CHEMISTRY
PAPER 3
(PRACTICAL)
INSTRUCTIONS TO CANDIDATES
- Write your name and index number in the spaces provided.
- Sign and write the date of examination in the spaces provided.
- Answer ALL questions in the spaces provided in the question paper.
- You are NOT allowed to start working with the apparatus for the first
- 15 minutes of the 2¼ hours allowed for this paper. This time is to enable you to read the question paper and make sure you have all the chemicals and apparatus you may need.
- Mathematical tables and silent electronic calculators may be used.
- All working must be clearly shown where necessary.
- This paper has 8 printed pages.
- You are provided with :
- 2M hydrochloric acid, solution
- 5 pieces of magnesium ribbon, each 2cm long
- 0.5M sodium carbonate, solution R
You are required to determine:- the rate of reaction between hydrochloric acid and magnesium
- the mass of 2cm of magnesium ribbon
- Procedure 1
- Using a clean measuring cylinder, measure 60cm 3 of 2M hydrochloric acid, solution W and place it into a clean conical flask. Take one piece of 2cm piece of magnesium ribbon provided and place it in the hydrochloric acid and immediately start the stopwatch.
- Measure and record the time taken for the magnesium ribbon to react completely with hydrochloric acid in table 1 below.
Retain the contents of conical flask 1 for use in procedure II. Label this solution P - Repeat the procedure using 50cm 3 , with 10 cm 3 of water , 40 cm 3 and 20cm3 of water ,, 30cm3 and 20cm3 of water , portions of 2M hydrochloric acid adding distilled water to make up to 60cm3 of solution and complete the table below.
NOTE: Do not retain the contents of the conical flask in experiments 2, 3, 4 and 5. Table 1
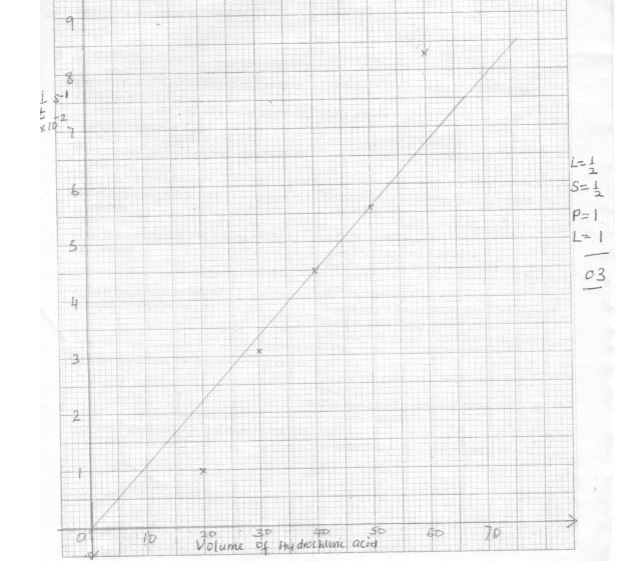
(6mks)Experiment number 1 2 3 4 5 Volume of 2M hydrochloric acid(cm 3) 60 50 40 30 20 Volume of water 0 10 20 30 40 Time taken for ribbon to disappear (s) 1/time (sec -1) - Plot a graph 1/time (sec -1 ) against volume of 2M hydrochloric acid (3mks)
- From your graph, determine the time taken for the ribbon to disappear when 36cm 3 of 2M hydrochloric acid were used. (1mks)
- In terms of rate of reaction, explain the shape of your graph. (1 mks)
- Procedure II
- Using a clean measuring cylinder measure 40cm 3 of distilled water and add it to the contents of the conical flask retained in procedure I labeled solution P
- Fill a burette with solution P. Pipette 25.0cm 3 of solution R 0.5M sodium carbonate and place it into a clean flask. Add 2-3 drops of methyl orange indicator to solution R. Titrate with solution P and record your results in table II below. Repeat the titration two more times and complete the table.
Table II
(4mks)1 2 4 Final volume(cm3 ) Initial volume (cm3 ) Final- initial volume (cm3 )
Calculate the :- Average volume of solution P used. (1 mks)
- Number of moles of sodium carbonate solution R used. (1 mks)
- Number of moles of hydrochloric acid used. (1 mks)
- Number of moles of hydrochloric acid present in 100cm 3 of solution P. (1 mks)
- Number of moles of hydrochloric acid present in 60cm 3 of solution W. (1 mks)
- Number of moles of hydrochloric acid that reacted with 2cm magnesium ribbon. (1 mks)
- Mass of magnesium present in 2cm ribbon. (Mg = 24.0) (1 mks)
- You are provided with solid K. Carry out the following tests below. Write your observations and inferences in the spaces provided.
- Place about one half of solid K in a dry test tube. Heat gently then strongly. Test any gases produced with blue and red litmus papers
Observation Inference - Place the remaining amount of solid K in a boiling tube. Add about 10cm3 of distilled water and shake the mixture.
Observation Inference - Divide solution K above into four portions of 2cm3 each in separate test tubes. Use the portions for tests (i) to (iv) below.
- To the first portion add sodium hydroxide solution dropwise until in excess.
Observation Inference - To the second portion add about 1cm 3 of sodium chloride solution
Observation Inference - To the second portion add aqueous ammonia drop wise till excess
Observation Inference - To the second portion add three drops of aqueous barium nitrate followed by five drops of nitric (v) acid .
Observation Inference
- To the first portion add sodium hydroxide solution dropwise until in excess.
- Place about one half of solid K in a dry test tube. Heat gently then strongly. Test any gases produced with blue and red litmus papers
- You are provided with an organic substance S. Carry out the following tests and record your observations and inferences in the spaces provided.
- Place about one third of substance S on a metallic spatula and ignite it with a bunsen burner flame.
Observation Inference - Place the remaining amount of substance S in a boiling tube. Add about 10cm3 of distilled water and shake well. Use about 2cm3 portions of the mixture obtained for tests (i) to (iii) below.
- To the first portion add solid sodium hydrogen carbonate.
Observation Inference - To the second portion add two drops of acidified potassium manganite (VI) solution.
Observation Inference - To the third portion add 3 drops of bromine water
Observation Inference
- To the first portion add solid sodium hydrogen carbonate.
- Place about one third of substance S on a metallic spatula and ignite it with a bunsen burner flame.
CONFIDENTIAL
Instructions to schools;
The information contained in this paper is to enable the head of the school and the teacher in charge of chemistry to make adequate preparation for Chemistry practical examination. NO ONE ELSE should have access to this paper or acquire knowledge of it’s content. Great care must be taken to ensure that the information herein does NOT reach the candidates either directly or indirectly. The teacher in charge of chemistry should not perform any of the experiments in the same room as the candidates nor make the results of the experiments available to the candidates or give any other information related to the
experiment to the candidates. Doing so will constitute an examination irregularity which is PUNISHABLE.
In addition to the normal laboratory fittings and apparatus, each candidates should have
the following:
Requirements;
- 250cm³ of solution W; 2.0 HCl (aq)
- Five 2cm long magnesium ribbons.
- 120cm³ of solution R; 0.5M Na2CO3(aq).
- One 100ml and one 10ml measuring cylinders.
- Three conical flasks.
- One stop watch.
- A piece of label
- 500ml distilled water.
- One burette (50ml).
- One 25ml pipette and pipette filler.
- One complete retort stand.
- One white tile.
- About 1.0g of solid K
- Six test tubes and one boiling tube on a test-tube rack.
- Bunsen burner and lighter.
- Blue litmus
- Red litmus
- Two boiling tubes.
- About 0.2g of NaHCO3 solid.
- About 1.0g of solid S
- Metallic spatula
ACCESS TO
- Methyl orange indicator.
- 2M NaOH (aq)
- 0.5M NaCl (aq)
- 2M NH 4 OH (aq)
- 0.5M Ba(NO3)2(aq)
- 2M HNO3(aq)
- MnO-4(aq)/H+(aq)
- Bromine water.
PREPARATIONS:
- Solid K is hydrated aluminium ammonium sulphate.
- Solid S is maleic acid.
- Preparation of KMnO4(aq) / H+(aq)
- Dissolve 3.16 of solid KMnO 4(S) in 200cm³ of 2MH 2 SO 4(aq) making it up to one litre using distilled water.
- Preparation of bromine water.
- Dissolve 1.0ml of bromine liquid in 100cm³ of distilled water.
MARKING SCHEME
- PROCEDURE
Table I
TableExperiment number 1 2 3 4 5 Volume of 2M hydrochloric acid(cm 3) 60 50 40 30 20 Volume of water 0 10 20 30 40 Time taken for ribbon to disappear (s) 12 18 22 32 96 1/time (sec -1) 0.0830 0.0560 0.0450 0.0313 0.0104
Marking areas-
- Complete table - 3 marks
Penalties /conditions- Penalise ½mk for each space not filled.
- Reject fractions for 1/Time and award a max of 1½ mks for the table.
- if fractions appear followed by an extra column of decimals ignore the fractions and award accordingly.
- penalise ½mk each for wrong arithmetic in the values of 1/Time not within an error of ± 2 units in the 3rd decimal place unless it divides exactly.
- Accept reciprocals given to at least 3 decimal places. otherwise penalise ½mk each for rounding off to the 2nd decimal place to a max of 1mark, unless it divides exactly.
- penalise ½mk for every reading <5 and >240 seconds in the time row.
- penalise ½mk for each entry not in seconds.
- Use of decimals - 1 mk (Tied to the 4th row only)
- Accept whole numbers or decimals upto the 2nd decimal place only used consistently, otherwise penalise fully.
- Accuracy - 1 mark (Tied to 4th row only)
- Compare the candidates 1st reading to the school’s value and if within ±2sec, award 1 mark. Otherwise penalise fully.
- Trend - 1 mark
(Tied to 4th row only- Award 1mk if time is continuously increasing otherwise penalise fully
- Complete table - 3 marks
- Graph - 3marks
marking areas- Labelling of both axes - ½mk
- Conditions and penalties.
- Penalise ½mk for wrong units used in any of the axis.
- Penalise ½mk for inverted axes.
- Accept if units not shown, otherwise if shown must be correct.
- Both axes must be labelled.marking areas.
- Scale - ½ mark
- are covered by the actual plots including the origin should be ¾ or more of the squares provided in both axes.
- The scale interval should be consistent.
- Plotting - 1 mark
- award 1mk if all plots are correctly plotted.
- award ½mk if 4 plots correct
- award 0mk if less than 4 plots correctly plotted.
- accept plots even if the axes are inverted.
- accept rounding off the values of 1/Time to the 3rd decimal place when plotting.
- Line - 1 mark
- Accept a straight line passing through at least 2 points.
- Correctly plotted and through the origin (0,0) for 1 mk.
OR
if extrapolated can pass through the origin. (0,0)
- Showing 1/T on the graph - ½ mk
- Stating the correct reading of 1/T at 36cm³ - ½mk
- applying the expression that
Time = 1/correct reading ½mk - correct answer ½mk
- Rate increases with increase in concentration of hydrochloric acid or vice versa - 1mark
OR
Rate of reaction is directly proportion to the concentration.
Conditions.- tied to the correct graph or trend in the table
- If volume is used instead of concentration award full mark.
-
- PROCEDURE II.
Table II
(use school value)
marking areas- Complete table - 1 mark
- Conditions
- award 1mk for complete table with 3 titrations done.
- award ½mk for incomplete table with 2 titrations done.
- Award 0 mark for 1 titration done.
- Penalties
- wrong arithmetic.
- inverted table.
- burette readings beyond 50cm³ unless explained.
- unrealistic titre values i.e. below 1.0cm³ or hundreds.
- NB- Penalise ½mk each. For a maximum of ½mk penalise once.
- Conditions
- use of decimals - 1 mark
(Tied to 1st and 2nd rows only)- Accept either 1 or 2 decimal places used consistently.
- Accept 2 decimals places only if the 2nd decimal place is 0 or 5.
- NB: Penalise fully is any of the conditions is not net.
- Accuracy - 1 mark
(Tied to the school’s value (S.V)
Conditions- If any of the titre values is within ± 0.1 of the S.V award 1mark.
- If any of the 3 titre values is within ± 0.2 of the S.V award ½mk
NB: If there is wrong arithmetic compare the
S.V with the worked out correct value and award accordingly.
- Principles of averaging - 1 mark I + II + III = correct answer
Conditions and penalties.- The values to be averaged MUST be within ±0.2 of each other (i.e. consistent)
- If 3 titrations are done and all are consistent, they must all be averaged.
- If 3 titrations are done and only 2 are consistent and averaged, award 1mk
- If 2 titrations are done and are consistent and averaged award 1mark.
- If the answer is correct but no working is shown award. ½mk
- Final answer - 1 mark
(Tied to the S.V. and to the correct average titre)- Conditions.
- If within ±0.1 of the S.V - 1 mark
- If within ± 0.2 of the S.V - ½ mark
- If wrong values are averaged pick for the candidates correct values. If any and average then for the candidate and award accordingly.
- NB: A total of 5 marks
- Moles of Na2CO3 = 0.5M × 25 ½mk
1000
= correct ans ½mk - Na2CO3(aq) + 2HCl(aq) → 2NaCl(aq) + CO2(g) + H2O(l)
Moles of HCl
= 2 × ans in (ii) above ½mk
= correct answer ½s - Moles of HCl in solution P
= 100cm³ × ans in b(iii) above ½mk
Average volume
= Correct answer ½ - Moles of HCl in 60cm³ of solution W.
= 60 × 2 ½mk
1000
= 0.12 moles ½mk - Moles of HCl reacted with magnesium ribbon.
= Ans in (V) - Ans in (iv) ½mk
= correct ans ½mk - Mass of magnesium present in 2cm ribbon
Mg(S) + 2HCl(aq) → MgCl2 + H2(g) ½mk -
Moles of = 1/2 x ans b(vi) above 1/2 mk= intermediate answer
Mass of Mf= Intermediate x 24 1/2mk = final answer
Conditions/ penalties
- penalise 1/2 mk in the final answer if units are wrong or missing.
Observation Inference a - colourless ✓½ liquid formed on the colour
parts of test tube
- red litmus paper turns blue while blue litmus
paper remains blue ✓½
- white residue is formed ✓½
Award 1mk for any two correct observations
Max. 1mkHydrated ✓½ salt / water of crystallisation
NH + present ✓½
4
Reject: Alkaline gas present
- Ammonia gas producedb Solid dissolves ✓½ to form a colourless✓½
solutionSoluble ✓½ salt / coloured ✓½ ionsAbsent c i White precipitate ✓½ soluble ✓½ in
excessAl 3+ , Zn 3+ , Pb 2+ present
Award ½ if only one mentioned
Penalise ½mk for any contradictory ionii No white precipitate formed ✓1
Reject: no precipitateAl 3+ Zn 2+ present ✓1
Award only if the ion is mentioned in c(i) above
Penalise ½mk if only one ion given
Penalise fully for any contradictory ion giveniii White precipitate ✓½ insoluble ✓ in excess Al 3+ ✓
Award only if the ion is mentioned in c(i)
and c(ii) above
Penalise fully for any contradictory ion giveniv A white precipitate ✓½ that does not dissolve
✓ ½ in nitric (V) acidSO 2- ✓1 4penalise fully for any contradictory ion 3 a Burns with a yellow ✓½ / sooty ✓½ / smoky
flame
Accept: unsaturated organic compound for ½mk
Reject: unsaturated hydrocarbon
- alkenes or alkynes written in wordsb i Effervescence / fizzing / bubbles ✓½ RCOOH ✓½ or H + present
- award fully if only RCOOH mentioned
- penalise fully (deny ½mk) it only H + mentionedii Potassium manganate (VII) is decolourised ✓
Or
Purple potassium manganate (VII) solutions
turns colourless
Reject: solution is decolourised
or ROHiii Bromine water ✓1 is decolourised or yellow
bromine water turns colourless
Reject: orange / red colour
- colour changes to colourless
- solution is decolourised
Award fully if only one functional group
mentioned
Penalise fully for any contradictory functional
group
- penalise 1/2 mk in the final answer if units are wrong or missing.
- Conditions.
- Complete table - 1 mark
Download Chemistry Paper 3 Questions and Answers with Confidentials - MECS Cluster Joint Mock Exams 2021/2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students