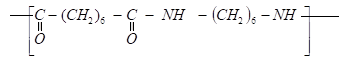Instructions to Candidates
- Answer ALL the questions in the spaces provided in the question paper
- KNEC Mathematical tables and electronic calculators may be used for calculations
- All working MUST be clearly shown where necessary
- Candidates should answer the questions in English

Questions
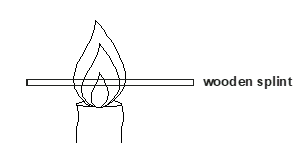
- Study the diagram below then use it to answer the questions that follow.
- Draw the wooden splint at the end of the experiment. If it was slipped then removed. (1 mark)
- Explain the appearance of the wooden splint in (a) above. (2 marks)
-
- The half-life of 210 83M is 7 days. Determine the mass of remaining if 100g decayed in 35 days. (1 mark)
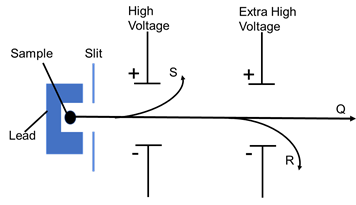
- The diagram below shows the radiations emitted by a radioactive sample.
- Identify radiation particles S and R. (1 mark)
S……………………………………………………
R…………………………………………………… - Which emission causes most harm to human cells. Give a reason. (1mark)
- Identify radiation particles S and R. (1 mark)
- Starting with copper metal, describe how a solid sample of copper (II) carbonate can be prepared. (3 marks)
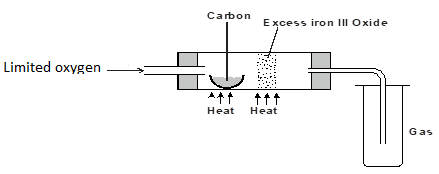
- The set-up below was used to obtain a sample of iron.
Write two equations for the reactions which occur in the combustion tube. (2 marks) - Below are the bond dissociation energies of some elements.
Bond
Bond dissociation energy
C – C
343 kJ mol-1
C – H
414 kJ mol-1
H – H
435 kJ mol-1
C = C
612 kJ mol-1
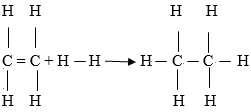
Use this information to calculate the heat of reaction for (3 marks)
C2H4(g) + H2(g) → C2H6(g) - Sulphur (IV) oxide is oxidized catalytically to sulphur (VI) oxide in the reaction.
2SO2(g) + O2(g) → 2SO3(g) ΔH = -197kJ- What information about the reaction is given by ∆H = -197kJ ? (1 mark)
- Name one catalyst that can be used in this reaction. (1 mark)
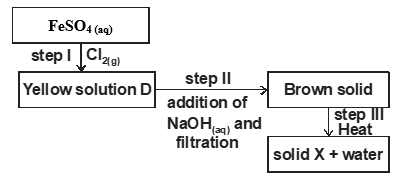
- Study the scheme below and answer the questions that follow.
- Write the formula of the cation present in solution D. (1 mark)
- What property of chlorine is shown in step 1. (1 mark)
- Write an equation for the reaction which occurred in step III. (1 mark)
- 0.63g of lead powder were dissolved in excess nitric (V) acid to form lead (II) nitrate solution. All the lead (II) nitrate was then reacted with sodium sulphate solution.
- Write an ionic equation for the reaction between sodium sulphate solution and lead (II) nitrate solution. (1 mark)
- Determine the mass of the lead salt formed in the reaction in (a) above (Pb = 207, S = 32, O = 16) (2 marks)
- Use the cell representation below to answer the questions that follow.
Cr(s)/Cr3+(aq)//Fe2+(aq)/Fe(s)- Write an equation for the cell reaction. (1 mark)
- If the emf of the cell is 0.30V and the E0 value for Fe2+/Fe(s) is -0.44V. Calculate the E0 value for Cr(s)/Cr3+(aq) (2 marks)
- An element Q has a relative atomic mass of 88. When a current of 0.5A was passed through the fused chloride of Q for 32 minutes and 10 seconds, 0.44g of Q were deposited at cathode. Determine the charge on the ion of Q. (1 Faraday = 96500 coulombs) (3 marks)
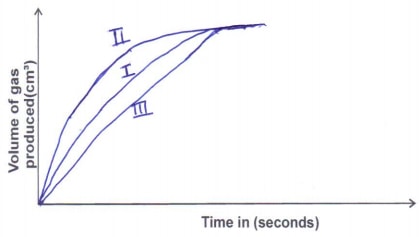
- The table below gives three experiments on the reaction of excess sulphuric (VI) acid and 0.5g Zinc done under different conditions. In each case the volume of gas liberated was recorded at different time intervals.
Experiment
Form of Zinc
Sulphuric (VI) acid
I
Powder
0.8M
II
Powder
1.0M
III
Granules
0.8M
On the axes below, draw and label the three curves that would be obtained from the results above. (3 marks) -
- Starting with red roses, describe how a solution containing the red pigments may be prepared? (2 marks)
- How can the solution be shown to be an indicator. (1 mark)
- The table below provides data on the successive ionisation energies of carbon.
Ionisation numbers
1st
2nd
3rd
4th
5th
6th
Ionisation energy (kJ/mol)
1090
2350
4610
6220
37800
47300
- Explain why each ionisation energy increase in nature. (2 marks)
- Write an equation for the 5th ionisation energy of carbon. (1 mark)
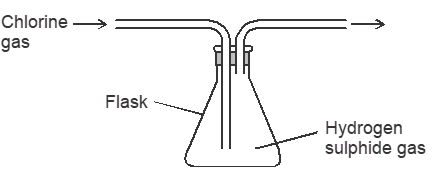
- The figure below was set by a student to investigate the reaction between chlorine gas and hydrogen sulphide gas.
- Write an equation for the reaction that took place in the flask. (1 mark)
- What observation was made in the flask ? (1 mark)
- What precaution should be taken in carrying out the experiment ? (1 mark)
- A certain carbonate, QCO3, reacts with dilute hydrochloric acid according to the equation given below.
QCO3(s) + 2HCl (aq) → QCl2(aq) + CO2(g) + H2O(l)
If 1g of the carbonate reacts completely with 20cm³ of 1M hydrochloric acid. Calculate the relative atomic mass of Q.(C = 12.0, O=16.0) (3 marks) - When bismuth (III) chloride is added to water, a reaction occurs and a white precipitate forms as shown below.
BiCl3(aq) + H2O(l) ⇌ BiOCl(s) + 2HCl(aq)
What would be the effect on the amount of the precipitate formed if sodium hydroxide solution is added to the equilibrium mixture? Explain your answer. (2marks) -
- State the Gay Lussac's Law. (1 mark)
- 10cm3 of a gaseous hydrocarbon, C2Hx required 30cm3 of oxygen for complete combustion. If steam and 20cm3 of carbon (IV) oxide were produced, what is the value of X? (2 marks)
-
- Give Bronsted and Lowry definition of an acid (1mk)
- Differentiate between a strong acid and a concentrated acid (2mks)
- When a hydrated sample of CaSO4.xH2O was heated until all water was lost, the following data was recorded
Mass of crucible = 30.296 g
Mass of crucible + hydrated salt = 33.111 g
Mass of crucible + anhydrous salt = 32.781 g
Determine the empirical formula of the hydrated salt. (CaSO4=136, H2O=18). (3marks) - Describe a chemical test used to distinguish butane from butene in the laboratory. (2marks)
- The table below gives the atomic numbers of elements W, X, Y and Z.
Element
W
X
Y
Z
Atomic number
14
17
16
19
- Name the type of bonding that exist in the compound formed when X and Z reacts. (1mark)
- Select the letter representing the strongest reducing agent. Give a reason for your answer. (2mks)
- In an electrochemical cell, the standard hydrogen electrode uses platinized platinum. State three functions of the platinized platinum. (3 marks)
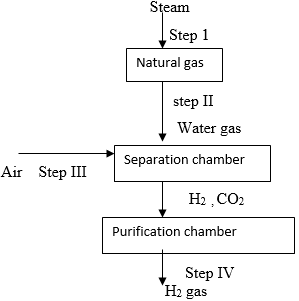
- The flowchart below shows the scheme for extraction of Hydrogen from hydrolysis of natural gas, study it and answer the questions that follow.
- In step II water gas is formed. State one use of water gas. (1marks)
- When air is added in step III CO is converted to CO2 name one chemical substance that can be used to separate CO2 from H2 in step IV (1marks)
- State one large scale use of Hydrogen gas formed. (1marks)
- Aluminium is obtained from the ore with the formula Al2O3.2H2O. The ore is first heated and refined to obtain pure aluminium oxide (Al2O3). The oxide is then electrolysed to get Aluminium and oxygen gas using carbon anodes and carbon as cathode.
- Give the common name of the ore from which aluminium is extracted. (1 mark)
- What would be the importance of heating the ore first before refining it ? (1 mark)
- The refined ore has to be dissolved in cryolite first before electrolysis. Why is this necessary? (1 mark)
- Why are the carbon anodes replaced every now and then in the cell for electrolysing aluminium oxide? (1 mark)
- Nylon polymer has the structure below.
- Determine the structures of the monomers. (2mks)
- State the type of polymerization. (1mk)
-
- Define the term solubility. (1 mark)
- The following were the results obtained in an experiment to determine solubility of potassium nitrate at room temperature.
Mass of evaporating dish = 14.32 g
Mass of evaporating dish + saturated solution = 35.70 g
Mass of evaporating dish + salt (residue) = 18.60 g
Calculate the solubility of potassium nitrate from the above results. (2 marks)
- Describe a simple laboratory experiment that can be used to distinguish between sodium sulphide and sodium carbonate. (2mks)
-
- Give one reason some of the laboratory apparatus are made of ceramics. (1 mark)
- Name two apparatus that can be used to measure approximately 75 cm3 of dilute sulphuric (VI) acid. (2 marks)

Marking Scheme
-
-
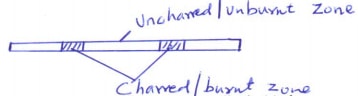
- Charred part is the hottest zone as a result of complete combustion of gases while uncharred zone is as a result of incomplete combustion of gases
-
-
-
No. of half lives
= 35/7
= 5 half lives
R = (1/2)n × original mass
R = (1/2)n × 100
= 1/32 × 100
= 3.125g
Alternatively
1st 2nd 3rd 4th 5th
100 — 50 — 25 — 12.5 — 6.25 — 3.125 -
- S - Beta (β) particles
R - Alpha (α) particles - Alpha. Has greater ionising effect
- S - Beta (β) particles
-
- - Heat copper metal in air to obtain copper(II)oxide
- React CuO with dilute HNO3 to obtain Cu(NO3)2 solution
- Filter to obtain Cu(NO3)2
- React Cu(NO3)2 with Na2CO3 to obtain CuCO3 and NaNO3
- Filter to obtain CuCO3 as residue and NaNO3 as filtrate
- Dry between the filter papers to get CuCO3(s) - 2C(s) + O2(g) → 2CO(g)
Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO2(g) -
ΔH = BBE + BFE
BBE
C = C = 612
C − C = 414 × 4 = 1656
H − H = 435
2703
BFE
C − C = 343
C − H = 414 × 6 = 2484
+2827
ΔH = -2703 + 2827
= +124 KJ/mol -
- Endothermic reaction
- Vanadium(V) oxide (Reject V2O5)
-
- Fe2(SO4)3
- Oxidising agent
- 2Fe(OH)3(s) → Fe2O3(s) + 3H2O(l)
-
- Pb2+(aq) + SO4(aq) → PbSO4
- Moles of Pb = 0.63/207 = 0.0030 moles
M.R. = 1:1
Moles of PbSO4 = 0.00304
Mass = 0.00304 × 303
= 0.92112g
-
- Fe2+(aq) + Cr(s) → Cr3+(aq) + Fe(s)
- Eº = EºRed − EºOxi
0.30V = − 0.44 − EºOxi
EºCr/Cr3+ = − 0.44 − 0.30
= − 0.74 V
- Mass = MIT/zF
0.44g = 88 × 0.5 × 1930
z × 96500
0.44 = 0.88/z
0.44z = 0.88
0.44 0.44
z = + 2
Q2+
Alternatively
Q = It
= 0.5 × 1930
= 965 C
0.44 = 965
88 = ?
88 × 965 = 193000
0.44 96500
= +2 -
-
- - Crush roses using pestle and mortar
- Add propane to dissolve the red pigments
- Filter to obtain red pigment solution as filtrate - Add drops of the pigment to different types of acids or bases. It will show some colour on acids and different but same colour on bases
- - Crush roses using pestle and mortar
-
- After the removal of an electron, the overall nuclear charge hold the remaining electrons firmly
-
- H2S(g) + Cl2(g) → S(s) + 2 HCl(g)
- Yellow deposits of sulphur is formed
- 1 mole → 100cm3
→ 20cm3
20×1 = 0.02 moles
1000
Mole ratio QCO3 : HCl
1 : 2
Moles of QCO3 = 0.02/2 = 0.01 moles
RFM = Mass/RFM
= 1/0.01 = 100
100 = Q + 12 + 48
100 = Q + 60
Q = 40 - White precipitate increases/intesifies; When hydroxide is introduced it reacts with H+ from the HCl hence system shift from left to right.
-
- When gases react, they do so in volumes that bear a simple whole number ratio to one another and to those of products if gaseous at constatnt remperature and pressure
- C2Hx(g) + 3O2(g) → 2CO2(g) + 2H2O(g)
10cm3 30cm3 20cm3
x = 4
-
- An acid is a proton donor
- STrong acid is type of acid that dissociate fully into its ions while concentrated acid is on acid has more H+ ions
- Mass of hydrated salt = 2.815
Mass of anhydrous salt = 2.485
Mass of water = 0.33
Compound CuSO4 H2O
Mass 2.485 0.33
RFM 136 18
No. of moles 0.01827 0.0183
0.018271 0.01827
Mole Ratio 1 : 1
CuSO4.H2O - - Add bromine water to butane and butene in different test tubes
- Test-tube containing butene will decolorize bromine water while butane does not. -
- Ionic
- Z, has the largest atomic radius/lowest ionization energy
- Performed in fume chamber/open air chlorine is poisonous gas.
-
- Acts as an inert metal connection to the H2/H+(aq) system
- Provides a surface area on which dissociation of H2 molecules take place
- Serves as an electrical conductor to the rxternal circuit
-
- - In fuel
- In cells - Calcium hydroxide
- Any correct
- - In fuel
-
- Bauxite
- To lower the melting point
- They are eaten away due to reaction with oxygen unde high temperature.
-

-
- The maximum amount of solute that can satuarte 100 g of solvent at a particular temperature
- Mass of solute = 18.60 − 14.32 = 4.28
Mass of solvent = 35.70 − 18.60 = 17.1g
4.28g → 17.1 of solvent
100g of solvent × 4.289 = 25.03g/100g of solvent
17.1
- - React with acid separately
- Test the gas produced using acidified K2Cr2O7
- Sulphide produces a gas that turns ornage acidified K2Cr2O7 green while for carbonate it ramins orange
(Accept any other correct) -
- Can withstand strong heating
- - 100cm3 measuring cylinder
- 100cm3 beaker
Download Chemistry Paper 1 Questions and Answers - Kassu Jet Joint Exams 2020/2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students