whose frequency is 100HZ
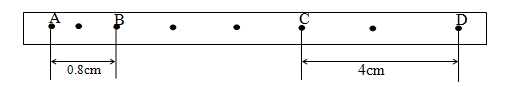
Determine:- Initial velocity at points AB. (2 marks)
- Velocity at points CD (2 marks)
- Acceleration of the trolley during the motion. (2 marks)
- Define angular velocity (1 mark)
-
- Give the reason why a density bottle is more accurate than a measuring cylinder when used to measure volume of liquids. (1 mark)
- State two precautions taken when using a density bottle. (2 marks)
- A form one student wanted to determine the density of copper. She wrote the following procedure: Study it and answer the questions that follow.
- Measure the mass x1 (g) of a clean dry empty density bottle
- Fill the bottle partly with copper turnings and measure the mass x2(g)
- Fill the bottle with water up to the neck and measure its mass x3(g)
- Empty the bottle and rinse it.
- Fill it with water and replace the stopper. Measure the mass x4(g) of the bottle filled with water.
Write an expression for:- Volume of the bottle (2 marks)
- Mass of copper turnings (1 mark)
- Volume of copper turnings. (2 marks)
- Density of copper. (2 marks)
-
- State the law of floatation (1 mark)
- A metal block weighs 0.8N is suspended by a string in water. If the block is completely immersed in water the tension in the string is 0.5N. Find
- The upthrust on the block (1 mark)
- The density of the block. (3 marks)
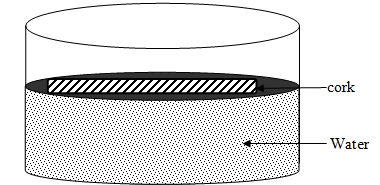
- The figure below shows a cork of mass 25g floating in water.
Determine the minimum volume of copper that must be attached so that the two will just submerge.( Relative density of copper = 9.0, Relative density of cork= 0.25)(3marks)
- Explain how a submarine can be made to.
- Float on water (1 mark)
-
- You are provided with the following apparatus: A filter funnel, a thermometer, a stop watch, ice at 0°C, an immersion heater rated P watts, a beaker, a stand, boss and clamp and a weighing machine. Describe an experiment to determine the specific latent heat of fusion of ice. Clearly state the measurements to be made. (3marks)
- 200 g of ice at 0°C is added to 400g water in a well lagged calorimeter of mass 40g. The initial temperature of the water was 40°C. If the final temperature of the mixture is X°C,(Specific latent of fusion of ice L = 3.36 x 105 Jkg-1, specific heat capacity of water, c = 4200Jkg-1K-1, specific heat capacity of copper = 400 Jkg-1K-1.)
- Derive an expression for the amount of heat gained by ice to melt it and raise its temperature to X°C (2marks)
- Derive an expression for the amount of heat lost by the calorimeter and its content when their temperature falls to X°C. (2marks)
- Determine the value of X. (3marks)
- A hydrogen balloon of volume 1.2 m3 is released at the ground level where the pressure is680 mmHg and a temperature of 20 °C. Determine the volume of the balloon at a height of2500m above the ground where the pressure drops to 500 mmHg and the temperature is 4°C. (4marks)
- A balloon seller has a cylinder of helium gas which he uses to blow up his balloons. The volume of the cylinder is 0.10m3. It contains helium gas at a pressure of 1.0 x 107Nm-2. The balloon seller fills each balloon to a volume of 1.0 x 10-2m3 and a pressure of 2.0 x 105N/m2
- Explain in terms of particles how the helium in the cylinder produces a pressure (1mark)
- Calculate the total volume that the helium gases occupy at a pressure of 1.2 x 105 N/m2. Assume the temperature of the helium does not change (3marks)
- Calculate the number of balloons of volume 1.0 x 10-2m3 that the balloon seller can fill using the gas (2marks)
- The graph below shows how the pressure of a gas trapped inside a sealed container changes with temperature. The pressure is caused by the gas particles continually hitting the sides of the container.
- Write down the name of the temperature at which the gas particles stop hitting the sides of the container (1mark)
- What is the momentum of the gas particles at this temperature? Give reason for the Answer (2marks)
- Give the value of the temperature in Kelvin (1 mark)

MARKING SCHEME
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download PHYSICS PAPER 1 - 2019 MOKASA II MOCK EXAMINATION.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students