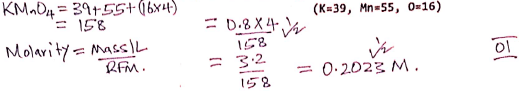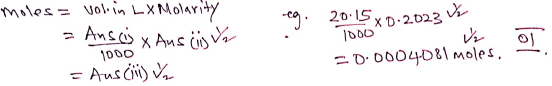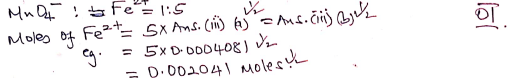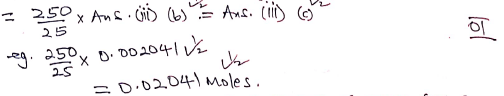CHEMISTRY PAPER 3 CONFIDENTIAL
In addition to the common laboratory apparatus and fittings, each candidate shall be supplied with the following
- 60 cm3of 0.5M copper (II) sulphate labeled solution K
- 5g of Solid L
- 90cm3 of acidified potassium manganate(VII), labelled as Solution M
- 60 cm3of 2M sulphuric (VI) acid, H2SO4
- 2ml of solution X
- 2ml of solution Y
- 2ml of solution Z
- About 8 cm3of liquid E in a stoppered test tube
- About 2g of solid Q in a stoppered container
- Burette
- Pipette and pipette filler
- Three conical flasks
- 10ml measuring cylinder.
- 250 ml plastic beaker
- 250 ml volumetric flask with a stopper
- 1 label
- Stop watch
- Clean glass rod/looped nichrome wire
- Clean and dry Metallic spatula
- Thermometer (-10-1100c)
- four test tubes
- test tube holder
- Filter paper
- Filter funnel
- Retort stand
- white tile
- 10ml measuring cylinder.
- 50ml measuring cylinder.
Access to the following:
- Distilled water
- Bunsen burner
- Acidified potassium dichromate (VI) supplied with a dropper.
- 2M Barium nitrate solution supplied with a dropper.
- 2M lead (II) nitrate supplied with a dropper
- 2M Nitric (V) acid supplied with a dropper
- Sodium hydrogen carbonate solid supplied with a spatula
Note
- Solid Q is Na2SO3
- Liquid E is ethanol
- Solution K is prepared by accurately weighing 125g of hydrated copper (II) sulphate and making up to 1000 cm3 of solution
- Solution M is prepared by accurately weighing 3.2g of KMnO4.then dissolving in 100ml of 2M sulphuric acid and topping up to 1000ml of the solution
- Solution X is 2M potassium nitrate
- Solution Y is made by dissolving One Spatulaful of blue Toss detergent in 100ml of distilled water (do not use tap water)then filter
- Solution Z is 2M copper (II) sulphate solution
CHEMISTRY PAPER 3 CHEMISTRY PRACTICAL QUESTIONS
Question 1
You are provided with the following reagents:
- Solution K- Copper (II) sulphate solution
- Solid L- Iron powder
- Solution M- Acidified Potassium Manganate (VII) solution, containing 0.8g of Potassium Manganate (VII) in 250cm3 of the solution.
You are required to determine the molar heat of displacement of copper in a solution of its ions by iron metal.
Procedure I
- Place 50cm3 of Solution K in a 100cm3 plastic beaker using a burette.
- Measure the constant temperature of the solution and record it in the Table 1 below.
- Add all of the Solid L provided at once and start a stop watch immediately.
- Using a thermometer, Stir the mixture thoroughly and continuously and record the temperature of the mixture after every one minute in the table 1.
- Retain the resultant mixture for use in the next Procedure II.
Table 1
| Time (Min) | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| Temperature(0C) |
(3 marks)
- Plot a graph of temperature (vertical axis) against time on the grid provided below. (3 marks)
- From the graph you have drawn, determine the;
- highest change in temperature, ∆T (1 mark)
- time taken for the reaction to completely occur (1 mark)
- Calculate the heat change for the reaction. (Take density of the solution to be 1g/cm3 and specific heat capacity of the solution to be 4200kJ/Kg/K) (2 marks)
Procedure II
- Swirl the mixture obtained in procedure I above and filter into a 250mL volumetric flask.
- Thoroughly rinse the beaker with 20cm3 of distilled water and ensure all the mixture has been transferred onto the filter paper.
- Add 50cm3 of 2M Sulphuric (VI) acid to the filtrate mixture in the volumetric flask.
- Add more distilled water to the solution in the volumetric flask to the mark. Mix the contents thoroughly and label this solution as Solution N.
- Fill the burette with Solution M.
- Place 25 cm3 of Solution N into a 250 cm3 conical flask using a pipette and a pipette filler.
- Titrate Solution N against Solution M until the first permanent pink colour is seen.
- Record your results in Table 2
- Repeat the titration twice and complete Table 2.
Table 2
| Titre | I | II | III |
| Final burette reading(cm3) | |||
| Initial burette reading (cm3) | |||
| Volume of solution M used (cm3) |
(3 marks)
- What is the average volume of Solution M used? (1 mark)
- Calculate the molarity of Solution M, KMnO4 (1 mark) (K=39, Mn=55, O=16)
- Calculate the number of moles of:
- Potassium manganate (VII) used, solution M (1 mark)
- Iron (II) ions in 25cm3 of solution N (1 mark)
The equation for the reaction is:
MnO4-(aq) + 8H+ (aq) + 5Fe2+ (aq) -> Mn2+ (aq) + 4H2O (l) + 5Fe3+ (aq) - Iron (II) ions in the 250cm3 of solution N (1 mark)
- Determine the molar heat of displacement of copper from a solution of its ions by iron metal (2 marks)
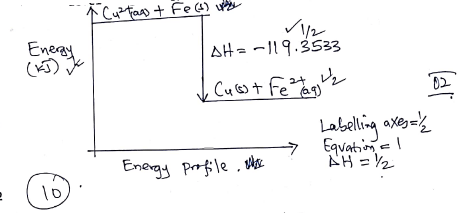
- Draw an energy level diagram for the reaction (2 marks)
Question 2
- You have been provided with solutions X, Y and Z. Carry out the flame tests for each and indicate the colour of the flames and inferences below.(3 marks)
Ions Flame Colour Inference X Y Z - You are provided with Solid Q. Carry out the tests below and Write your observations and inferences in the spaces provided.
- Place all Solid Q in a clean test tube. Add about 8cm3 of distilled water and shake. Divide the solution into 3 portions
Observation Inference (1/2 mark) (1/2 mark) - To the first portion add a few drops of Lead (II) nitrate solution and warm
Observation Inference (1 mark) (1 mark) - To the first portion add a few drops of Barium nitrate solution followed by few drops of dilute hydrochloric acid.
Observation Inference (1 mark) (1 mark) - To the third portion add a few drops of acidified potassium dichromate (VI) then warm gently.
Observation Inference (1 mark) (1 mark)
- Place all Solid Q in a clean test tube. Add about 8cm3 of distilled water and shake. Divide the solution into 3 portions
- You have been provided with Liquid E.
- Place about 2cm3 of the Liquid E in a clean test tube. Add an equal amount of distilled water and shake the mixture. Allow to settle.
Observation Inference (1 mark) (1 mark) - Place about 2cm3 of the Liquid E in a clean test tube. Add a half spatulaful of sodium hydrogen carbonate.
Observation Inference (1 mark) (1 mark) - To about 2cm3 the Liquid E add 3 drops of acidified potassium dichromate (VI) solution and warm gently.
Observation Inference (1 mark) (1 mark) - Take a few drops of Liquid E on a clean and dry metallic spatula and ignite over a non-luminous Bunsen flame.
Observation Inference (1 mark) (1 mark)
- Place about 2cm3 of the Liquid E in a clean test tube. Add an equal amount of distilled water and shake the mixture. Allow to settle.

Marking Scheme
Question 1
You are provided with the following reagents:
- Solution K- Copper (II) sulphate solution
- Solid L- Iron powder
- Solution M- Acidified Potassium Manganate (VII) solution, containing 0.8g of Potassium Manganate (VII) in 250cm3 of the solution.
You are required to determine the molar heat of displacement of copper in a solution of its ions by iron metal.
Procedure I
- Place 50cm3 of Solution K in a 100cm3 plastic beaker using a burette.
- Measure the constant temperature of the solution and record it in the Table 1 below.
- Add all of the Solid L provided at once and start a stop watch immediately.
- Using a thermometer, Stir the mixture thoroughly and continuously and record the temperature of the mixture after every one minute in the table 1.
- Retain the resultant mixture for use in the next Procedure II.
Table 1
| Time (Min) | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| Temperature(0C) | 21.0 | 22.5 | 24.0 | 25.0 | 27.0 | 28.0 | 29.5 | 31.0 | 31.0 | 31.0 | 29.5 |
ct - 1, D - 1/2, AC - 1, Trend - 1/2
(3 marks)
- Plot a graph of temperature (vertical axis) against time on the grid provided below. (3 marks)
- From the graph you have drawn, determine the;
- highest change in temperature, ∆T (1 mark)
32.6-21.0 = 11.6oC or 11.6K
(Must be shown on graph) - time taken for the reaction to completely occur (1 mark)
8 minutes
(must be shown on graph)
- highest change in temperature, ∆T (1 mark)
- Calculate the heat change for the reaction. (Take density of the solution to be 1g/cm3 and specific heat capacity of the solution to be 4200kJ/Kg/K) (2 marks)
Procedure II
- Swirl the mixture obtained in procedure I above and filter into a 250mL volumetric flask.
- Thoroughly rinse the beaker with 20cm3 of distilled water and ensure all the mixture has been transferred onto the filter paper.
- Add 50cm3 of 2M Sulphuric (VI) acid to the filtrate mixture in the volumetric flask.
- Add more distilled water to the solution in the volumetric flask to the mark. Mix the contents thoroughly and label this solution as Solution N.
- Fill the burette with Solution M.
- Place 25 cm3 of Solution N into a 250 cm3 conical flask using a pipette and a pipette filler.
- Titrate Solution N against Solution M until the first permanent pink colour is seen.
- Record your results in Table 2
- Repeat the titration twice and complete Table 2.
Table 2
| Titre | I | II | III |
| Final burette reading(cm3) | 20.5 | 20.2 | 40.3 |
| Initial burette reading (cm3) | 0.0 | 0.0 | 20.2 |
| Volume of solution M used (cm3) | 20.5 | 20.2 | 20.1 |
(3 marks)
CT = 1, AC = 1, D = 1/2, PA = 1/2, FA = 1
- What is the average volume of Solution M used? (1 mark)
(20.2 + 20.1)/2 = 20.15 - Calculate the molarity of Solution M, KMnO4 (1 mark) (K=39, Mn=55, O=16)
- Calculate the number of moles of:
- Potassium manganate (VII) used, solution M (1 mark)
- Iron (II) ions in 25cm3 of solution N (1 mark)
The equation for the reaction is:
MnO4-(aq) + 8H+ (aq) + 5Fe2+ (aq) -> Mn2+ (aq) + 4H2O (l) + 5Fe3+ (aq)
- Iron (II) ions in the 250cm3 of solution N (1 mark)
- Potassium manganate (VII) used, solution M (1 mark)
- Determine the molar heat of displacement of copper from a solution of its ions by iron metal (2 marks)
- Draw an energy level diagram for the reaction (2 marks)
Question 2
- You have been provided with solutions X, Y and Z. Carry out the flame tests for each and indicate the colour of the flames and inferences below.(3 marks)
Ions Flame Colour Inference X Purple flame K+ present Y Yellow flame Na+ present Z Green flame Cu2+ present - You are provided with Solid Q. Carry out the tests below and Write your observations and inferences in the spaces provided.
- Place all Solid Q in a clean test tube. Add about 8cm3 of distilled water and shake. Divide the solution into 3 portions
Observation Inference Dissolves to form a colourless solution(1/2 mark) -Polar substance/soluble substance
-Absence of Cu2+, Fe2+, Fe3+(1/2 mark) - To the first portion add a few drops of Lead (II) nitrate solution and warm
Observation Inference White precipitate, insoluble on warming(1 mark) CO32-, SO42-, SO32- present(1 mark) - To the first portion add a few drops of Barium nitrate solution followed by few drops of dilute hydrochloric acid.
Observation Inference -White precipitate soluble in the acid to form a colourless solution.
-Effervescence of a colourless gas (1 mark)CO32-,SO32-(1 mark) - To the third portion add a few drops of acidified potassium dichromate (VI) then warm gently.
Observation Inference Orange H+/K2Cr2O7 turns green(1 mark) SO32- present(1 mark)
- Place all Solid Q in a clean test tube. Add about 8cm3 of distilled water and shake. Divide the solution into 3 portions
- You have been provided with Liquid E.
- Place about 2cm3 of the Liquid E in a clean test tube. Add an equal amount of distilled water and shake the mixture. Allow to settle.
Observation Inference Miscible with water/ forms uniform mixture(1 mark) Liquid E is polar(1 mark) - Place about 2cm3 of the Liquid E in a clean test tube. Add a half spatulaful of sodium hydrogen carbonate.
Observation Inference No fizzling/no bubbles or no effervescence or a colourless gas(1 mark) RCOOH, H+, H3O+,COOH absent(1 mark) - To about 2cm3 the Liquid E add 3 drops of acidified potassium dichromate (VI) solution and warm gently.
Observation Inference Colour of H+/K2Cr2O7 changes from orange to green. (1 mark) ROH present(1 mark) - Take a few drops of Liquid E on a clean and dry metallic spatula and ignite over a non-luminous Bunsen flame.
Observation Inference Burns with a blue non-sooty/non-smoky flame(1 mark) ROH confirmed(1 mark)
- Place about 2cm3 of the Liquid E in a clean test tube. Add an equal amount of distilled water and shake the mixture. Allow to settle.
Download Chemistry Paper 3, with Confidential - 2020 MOKASA JOINT MOCKS EXAMINATION (QUESTIONS AND ANSWERS).
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students