Questions
Instructions to candidates
- Answer ALL the questions in the spaces provided, and working MUST be clearly shown
- A magnesium ribbon sample was heated in separate volumes of pure oxygen and air.
- In which sample was the mass of the product higher? Explain. (2 Marks)
- Write the equations for the reactions in the sample with air. (2 Marks)
- Give the systematic name of the following compound and draw the structure of the polymer it forms: CH2CHCl
Name __________________________________________________________________(1 Mark)
Structure (1 Mark) - When aqueous sodium hydroxide solution was added to freshly prepared acidified iron (II) sulphate solution, a green precipitate was formed. When hydrogen peroxide was first added to iron (II) sulphate solution followed by sodium hydroxide solution, a brown precipitate was formed. Explain these observations. (3 Marks)
- Study the following nuclear reaction and complete it by giving the values of m and n
232 92 X → m n Y + 2 0 -1 e- + 4 2 He
m _____________________________ (1 Mark) n _______________________ (1 Mark) -
- State Charles’ Law (1 Mark)
- A certain mass of carbon (IV) oxide gas occupied 200cm3 as 25oC and 750mmHg pressure. Calculate the volume occupied by the same mass of gas if pressure is lowered to 300mmHg and the temperature raised to 30oC. (2 Marks)
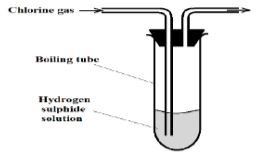
- Chlorine gas was bubbled into as solution of hydrogen sulphide as shown in the diagram below.
- Explain the observation made in the boiling tube (2 Marks)
- What precaution should be taken in this experiment? (1 Mark)
- Distinguish between the bleaching action of chlorine and that of sulphur (IV) oxide. (1 Mark)
- Concentrated sulphuric (VI) acid was left exposed in air for a few days. It was found that the level of the acid had risen.
- Why did the level of the acid in the container rise? (1 Mark)
- How is this property useful in the laboratory? (1 Mark)
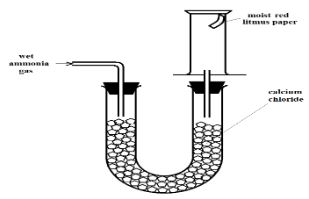
- The setup below can be used to dry and collect ammonia gas. Use it to answer the questions that follow.
- The wet red litmus paper remained red. Explain. (1 Mark)
- Name the method used when collecting ammonia gas. (1 Mark)
- 400cm3 of gas D diffuses from a porous plug in 50 seconds while 600cm3 of oxygen gas diffuses from the same apparatus in 30 seconds. Calculate the relative molecular mass of gas D. (3 Marks)
- Use the information in the table below on solubility to answer the questions that follow.
Salt Solubility at 70°C 35°C CuSO4 38 28 Pb(NO3)2 78 79
A mixture containing 38g copper (II) sulphate and 78g of lead (II) nitrate in 100g of water at 70oC is cooled to 35oC.- Which of the two salts will crystallize? (1 Mark)
- Calculate the mass of crystals formed. (1 Mark)
- State the salt that will be unsaturated at 35oC (1 Mark)
- How much of the salt in c) above would be required to make a saturated solution at 35oC? (1 Mark)
- Methane burns in oxygen as shown by the equation below.
CH4(g) + 2O2(g) → CO2(g) + 2H2O(g)
Given the following bond energies:
Bond Bond/Energy
(kj/mole)C- H 413 O = O 497 C = O 740 O - H 463 - Calculate the heat change for the reaction. (2 Marks)
- Define molar heat of combustion. (1 Mark)
- Given solid sodium carbonate, lead (II) nitrate crystals and water, explain how you can obtain a solid sample of lead (II) carbonate. (3 Marks)
- Calculate the volume of oxygen produced when 10g of silver nitrate was completely decomposed by heating at s.t.p. (Ag = 108, N = 14, O = 16, MGV at s.t.p. = 22.4dm3) (3 Marks)
- A solution of hydrogen chloride gas in water conducts an electrical current, while that of hydrogen chloride in methylbenzene does not conduct. Explain. (2 Marks)
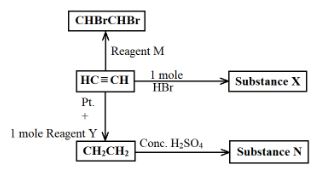
- The scheme below shows some reactions, starting with ethyne. Study it and answer the questions that follow.
- Name substance
- X ______________________________________________________________ (½ Mark)
- N ______________________________________________________________ (½ Mark)
- M ______________________________________________________________ (½ Mark)
- Ethene undergoes polymerization to form a polymer. Give an equation for the reaction and name the product. (1½ Marks)
- Name substance
- When 16g of ammonium nitrate was dissolved in 100cm3 of water at 25oC, the temperature of the solution drops to 19oC.
- Calculate the molar enthalpy of solution of ammonium nitrate (3 Marks)
(N = 14, O = 16, H = 1, Specific Heat Capacity for Water = 4.2kJ/kg/k) - Is the enthalpy change endothermic or exothermic? Give a reason (1 Mark)
- Calculate the molar enthalpy of solution of ammonium nitrate (3 Marks)
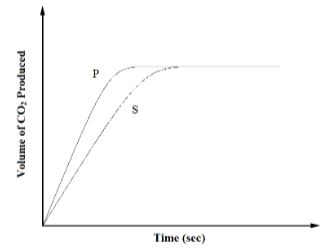
- The curves below represent the volume of carbon (IV) oxide gas evolved when 2M hydrochloric acid was reacted with 100g of powdered calcium carbonate and when 1M hydrochloric acid was reacted with the same quantity of calcium carbonate.
- Which of the two curves represents the reaction of 2M concentrated hydrochloric acid? Explain. (2 Marks)
- Why do the two curves flatten at the same level of production of CO2? (1 Mark)
- The electron arrangement of ions X3+ and Y2- are 2.8, and 2.8.8 respectively.
- In which groups do X and Y belong? (1 Mark)
X___________________________ Y_____________________________________ - State the formula of the compound that would be formed between X and Y (1 Mark)
- In which groups do X and Y belong? (1 Mark)
-
- State two ores from which sodium metal can be extracted. (1 Mark)
- During the extraction, calcium chloride solid is added into the sodium chloride solid. Why is calcium chloride added to the sodium chloride? (1 Mark)
- State two uses of sodium metal. (2 Marks)
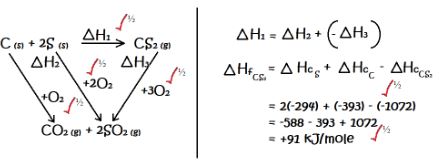
- Using and energy cycle diagram, calculate the enthalpy change of formation of carbon disulphide, given: (3 Marks)
S(s) + O2(g) → SO2(g) ∆H = -294kJ/mole
CS2(g) + 3O2(g) → CO2(g) + 2SO2(g) ∆H = - 1072kj/mole
C(s) + O2(g) → CO2(g) ∆H = -393kJ/mole
- The table below shows tests carried out in a sample of water and the results obtained.
Sample Results observations A Addition of sodium hydroxide dropwise until excess White precipitate which dissolves in excess B Addition of excess ammonia solution White precipitate C Addition of dilute nitric (V) acid followed by barium chloride White precipitate - Identify the anion present in the water sample (1 Mark)
- Write an ionic equation for the reaction in C (1 Mark)
- Use the following information to answer the questions that follow:
Sn2+ (aq) + 2e- → Sn (s) Eθ = -0.14V
Cu2+ (aq) + 2e- → Cu (s) Eθ = +0.34V- Write the cell representation for the cell made up of the two half cells (1 Mark)
- Identify the reducing species (1 Mark)
- Calculate the Eθ value for the cell (1 Mark)
- The following is a reaction of an equilibrium mixture:
2SO2(g) + O2(g) ⇌ 2SO3(g)
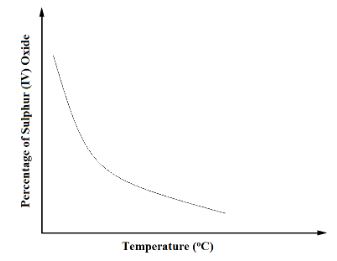
The percentage of sulphur (VI) oxide in the equilibrium mixture varies with temperature as illustrated in the sketch graph below- How does the percentage of sulphur (VI) oxide in the equilibrium mixture vary as the temperature increases? Explain. (1½ Mark)
- Is the forward reaction in the equilibrium exothermic or endothermic? Give a reason for your answer. (1½ Mark)
- Radioactive polonium (Po) with a mass number of 212 and atomic number of 84 was detected in a sample of water. The water had an activity of 1000 counts per second.
- If the water is boiled, explain whether the activity would be affected or not. (1 Mark)
- Given that polonium resulted from bitumen (B) following emission of a beta (β) particle, write a nuclear equation for the decay. (1 Mark)
- State one medical application of radioactivity. (1 Mark)
- Name and give the formula of:
- The chief ore from which zinc is extracted (1 Mark)
- The main impurity in the ore. (1 Mark)
- The ore is concentrated by froth floatation. What is froth floatation? (1 Mark)
- The atomic number of sulphur is 16. Write the electron arrangement of sulphur in the following compounds
- H2S (1 Mark)
- SO32- (1 Mark)
- For the reaction: Cl2(g) + 2I- (aq) → 2Cl- (aq) + I2(s)
Using oxidation numbers, state and explain the reducing species. (2 Marks) - The setup below was used to investigate the effect of carbon (II) oxide on zinc oxide.
- State the observations made on the setup. (2 Marks)
- Write equations for the reactions that took place. (2 Marks)
Marking Scheme
- A magnesium ribbon sample was heated in separate volumes of pure oxygen and air.
- In which sample was the mass of the product higher? Explain. (2 Marks)
- In the sample with air. Magnesium combined with both oxygen and nitrogen components of air, while in the oxygen sample it only combines with oxygen.
- Write the equations for the reactions in the sample with air. (2 Marks)
- 2Mg (s) + O2 (g) 2MgO (s) 3Mg (s) + N2 (g) Mg3N2 (s)
[rules for writing chemical equations apply; ½ mark penalty for no/wrong state symbols; full penalty for wrong symbols/formula and not balanced]
- 2Mg (s) + O2 (g) 2MgO (s) 3Mg (s) + N2 (g) Mg3N2 (s)
- In which sample was the mass of the product higher? Explain. (2 Marks)
- Give the systematic name of the following compound and draw the structure of the polymer it forms: CH2CHCl
- Name chloroethene (1 Mark)
Structure (1 Mark)
- Name chloroethene (1 Mark)
- When aqueous sodium hydroxide solution was added to freshly prepared acidified iron (II) sulphate solution, a green precipitate was formed. When hydrogen peroxide was first added to iron (II) sulphate solution followed by sodium hydroxide solution, a brown precipitate was formed. Explain these observations. (3 Marks)
- Fe(OH)2 formed when NaOH is added to FeSO4 and is a green precipitate.
- Hydrogen peroxide is an oxidising agent and first oxidised iron (II) sulphate to iron (III) sulphate. Addition of NaOH resulted in formation of brown precipitate of Fe(OH)3.
- Study the following nuclear reaction and complete it by giving the values of m and n
X → Y + 2 e- + H
m → 228 (1 Mark) n → 92 (1 Mark) -
- State Charles’ Law (1 Mark)
- The volume of a fixed mass of gas is directly proportional to its absolute temperature at constant pressure.
- A certain mass of carbon (IV) oxide gas occupied 200cm3 as 25oC and 750mmHg pressure. Calculate the volume occupied by the same mass of gas if pressure is lowered to 300mmHg and the temperature raised to 30oC. (2 Marks)
- P₁V₁ = P₂V₂
T₁ T₂
750 x 200 = 300 x V₂
298 303
V₂ = 750 x 200 x 303
298 x 300
V₂ = 508.39cm3
[1mk correct substitution of values into formula; 1mk correct answer]
- P₁V₁ = P₂V₂
- State Charles’ Law (1 Mark)
- Chlorine gas was bubbled into as solution of hydrogen sulphide as shown in the diagram below.
- Explain the observation made in the boiling tube (2 Marks)
- Yellow solid / Yellow precipitate observed. [1 mk – observation]
- Hydrogen sulphide is oxidised to yellow sulphur [1 mk – explanation]
- What precaution should be taken in this experiment? (1 Mark)
- The experiment should be done in a fume chamber // The experiment should be done outside in the open air.
- Distinguish between the bleaching action of chlorine and that of sulphur (IV) oxide. (1 Mark)
- Chlorine bleaches by oxidation while sulphur (IV) oxide bleaches by reduction.
- Explain the observation made in the boiling tube (2 Marks)
- Concentrated sulphuric (VI) acid was left exposed in air for a few days. It was found that the level of the acid had risen.
- Why did the level of the acid in the container rise? (1 Mark)
- It absorbed water from the atmosphere.
- How is this property useful in the laboratory? (1 Mark)
- It is used for drying gases produced in experiments.
- Why did the level of the acid in the container rise? (1 Mark)
- The setup below can be used to dry and collect ammonia gas. Use it to answer the questions that follow.
- The wet red litmus paper remained red. Explain. (1 Mark)
- Ammonia gas reacted with the calcium chloride [to form NH3•CaCl2] hence no gas was collected.
- Name the method used when collecting ammonia gas. (1 Mark)
- Upward delivery of ammonia // Downward displacement of air.
- The wet red litmus paper remained red. Explain. (1 Mark)
- 400cm3 of gas D diffuses from a porous plug in 50 seconds while 600cm3 of oxygen gas diffuses from the same apparatus in 30 seconds. Calculate the relative molecular mass of gas D. (3 Marks)
- RD = 400/50 = 8cm3/sec
RO2 = 600/30 = 20cm3/sec
RD/RO2 = √MO2/MD
8/20 = √32/MD
(0.4)2 x MD =32
MD = 32/0.42 = 32/0.16
MD= 200
- RD = 400/50 = 8cm3/sec
- Use the information in the table below on solubility to answer the questions that follow.
A mixture containing 38g copper (II) sulphate and 78g of lead (II) nitrate in 100g of water at 70oC is cooled to 35oC.- Which of the two salts will crystallize? (1 Mark)
- CuSO4 / Copper (II) sulphate
- Calculate the mass of crystals formed. (1 Mark)
- 38 g – 28 g = 10 g [½mk substitution; ½mk correct answer]
- State the salt that will be unsaturated at 35oC (1 Mark)
- Pb(NO3)2
- How much of the salt in c) above would be required to make a saturated solution at 35oC? (1 Mark)
- 79 g
- Which of the two salts will crystallize? (1 Mark)
- Methane burns in oxygen as shown by the equation below.
CH4 (g) + 2O2 (g) CO2 (g) + 2H2O (g)- Calculate the heat change for the reaction. (2 Marks)
- ∆H = Hreactants + H formation
Hreactants = (413 x 4) + (2 x 497) = +2646
Hproducts = (740 x 2) + (463 x 4) = -3332
∆H = +2646 + (- 3332) = -686kJ/mol
- ∆H = Hreactants + H formation
- Define molar heat of combustion. (1 Mark)
- This is the enthalpy change/heat change that occurs when one mole of a substance completely burns in oxygen.
- Calculate the heat change for the reaction. (2 Marks)
- Given solid sodium carbonate, lead (II) nitrate crystals and water, explain how you can obtain a solid sample of lead (II) carbonate. (3 Marks)
- Add water to solid sodium carbonate to form sodium carbonate solution. Add excess lead (II) nitrate to a volume of the sodium carbonate solution and allow the mixture to settle. Filter the resultant mixture to obtain lead (II) carbonate as the residue. Wash/rinse the residue with distilled water. Dry the residue between filter papers.
[the underlined words form the marking points; each point scores ½ mark]
- Add water to solid sodium carbonate to form sodium carbonate solution. Add excess lead (II) nitrate to a volume of the sodium carbonate solution and allow the mixture to settle. Filter the resultant mixture to obtain lead (II) carbonate as the residue. Wash/rinse the residue with distilled water. Dry the residue between filter papers.
- Calculate the volume of oxygen produced when 10g of silver nitrate was completely decomposed by heating at s.t.p. (Ag = 108, N = 14, O = 16, MGV at s.t.p. = 22.4dm3) (3 Marks)
- 2AgNO3 (s) → 2Ag (s) + 2NO2 (g) + O2 (g)
Moles of AgNO3 = R.F.M. = 108 + 14 + (3 x 16) = 170
moles = 10/170 = 0.0588 moles
moles O2
AgNO3 : O2
2:1
0.0588: 0.0588 x 1
2
=0.02941
Volume of O2
Vol O2 = moles x MGV
= 0.02941 x 22.4
= 0.6588L
OR 0.6588dm3
or 658.82cm3
- 2AgNO3 (s) → 2Ag (s) + 2NO2 (g) + O2 (g)
- A solution of hydrogen chloride gas in water conducts an electrical current, while that of hydrogen chloride in methylbenzene does not conduct. Explain. (2 Marks)
- In water HCl dissociates to yield mobile ions but in methylbenzene, HCl does not dissociate and remains in molecular state.
[marking points are underlined; 1mk for each point]
- In water HCl dissociates to yield mobile ions but in methylbenzene, HCl does not dissociate and remains in molecular state.
- The scheme below shows some reactions, starting with ethyne. Study it and answer the questions that follow.
- Name substance
- X - bromoethane (½ Mark)
- N - ethyl hydrogen sulphate (½ Mark)
- M - bromine (½ Mark)
- Ethene undergoes polymerization to form a polymer. Give an equation for the reaction and name the product. (1½ Marks)
-
polyethene
[1mk for equation and ½mk for name]
-
- Name substance
- When 16g of ammonium nitrate was dissolved in 100cm3 of water at 25oC, the temperature of the solution drops to 19oC.
- Calculate the molar enthalpy of solution of ammonium nitrate (3 Marks)
(N = 14, O = 16, H = 1, Specific Heat Capacity for Water = 4.2kJ/kg/k)
- ΔH= mCΔT
= 0.1kg x 4.2KJ/Kg/K x 6k
<+ 2.52KJ
FRM NH4NO3 = 80
since 80g < 1 mole
if 16g evo0loved + 2.52KJ
80g evolve 80 x 2.52
16
= +12.6KJ
- ΔH= mCΔT
- Is the enthalpy change endothermic or exothermic? Give a reason (1 Mark)
- Endothermic. The net ∆H is positive.
[½ mk for statement; ½ mk for explanation]
- Endothermic. The net ∆H is positive.
- Calculate the molar enthalpy of solution of ammonium nitrate (3 Marks)
- The curves below represent the volume of carbon (IV) oxide gas evolved when 2M hydrochloric acid was reacted with 100g of powdered calcium carbonate and when 1M hydrochloric acid was reacted with the same quantity of calcium carbonate.
- Which of the two curves represents the reaction of 2M concentrated hydrochloric acid? Explain. (2 Marks)
- P. It is steeper than curve S // The reaction stops earlier than S // The curve flattens out earlier than in S.
[1mk for stating P; 1mk for explanation]
- P. It is steeper than curve S // The reaction stops earlier than S // The curve flattens out earlier than in S.
- Why do the two curves flatten at the same level of production of CO2? (1 Mark)
- The same mass of powdered calcium carbonate reacted completely with the acid.
- Which of the two curves represents the reaction of 2M concentrated hydrochloric acid? Explain. (2 Marks)
- The electron arrangement of ions X3+ and Y2- are 2.8, and 2.8.8 respectively.
- In which groups do X and Y belong? (1 Mark)
- X Group III Y Group VI
- State the formula of the compound that would be formed between X and Y (1 Mark)
- X2Y3 OR Al2S3
- In which groups do X and Y belong? (1 Mark)
-
- State two ores from which sodium metal can be extracted. (1 Mark)
- Rock salt, Chile saltpetre // Solid sodium nitrate, Glaubers salt
[first two ores mentioned correctly by the candidate]
- Rock salt, Chile saltpetre // Solid sodium nitrate, Glaubers salt
- During the extraction, calcium chloride solid is added into the sodium chloride solid. Why is calcium chloride added to the sodium chloride? (1 Mark)
- To lower the melting point of NaCl from 800oC to about 600oC
[temperature value MUST be indicated by the candidate]
- To lower the melting point of NaCl from 800oC to about 600oC
- State two uses of sodium metal. (2 Marks)
- Manufacture of sodium cyanide for gold extraction
- Sodium chloride is used as a food additive
- Sodium vapour is used in streetlights/streetlamps which gives yellow glow
[two uses for 1 mark each]
- State two ores from which sodium metal can be extracted. (1 Mark)
- Using and energy cycle diagram, calculate the enthalpy change of formation of carbon disulphide, given: (3 Marks)
- S (s) + O2 (g) SO2 (g) ∆H = -294kJ/mole
- CS2 (g) + 3O2 (g) CO2 (g) + 2SO2 (g) ∆H = - 1072kj/mole
- C (s) + O2 (g) CO2 (g) ∆H = -393kJ/mole
- The table below shows tests carried out in a sample of water and the results obtained.
- Identify the anion present in the water sample (1 Mark)
- SO42- // Sulphate ion
- Write an ionic equation for the reaction in C (1 Mark)
- Ba2+(aq) + SO42-(aq) → BaSO4 (s)
[½mk penalty for wrong state symbols or omission of state symbols; full mark penalty for wrong balancing or wrong formulae]
- Ba2+(aq) + SO42-(aq) → BaSO4 (s)
- Identify the anion present in the water sample (1 Mark)
- Use the following information to answer the questions that follow:
Sn2+ (aq) + 2e- Sn (s) Eθ = -0.14V
Cu2+ (aq) + 2e- Cu (s) Eθ = +0.34V- Write the cell representation for the cell made up of the two half cells (1 Mark)
- Sn (s) |Sn2+(aq) ||Cu2+(aq) |Cu (s) Eθ = +0.48V
[the Eθ value must be included for the candidate to score the 1 mark)
- Sn (s) |Sn2+(aq) ||Cu2+(aq) |Cu (s) Eθ = +0.48V
- Identify the reducing species (1 Mark)
- Sn (s)
[must be written in symbol and state symbol included in order to score 1mk]
- Sn (s)
- Calculate the Eθ value for the cell (1 Mark)
- Eθ = Ered – Eox
= +0.34 – (-0.14)
= + 0.48V
- Eθ = Ered – Eox
- Write the cell representation for the cell made up of the two half cells (1 Mark)
- The following is a reaction of an equilibrium mixture:
The percentage of sulphur (VI) oxide in the equilibrium mixture varies with temperature as illustrated in the sketch graph below- How does the percentage of sulphur (VI) oxide in the equilibrium mixture vary as the temperature increases? Explain. (1½ Mark)
- Percentage reduces. An increase in temperature favours the backward reaction // An increase in temperature causes equilibrium to shift to the left.
[½ mk for statement and ½ mk for explanation]
- Percentage reduces. An increase in temperature favours the backward reaction // An increase in temperature causes equilibrium to shift to the left.
- Is the forward reaction in the equilibrium exothermic or endothermic? Give a reason for your answer. (1½ Mark)
- Exothermic. The percentage of SO3 increases with the reduction in temperature.
[½mk for statement; 1mk for explanation]
- Exothermic. The percentage of SO3 increases with the reduction in temperature.
- How does the percentage of sulphur (VI) oxide in the equilibrium mixture vary as the temperature increases? Explain. (1½ Mark)
- Radioactive polonium (Po) with a mass number of 212 and atomic number of 84 was detected in a sample of water. The water had an activity of 1000 counts per second.
- If the water is boiled, explain whether the activity would be affected or not. (1 Mark)
- Not affected. Radioactive decay is not affected by physical factors like temperature.
[½mk for statement; ½mk for explanation]
- Not affected. Radioactive decay is not affected by physical factors like temperature.
- Given that polonium resulted from bitumen (B) following emission of a beta (β) particle, write a nuclear equation for the decay. (1 Mark)
- B → Po + e-
- State one medical application of radioactivity. (1 Mark)
- Sterilization of surgical instruments by radiating them
- Regulating iodine level, hence treating thyroid gland deformities
- If the water is boiled, explain whether the activity would be affected or not. (1 Mark)
- Name and give the formula of:
- The chief ore from which zinc is extracted (1 Mark)
- Zinc blende – ZnS
- The main impurity in the ore. (1 Mark)
- Lead (II) sulphide
- The ore is concentrated by froth floatation. What is froth floatation? (1 Mark)
- The process of adding frothing agent to finely ground ore and agitating it to separate the ore from impurities, hence concentrate it.
- The chief ore from which zinc is extracted (1 Mark)
- The atomic number of sulphur is 16. Write the electron arrangement of sulphur in the following compounds
- H2S S2- = 2.8.8 (1 Mark)
- SO32- S4+ = 2.8.2 (1 Mark)
- For the reaction: Cl2(g) + 2I- (aq) → 2Cl-(aq) + I2 (s)
Using oxidation numbers, state and explain the reducing species. (2 Marks)- Iodide ions // I-(aq)
- Oxidation number of reducing species increases. Oxidation number of iodide ions increases from -1 to zero.
[1mk statement; 1mk explanation]
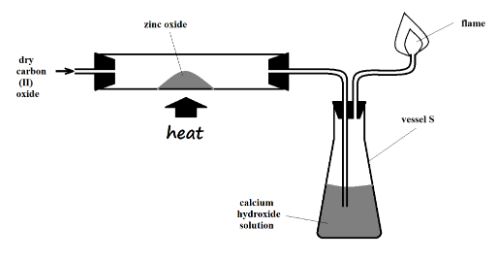
- The setup below was used to investigate the effect of carbon (II) oxide on zinc oxide.
- State the observations made on the setup. (2 Marks)
- Yellow zinc oxide changes to a grey substance
- A white precipitate forms in the calcium hydroxide solution // Calcium hydroxide solution turns from colourless to white
- Reject: Lime water//calcium hydroxide solution turns milky.
- Write equations for the reactions that took place. (2 Marks)
- ZnO (s) + CO (g) Zn (s) + CO2 (g)
- CO2 (g) + Ca(OH)2 (aq) ---- CaCO3 (s) + H2O (l)
- State the observations made on the setup. (2 Marks)
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - KCSE 2022 Mock Exams Set 2.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students