INSTRUCTIONS TO CANDIDATE
- Write your name and admission number in the spaces provided.
- Sign and write the date of examination in the spaces provided
- Answer all the questions in the spaces provided
- All working must be shown where necessary
- Electronic calculators and mathematical tables may be use.

QUESTIONS
- The table below gives information about some elements. The letters do not represent the actual symbols of the elements. Study the information and answer the questions that follow.
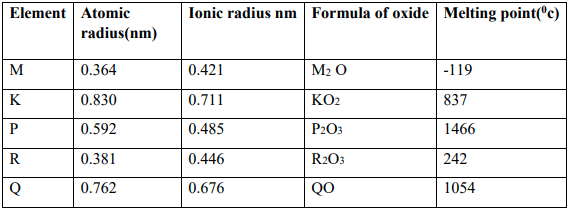
- Identify two elements that are non-metals. Give a reason? (2mks)
-
- State the valency of elements Q and R. (1mk)
Q………………………………………………………………………………………………..
R………………………………………………………………………………………………. - Write a formula of a compound formed when Q combines with M. (1mk)
- What type of bond exists between Q and K. (1mk)
- State the valency of elements Q and R. (1mk)
- The melting point of the oxide of R is lower than that of the oxide of P. Explain. (2mks)
- Identify two elements that would react with each other most vigorously. Give a reason(2mks)
- Element P would be suitable for making utensils for boiling water. State two properties that make the element suitable for the use. (2mks)
-
- The table below gives the solubility of hydrated Copper (II) Sulphate in moldm-3 at different temperatures.
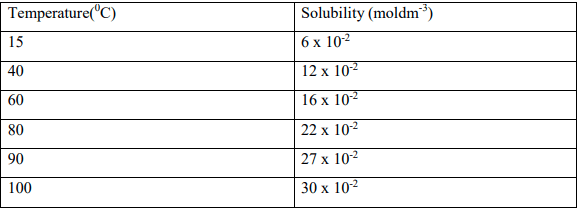
- On the grid provided plot a graph of solubility of Copper(II) Sulphate (vertical axis) against temperature. (3mks)

- From the graph, determine the mass of copper(II) Sulphate pentahydrate deposited when the solution is cooled from 90º
c to 60º (Cu=64, S=32, O=16, H=1). (3mks)
- On the grid provided plot a graph of solubility of Copper(II) Sulphate (vertical axis) against temperature. (3mks)
- In an experiment to determine the solubility of sodium chloride ,5.0 cm3 of a saturated solution of sodium chloride weighing 5.35g were placed in a volumetric flask and diluted to a total volume of 250cm3.
25.0 cm3 of the dilute solution of sodium chloride completely reacted with 24.1 cm3 of 0.1 M silver nitrate solution.
Ag NO3(aq) + NaCl(aq) → Ag Cl(s) + NaNO3(aq)
Calculate;- Moles of silver nitrate in 24.1cm3 of solution. (1mk)
- Moles of sodium chloride in 25.0cm3 of solution. (1mk)
- Moles of sodium chloride in 250cm3 of solution. (1mk)
- Mass of sodium chloride in 5.0cm3 of saturated chloride solution (Na=23.0 Cl=35.5). (1mk)
- Mass of water in 5.0 cm3 of saturated solution of sodium chloride. (1mk)
- The solubility of sodium chloride in g/100 g of water. (2mks)
- The table below gives the solubility of hydrated Copper (II) Sulphate in moldm-3 at different temperatures.
- In order to find the portion by volume of one of the main constituents of air, a sample of air was passed through two wash bottles; the first containing aqueous sodium hydroxide and the second containing concentrated Sulphuric (VI) acid and was then collected in a gas syringe.
- Suggest a reason for passing air through:
- Aqueous sodium hydroxide. (1mk)
- Concentrated Sulphuric (VI) acid. (1mk)
- The volume of the gas collected in the syringe was 80cm3 . This was passed several times over hot copper powder until no further contraction of volume took place. After cooling to the original temperature, the volume was found to be reduced to 63.2cm3
- Explain the change in appearance of copper powder as the experiment progressed. (2mks)
- Calculate the percentage of the air used up. (2mks)
- Name the main gas remaining in the syringe. (1mk)
- Explain the purity of the main gas remaining in the syringe. (2mks)
- A piece of sodium metal was exposed to air for some time. Write an equation for any one reaction that occurred. (1mk)
- Suggest a reason for passing air through:
- Study the flow chart below and answer the questions that follow.
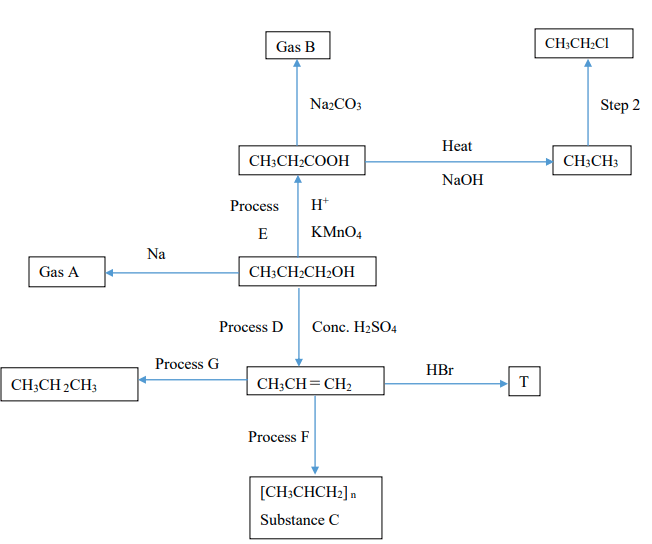
- Name:
- Gas A ……………………………………………………………………………. (1mk)
- Gas B ……………………………………………………………………………. (1mk)
- Substance C ….…………………………………………………………………. (1mk)
- Give the structural formula and name of T (2mks)
Structural Formula Name………………………………………… - Identify process
D …………………………………………………………………………………………. (1mk)
E …………………………………………………………………………………………. (1mk)
F …………………………………………………………………………………………. (1mk)
G …………………………………………………………………………………………. (1mk) - Name the condition and reagent for Step 2 (2mks)
Condition ………………………………………………………………………………........................
Reagent ……………………………………………………………………………………………….. - Write equation for the complete combustion of the product of process G. (1mk)
- To what class of compounds does the products of process D and E belong?
D …………………………………………………………………………………………. (1mk)
E …………………………………………………………………………………………. (1mk) - If the relative molecular mass of C is 63,000, determine n (C = 12, H = 1) (1mk)
- Name:
-
- The standard electrode potential for the elements chlorine and magnesium are: -
Cl2(g) + 2e- → 2Cl-(aq) Eθ + 1.36V
Mg2+(aq) + 2e- → Mg(s) Eθ - 2.36V- Which one of the two elements will act as an oxidizing agent? Explain. (2mks)
- Calculate the electromotive force of a cell where the overall reaction is: - (1mk)
Cl2(g) + Mg(s) Eθ + MgCl2(s)
- The table below gives the reduction standard electrode potentials for divalent metals.
The letters are not their actual symbols. Use them to answer the questions that follow: -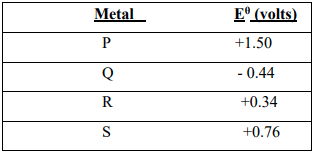
- Select two metals whose half cells can produce the highest voltage when connected.(1mk)
- Draw a well labelled diagram of electrochemical cell formed by half-cells of metals P and Q. (2mks)
- Calculate the voltage produced by the cell in (ii) above. (1mk)
- When nitrate solution of a certain metal X was electrolysed, 1.174g of metal X was deposited by a current of 4 amperes flowing for 16minutes. Determine the formula of the metal nitrate. (1F= 96,500, R.A.M of X= 59). (3mks)
- The standard electrode potential for the elements chlorine and magnesium are: -
-
- Below is a simplified diagram of the Down’s cell used for the manufacture of sodium. Study it and answer the questions that follow.
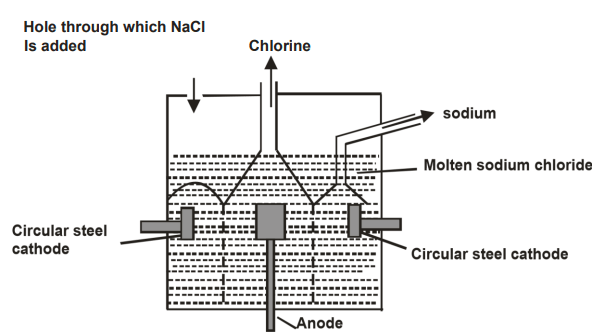
- What material is the anode made of? Give a reason. (2mks)
- What precautions is taken to prevent chlorine and sodium from re-combining.(1mk)
- Write an ionic equation for the reaction in which chlorine gas is formed. (1mk)
- In the Down’s process, a certain salt is added to lower the melting point of sodium chloride from about 800ºc to about 600ºc.
- Name the salt that is added. (1mk)
- State why it is necessary to lower the temperature. (1mk)
- Explain why aqueous sodium chloride is not suitable as an electrolyte for the manufacture of sodium in the Downs process. (2mks)
- State two uses of sodium metal. (2mks)
- Below is a simplified diagram of the Down’s cell used for the manufacture of sodium. Study it and answer the questions that follow.
- The set-up below was used to prepare dry sample of hydrogen Sulphide gas
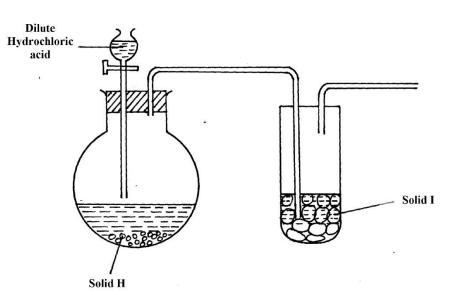
-
- Complete the diagram to show how the gas was collected. (1mk)
- Identify the following: - (2mks)
- Solid H ……………………………………………………………………………………
- Solid I ………………………………………………………………………………………..
- Write an equation for the reaction that occurred in the flask between solid H and dilute Hydrochloric acid. (1mk)
- When hydrogen Sulphide gas was passed through a solution of Iron (III) chloride, the following observations were made: -
- the color of the solution changed from reddish-brown to green and
- a yellow solid was deposited. Explain the observations. (2mks)
- In the manufacture of Sulphuric (VI) acid by contact process Sulphur (IV) oxide is made to react with air to form Sulphur (VI) oxide as shown: -
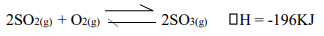
- Name the catalyst in this reaction. (1mk)
- State and explain the effect of the following changes on the yield of Sulphur (VI) oxide
- Increasing the pressure. (1mk)
- Using a catalyst. (1mk)
- Explain why sulphur (VI) oxide gas is absorbed in concentrated sulphuric (VI) acid before dilution. (1mk)
-

MARKING SCHEME
-
- M 1/ mk and R1/2
The ionic radius is larger 1mk than the atomic radius implying they gain electrons. -
- Q=2 1mk
R=3 1mk - QM2 1mk
- Metallic (1mk
- Q=2 1mk
- The oxide of P is ionic (1mk) with a giant ionic structure that requires a lot of energy to break, the oxide of R is molecular. 1mk
- M and K 1mk
M is a non-metal with the smallest (½mk) atomic radius hence most electronegative K is a metal with the largest atomic radius (½mk) hence most electropositive. - Has high melting point (1mk) and good conductor (1mk) of heat being a metal.
- M 1/ mk and R1/2
-
-
- - scale(1mk)
-Plotting all points correctly (1mk)
- Curve (1mk) - 0.188- 0.12 = 0.068 mol (1mk)
Therefore, mass of hydrated copper(II) sulphate
= 0.068 x 250 (1mk)
= 17g (1mk)
- - scale(1mk)
-
- Moles of AgNO3 = 0.1 x 24.1 = 2.41 x 10-3 (1mk)
1000 - Moles of NaCI = Moles of AgNO3
= 241 x 10 -3 (1mk) - Moles of NaCl in 250cm3 = 41 x 10-3 x 250
25
=2.41 x 10-2 (1mk) - R.F.M Na CI = 23 + 35 .5 = 58.5
Mass of NaCl in 5cm3 = 2.41 x 10-2 x 58.5
= 1.41g (1mk) - Mass of water = 5.35 – 1.41
= 3.94g (1mk) - 3.94 of water contains 1.41g of NaCl
100 g of water = 1.41 x 100 (1mk)
3.94
=35.7 (1mk)
- Moles of AgNO3 = 0.1 x 24.1 = 2.41 x 10-3 (1mk)
-
-
-
- To remove carbon(iv) (1mk)
- To dry the air (1mk)
-
- Turn from red brown to black (1mk)
Copper is oxidized to copper (ii) oxide (1mk) - 80-63.2cm3 = 16 .8 cm3 (1mk)
16.8 × 100
80
= 21% (1mk) - Nitrogen (1mk)
- Impure (1mk) it contains noble gases like (1mk)
- Turn from red brown to black (1mk)
- 4Na (s) + O2(g) → 2Na2O(g)
2Na (s) + O2(g) → Na2O2(g)
-
-
- A: Hydrogen Gas (1mk)
B: Carbon (IV) oxide gas. (1mk)
C: Polypropene. (1mk)
H H H
| | | (1mk)
H—C—C—C—H
| | |
H Br H 2 – bromopropane (1mk)- D: Dehydration (1mk)
E: Oxidation (1mk)
F: Polymerization (1mk)
G: Hydrogenation/additional hydrogenation (1mk) - Condition: Sunlight/U.V rays/U.V light (1mk) Reagent: Chlorine (1mk)
- C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(l) (1mk)
- D: Alkenes. (1mk)
E: Alkanoic acids. (1mk) - (CH3CHCH2)n = 63000
42n = 63000
n = 1500. (1mk)
- A: Hydrogen Gas (1mk)
-
-
- Chlorine (1mk) Has a higher reduction potential (1mk)
- +1.36 - (- 36) = +3.72
-
- P and Q
- +1.50 – (-0.44) += + 1.94v
- Q = 4 X 16 X 60 = 3840C (1mk)
1.174g ....................3840
59 g...............59 × 3840 = 192,981.261 C (1mk)
1.174
If 96,500C.................................IF
192891.261C.....................192981.261 × 1
96500
Charge of X = +2 Formula X(NO3)2 (1mk)
-
-
-
- Graphite/ Titanium (1mk). They do not react with chlorine gas (1mk)
- A steel diaphragm is suspended between the (1mk)
- 2Cl- (aq) → Cl2(g) + 2e (1mk)
-
- Calcium Chloride (1mk)
- To reduce the cost of production. (1mk)
- Hydrogen ions will be preferentially discharged at the expense of sodium ions at the cathode. (1mk)
At the anode, hydroxyl ions will be preferentially discharged at the expense of chloride ions (1mk) - making sodium compounds such as sodium cyanide which is used in extraction of gold (1mk) sodium and potassium alloy is used as a coolant in nuclear reactors (1mk)
-
-
-
 (1mk)
(1mk)-
- iron II Sulphide or copper II Sulphide (1mk)
- anhydrous Calcium Chloride. (1mk) (Reject Calcium chloride)
- FeS(s) + HCl(aq) FeCl2(aq) + H2S(g) (1mk)
- Fe3+ is reduced or Fe2+ (1mk)
H2S is oxidized to (1mk) -
- Vanadium V oxide or platinised asbestos (1mk)
-
- The yield of SO3 increase because increase in pressure favour the forward reaction since less number of molecules of SO3 (1mk)
- The yield of SO3 is the same because catalyst only speeds the rate at which equibrium is attained. (1mk)
- Exothermic reaction occurs. When dissolved in water produce acid spray (fumes) cause pollution. (1mk)
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry P2 Questions and Answers - Nambale Mock Exams 2021/2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

