-
- Define radioactivity (1mark)
- Give two differences between chemical reactions and nuclear reactions. (2marks)
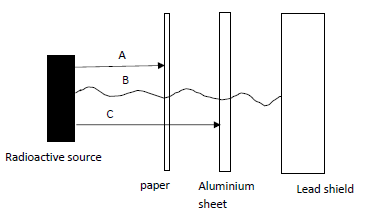
- Study the diagram below and answer the questions that follow
- What property of radiations is being investigated by the illustration above 1mark)
- Give the name of the radiation B and give a reason. (1mark)
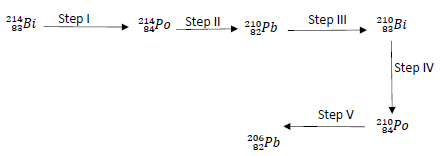
- B below is the radioactive decay starting with 21483Bi, study it and answer the questions that follow.
- Identify the radiations emitted at:
- Step I ………………………………………………………………… (1mark)
- Step V ………………………………………………………………… (1mark)
- Write a nuclear equation for step II (1mark)
- Identify the radiations emitted at:
- State one danger associated with frequent exposure to radiations. (1mark)
-
- The amount of salt A that can dissolve in water at different temperatures is shown in the table below
Temperature (oC) 0 10 20 30 40 60 80 90 Solubility of salt A g/100g of water. 36.1 35.5 34.8 34.2 33.7 32.6 31.4 30.9 - Draw a graph of solubility of salt A against temperature. (3marks)
- How does solubility of salt A vary with temperature? (1mark)
- From the graph determine the:
- Solubility when the temperature would be 500C (1mark)
- Temperature at which the solubility will be 31.8g/ 100g of water. (1mark)
- State one industrial application of solubility (1mark)
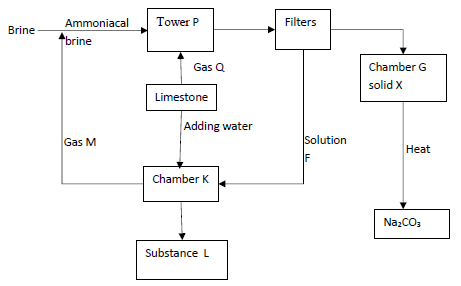
- The flow chart below shows industrial manufacture of sodium carbonate by solvay process. Study it and answer the questions that follow.
- Name
- Gas Q …………………………………………………………… (½mark)
- Gas M …………………………………………………………… (½mark)
- Solution F ……………..………………………………………… (½mark)
- Substance L ……………………………………………………… (½mark)
- Write equations for the reactions that occurred;
- Chamber K (1mark)
- Heating solid X (1mark)
- Give one use for each of the compounds:
- Substance L (1mark)
- Na2CO3 (1mark)
- Name
- The amount of salt A that can dissolve in water at different temperatures is shown in the table below
-
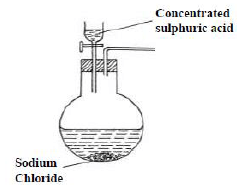
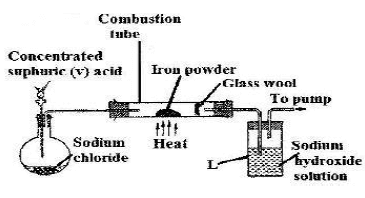
- The diagram below was a setup used by a form three student in Kapsabet Boys to prepare, dry and collect hydrogen chloride gas.
- Complete the diagram to show how a sample of hydrogen chloride gas was prepared, dried and collected. (3marks)
- Write a balanced chemical equation to shows how hydrogen chloride is formed in the flask. (1mark)
- Dry hydrogen chloride gas was passed through hot iron filings as shown below.
- State and explain observation that would be made in the combustion tube. (2marks)
- What is the purpose of having sodium hydroxide in beaker labelled L. (1mark)
- In the experiment above 600cm3 of hydrogen chloride gas were used completely. Determine the mass of the product that would be formed in the combustion tube.
(Fe = 56.0, Cl =35.5, Molar Gas Volume =22.4 litres at s.t.p) (3marks)
- The diagram below was a setup used by a form three student in Kapsabet Boys to prepare, dry and collect hydrogen chloride gas.
-
- A sample of an ore was suspected to have a compound of iron, describe how it can be established that the ore contains iron. (3marks)
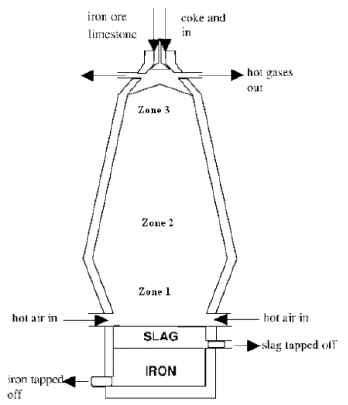
- The diagram below represents a blast furnace used in the extraction of iron. Study it and answer the questions that follow.
- Name two ores from which iron can be extracted. (2marks)
- Write chemical equations for the reactions that occur in the zones:
Zone 1 (1mark)
Zone 2 (1mark)
Zone 3 (1mark) - What are the two impurities found in the ore of iron? (1mark)
- Using chemical equations explain how the impurities are removed from
the ore. (3marks) - State the effect of the gases produced in this process on the environment. (1mark)
-
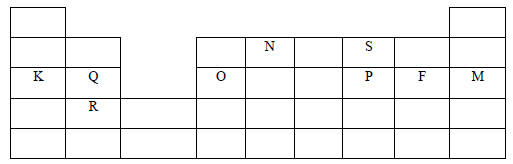
- The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters used are not the actual symbols of the elements.
- What name is given to the group of elements to which Q and R belong? (1mark)
- Write a chemical equation for the reaction that would occur when R and F react. (1mark)
- Compare the atomic radius of O and P. (2marks)
- Draw a dot (.) and across (X) diagram for the compound formed between N and F (2marks)
- Describe how a pure sample of carbonate of K can be obtained from a mixture with lead (II) carbonate. (3marks)
- The melting point of silicon (IV) oxide is 1728oC while that of sulphur (IV) oxide is -76oC. Explain (2marks)
- The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters used are not the actual symbols of the elements.
-
- Draw and give names of the structures of the compounds below.
CH3CHCHCH3 (1mark)
CH3CH2CH2COOH (1mark) - The formular below represent a type of detergent.
C17H35COONa- Name the type of detergent represented (1mark)
- Give one advantage and one disadvantage of using the detergent in (i)
Above. (2marks)
- Compound P whose formular is given below was a product of a reaction between compound M and compound N.
CH3CH2COOCH2CH3- Draw the structures of compounds M and N
M (1mark)
N (1mark) - Name the process that took place for formation of compound P. State the conditions necessary for the process named. (2marks)
- Draw the structures of compounds M and N
- Compound Q has empirical formula CH2 and molecular mass 42:
- Determine the Molecular mass of Q. (1mark)
- Draw a structure of polymer having three units of the structure in (i) (1mark)
- Draw and give names of the structures of the compounds below.
-
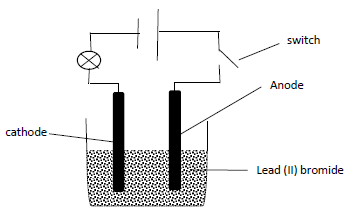
- The diagram below was used in electrolysis of lead (II) bromide. Study it and answer the questions that follow.
After the switch was closed, the bulb did not light. Explain. (1mark)- Write ionic equations for the reactions that occurred at:
- anode (1mark)
- Cathode (1mark)
- State the precaution that should be taken during carrying out this
experiment. (1mark) - During the electrolysis above 51.75g of lead was deposited in 3hours, determine the amount of current that was used.(1F= 96500 coulombs,
Pb = 207) (3marks)
- Write ionic equations for the reactions that occurred at:
- Use the reduction potentials given below to answer the questions that follow.
Reaction EƟ (volts) A2+(aq) + 2e- → A(s) -0.76 B2+(aq) + 2e- → B(aq) -0.44 C+(aq) + e- → C2 (g) 0.00 D2+(aq) + 2e- → D(s) +0.34 ½E2(g) + e- → E-(aq) +1.09 - Identify the strongest reducing agent. Give a reason. (1mark)
- Give two half-cell reactions that would produce the largest e.m.f when connected. (1mark)
- Draw and label an electrochemical cell that can be obtained when the two half-cell reactions mentioned above would be connected (3marks)
- Is it possible to store a solution containing ions of A in a container made of B? (1mark)
- The diagram below was used in electrolysis of lead (II) bromide. Study it and answer the questions that follow.
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions No Answers - Kapsabet Mocks 2020/2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students