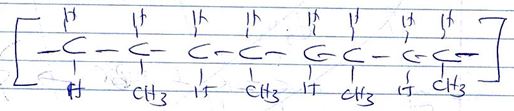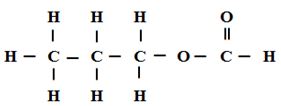Questions
Instructions to the candidates:-
- Answer all the questions.
- Mathematical tables and electronic calculators may be used
- All working MUST be clearly shown where necessary.
-
- State Boyle’s law [1mk]
- At 4000C, 850cm3 of a gas exert a pressure of 560mmHg. What volume of the same gas would exert a pressure of 640mmHg at the same temperature? [3mks]
- When burning magnesium is lowered into a gas jar containing nitrogen (I) oxide, it continues to burn forming a white solid
- Name the white solid 1mk]
- Write a chemical equation of the reaction that occurred [1mk]
- Carbon {IV} oxide is one of the gases used in fire extinguishers
- State any other possible use of carbon {IV} oxide [1mk]
- Name any two reagents that can be reacted together ti generate carbon {IV} oxide [2mks]
- Rusting is a process that causes massive destruction of iron structures
- State one condition that accelerates rusting [1mk]
- State one advantage of rusting [1mk]
- At 60°C, 38 grams of lead{II} nitrate saturate 56cm3 of water. Determine the solubility of lead {II} nitrate at this temperature [2mks]
- Explain why molten sodium chloride conducts electricity, but solid sodium chloride does not [2mks]
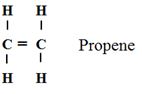
- A polymer can be represented as
- Name and draw the structure of the monomer [2mks]
- What type of polymerization occurs in the above case? [1mk]
- Given that the molecular mass of the polymer is 25620, how many units of the monomer make the polymer [2mks]
- A reaction can be represented as;
C2H4[g] +HBr[g] → C2H5Br[g]
Given the bond energies of C-H, C=C, C-C, C-Br, and H –Br as 20kJ/mol, 580Kj/mole, 446Kj/mole, 438KJ/mole and 396kJ/mole respectively. Determine the heat of formation of C2H5Br [3mks] -
- Define the term, dynamic equilibrium [1mks]
- A reaction at equilibrium can be represented as
2CrO2-4[aq] +2H+[aq] ⇌ Cr2O72-[aq] + H2O{l}
Yellow orange
State and explain the observation made when;- NaOH is added to the equilibrium mixture [2mks]
- HCl is added to the equilibrium mixture [2mks]
- During the electrolysis of dilute copper {II} chloride using carbon electrodes, a current of 1.5A was passed through the solution for 2 hours and 30 minutes
- Write the ionic equation of the reaction that occurred at the cathode [1mk]
- Given R.A.M of copper = 64 and 1F = 96500C, calculate the change in mass of the cathode [3mks]
-
- Define the term half-life [1mk]
- Name two particles likely to be emitted when a radioactive nuclide undergoes radioactivity [2mks]
- The half-life of a radioactive nuclide is 3 hours. Given that its initial mass is 288g, determine the remaining mass after 12 hours. [2mks]
- The reduction potentials of elements M and N are;
M2+[aq] + 2e- ⇌ M[s],Eθ=+0.44V
N+[aq] e- ⇌ N[s], Eθ = -1.64V
Using the above reduction potentials, predict whether a reaction would occur between
N+[aq] and M[s] [3mks] - An hydrocarbon can be represented as: C2 H2
- Name the hydrocarbon [1mk]
- State two reagents that can be reacted together to generate the hydrocarbon [2mks]
- Identify the group of hydrocarbons into which C2H2 belongs to [1mk]
-
- Name two allotropes of sulphur [2mks]
- In an experiment to investigate a certain property of sulphur, Maina added few drops of conc HNO3 to sulphur in a test tube and warmed the mixture
- State one observation made [1mk]
- Write a chemical equation of the reaction that occurred [1mk]
- Chlorine is commonly used in the manufacture of Ca (OCl)2
- State one use of the above compound of chlorine [1mk]
- Write a chemical equation leading to the production of Ca (OCl)2 [1mk]
- A compound can be represented as
- What name is given to the above class of compounds [1mk]
- Name two reagents that can be reacted together to generate the above compound [2mks]
- State two conditions necessary for the reaction leading to formation of the above compound to occur [2mks]
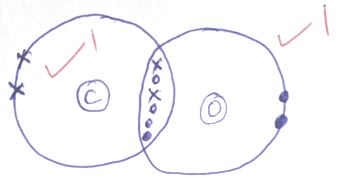
- Using dots and crosses, show bonding in carbon{II} oxide [2mks]
- When 20g of a compound containing carbon, hydrogen and oxygen was burnt in the air, 29.3g of carbon{IV} oxide and 11.7g of water were produced. Determine its empirical formulae.
{C=12, H=1 , O=16} [3mks] - Few drops of hydrochloric acid were added into a test tube containing lead {II} Nitrate solution
- State one observation made [1mk]
- Write an ionic equation of the reaction that occurred in the test tube [1mk]
- In the industrial manufacture of Ammonia one of the raw materials is nitrogen gas
- Name one other raw material [1mk]
- Name two possible sources of the raw material you have named in {a} above [2mks]
- Name two substances that can be used as catalyst in this process [2mks]
- State one use of ammonia [1mk]
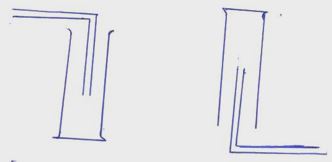
- Gas X and Y can be collected as shown below
- Name the method used to collect gas Y [1mk]
- How do densities of gas X and gas Y compare? [1mk]
- Give an example of a gas that can be collected using the same method as gas Y [1mk]
- Element W has two isotopes W – 36 and W-40 which occur in the ratio x:4. Given that R.A.M of W is 37.25, find the value of x [2mks]
- Describe an experiment that can be used to determine whether a given sample of a liquid is pure [2mks]
- A given mass of gas T diffuses through a porous plug in 48 seconds while a similar mass of gas R diffuse in 70 seconds. Given that the density of gas T is 0.6g/cm3, find the density of gas R [2mks]
- The electron configuration of elements A, B, C, D and E are as given below
Element Electron configuration A
B
C
D
E2, 8, 1
2, 8
2, 7
2, 8, 6
2, 8, 3- Which element has the highest electrical conductivity [1mk]
- Which letter represents the most reactive metal [1mk]
- Which letter represents the most reactive non-metal [1mk]
Marking Scheme
-
- Bolyles law states that volume of a given mass of a gas inversely proportional to its pressure
at constant temperature - P1V1=P2V2
560 x 850 = V2
640
V2=743.75 cm3
- Bolyles law states that volume of a given mass of a gas inversely proportional to its pressure
-
- Magnesium oxide
- Mg[s] + N2O[S] MgO + N2[g]
-
-
- Manufacture of fizzy drink
- Used as a refrigerant
[any one correct]
- Marble chips [solid calcium carbonate and dilute hydrochloric acid (Any correct 2)
-
-
-
- Salty condition
- Acidic condition
-
- Addition of minerals to the soil
- decomposition of iron waste
-
- 38g → 56g of water
? → 100g of water
35x 100
56
= 67.85g/100g of water - Molten sodium chloride has mobile ions while solid sodium chloride does not have mobile ions
-
-
- Addition polymerization
- [48] n = 25620
48 48
N=533.75 units
-
- [580 X1]+420X4 +[396]1 → [446]1 + [420 X5]+[438]1
580+1680 +396 → 446+2100+438
+ 2656 → -2984
+2656 -2984
=-328kJmol-1 -
- Dynamic equilibrium is attained when the rate of the forward reaction is equal to that of the reverse reaction
-
- The intensity of the yellow colour in the equilibrium mixture increased .
Additional of NaOH reduces the concentration ofH+ ions hence equilibrium shifts to the left - the intensity of the yellow colour in the equilibrium mixture decreases.
Additional of HCL increased the concentration of H+ ions hence equilibrium shifts to the right
- The intensity of the yellow colour in the equilibrium mixture increased .
-
- Cu2+[aq] + 2e- → Cu[s]
- Q= It
=1.5x150x60
=13,500C
2moles of electrons are depositing 1 mole of Cu metal
1 mole of electrons=96500C
2 moles of electrons =
193000C deposited 64g of copper
13500
13500 ×64
193000
4.476g
-
- Half life of a radioactive isotope is the time taken for a given or number of nuclides to decay to half its original mass or number
- Alpha particle
Beta particles - the number of half life
12/3 =4
[1/2 ]4x288=18g
- Eθ= ERHS - ELHS
=- 1.64 - + 0.44
=-2.08V
The reaction will not take place because the e.m.f is negative -
- Ethyne
- Calcium carbide and water
- Alkynes
-
- Rhombic sulphur
Monoclinic sulphur -
- Red brown gas of fumes were observed
- S[s] + 6HNO3[aq] H2SO4[aq] +6NO2[g]+2H2O
- Rhombic sulphur
-
- Acts as a bleaching agent
- 2Ca[OH]2[aq] + Cl2[g] → CaCl2[aq] +Ca(OCl)2{aq} +2H[g]
-
- Ester
- propanol and methanoic acid
- Concentrated sulphuric {IV}acid catalyst
Warming
-
- Mass of carbon
=12/44 x 29.3
Mass of H =2/18 x11.7= 7.99
=7.99
Mass of O2
20 – [7.99+1.3] =10.71
Element C H O Mass of the element
R.A.M
Divide by R.A.M7.99
12
7.991.30
1
1.3010.71
16
10.7112 1 16 Divide by smallest value 0.6658
0.66581.3
0.66580.6693
0.6658Mole ratio 1 2 1 E.F CH2O -
- White precipitate was formed
- Pb2[q] + 2Cl [aq] → PbCl2[s]
-
- hydrogen
- Electrolysis of brine
Cracking of larger alkanes - Finely divided iron
Plantinum catalyst - Manufacture of nitrogen fertilizer
Used as a refrigerant
Softening of water
-
- Upward delivery
- gas x is denser than gas y
- Hydrogen, ammonia,methane
22.x+4 x 36x x +40+4=37.25 x [x + 4]
36x + 160 = 37.25x +149
36x – 37,25x=149 – 160
- -1.25x=+11
-1.25 =1.25
X=8.8 - A liquid is boiled when constant boiling point is maintained the liquid is pure
- Time in T = √Density T
Time in R √Density R
48 sec = √0.16
70 sec √Density R
48 sec = √0.16
70 sec2 √Density R2
2304 = 0.16
4900 Density of R
Density of R=0.3402 g/cm3 -
- E
- A
- C
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - Asumbi Girls High School KCSE Mock 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students