Questions
Instructions to candidates
- Answer ALL the questions in the spaces provided, and working MUST be clearly shown
-
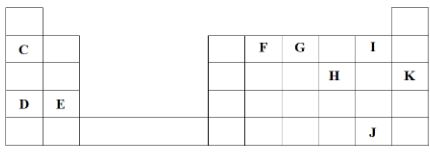
- The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters do not represent actual symbols of the elements
- Identify the most reactive non-metal. Explain (2 Marks)
- What is the name given to the family of elements to which I and J belong? (1 Mark)
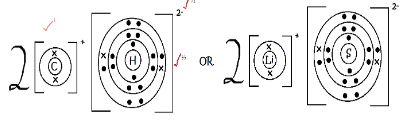
- Using dots (•) and crosses (×) to represent electrons, show bonding in the compound formed between C and H. (2 Marks)
- How does the atomic radius of F compare with that of I? Explain. (2 Marks)
- Study the table below and answer the questions that follow.
Substance M N O P Q R Melting Point (°C) 801 1356 -101 26 -39 113 Boiling Point (°C) 1410 2850 -36 154 457 445 Electrical conductivity in solid state Poor Poor Poor Poor Good Poor Electrical conductivity in molten state Good Poor Poor Poor Good Poor - Explain why substance M is a good conductor of electricity in the molten state but not in the solid state. (2 Marks)
- What is the most likely structure and bond in substance N? Explain. (2 Marks)
Structure ____________________________ Bond ___________________________ - Identify, with a reason, a substance that exists as a liquid at room temperature. (2 Marks)
- The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters do not represent actual symbols of the elements
-
-
- What name is given to different forms of an element which exist in the same physical state? (1 Mark)
- Name two crystalline forms of carbon (1 Mark)
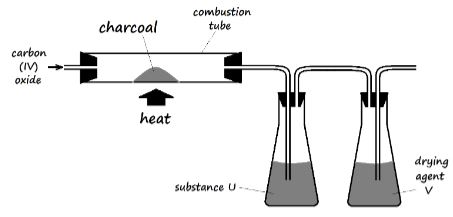
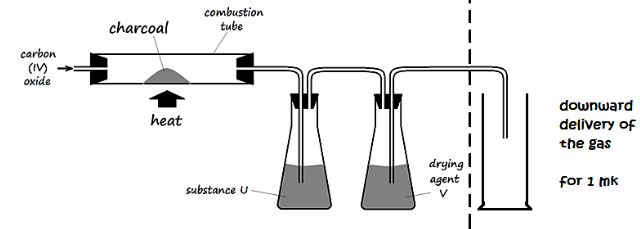
- The figure below is part of a setup used to prepare and collect dry carbon (II) oxide from carbon (IV) oxide.
- Complete the diagram to show how dry carbon (II) oxide gas is collected. (1 Mark)
- Identify:
- Substance U and state its use
- Drying agent Y
- Write a chemical equation for the reaction which takes place in the combustion tube (1 Mark)
- Carbon (II) oxide is a major environmental pollutant.
- Give one major source of carbon (II) oxide in the atmosphere (1 Mark)
- Explain how carbon (II) oxide causes poisoning (1 Mark)
- State one use of carbon (II) oxide (1 Mark)
- Write an equation for the formation of water gas. (1 Mark)
- Explain why sodium hydroxide solution is not used in testing for carbon (IV) oxide gas, while calcium hydroxide is preferably used. (2 Marks)
-
-
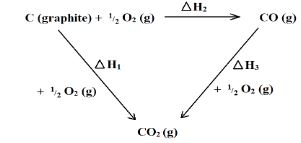
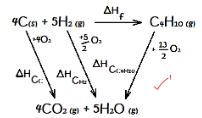
- Study the following energy cycle diagram and then answer the questions that follow.
- Name the enthalpy change represented by ∆H2. (1 Mark)
- Use the following information to calculate the value of ∆H1 for 144g of graphite. (2 Marks)
∆H2 = -110 kJ/mole ∆H3 = -283kJ/mole
- The following are thermochemical equations for molar enthalpies of combustion for some substances. Study them and answer the questions that follow.
C4H10 (g) + 13/2O2 (g) ? 4CO2 (g) + 5H2O ∆Hθc = -2877kJ/mole
C (s) + O2 (g) ? CO2 (g) ∆Hθc = -399kJ/mole
H2 (g) + ½O2 (g) ? H2O (l) ∆Hθc = -286kJ/mole- What is molar enthalpy of combustion of a substance? (1 Mark)
- Calculate the molar enthalpy of formation of butane (C4H10) using the information given above. (3 Marks)
- The following results were obtained in an experiment, to determine the heat of neutralization of 25cm3 of 2M sodium hydroxide solution, using 25cm3 of hydrochloric acid:
Initial temperature of acid = 25.0°C
Initial temperature of alkali = 26.0°C
Final temperature of mixture of acid + alkali = 38.5°C
Density of solution =1g/cm3
Specific heat capacity of solution =4.2 J/g/- Define molar heat of neutralization (1 Mark)
- Write an ionic equation for the neutralization reaction involving hydrochloric acid and sodium hydroxide solution. (1 Mark)
- Calculate:
- The enthalpy change during this experiment. (2 Marks)
- The molar enthalpy of neutralization for this reaction (2 Marks)
- Study the following energy cycle diagram and then answer the questions that follow.
-
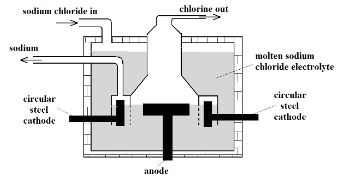
- Below is a simplified diagram of the Down’s Cell, used for the manufacture of sodium. Study it and answer the questions that follow.
- What material is the anode made of? Give the reason why that material is used. (2 Marks)
- What precaution is taken to prevent chlorine and sodium from re-combining? (1 Mark)
- Write an ionic equation for the reaction in which chlorine gas is formed (1 Mark)
- In the Downs process, (used for manufacture of sodium), a certain salt is added to lower the melting point of sodium chloride from about 800oC to about 600°C.
- Name the salt that is added. (1 Mark)
- State why it is necessary to lower the temperature in b) above (1 Mark)
- Explain why aqueous sodium chloride is not suitable as an electrolyte for the manufacture of sodium in the Down’s Process. (2 Marks)
- Sodium metal reacts with air to form two oxides. Give the formulae of the two oxides (1 Mark)
- State two uses of sodium (2 Marks)
- Below is a simplified diagram of the Down’s Cell, used for the manufacture of sodium. Study it and answer the questions that follow.
-
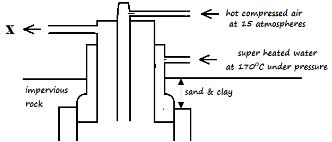
- The diagram below shows part of the Frasch process, used for the extraction of sulphur. Use it to answer the questions that follow.
- Identify X ________________________________________________________(1 Mark)
- Why is it necessary to use superheated water and hot compressed air in this process? (2 Marks)
- State two physical properties of sulphur that makes it possible for it to be extracted by this method. (2 Marks)
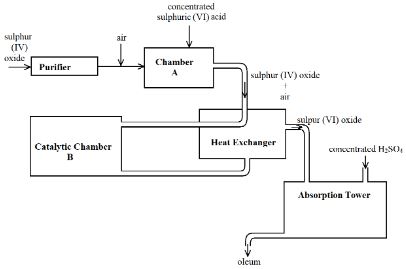
- The diagram below shows part of the process in the manufacture of sulphuric (VI) acid. Study it and use it to answer the questions that follow.
- Give two reasons why air is referred to as a mixture (2 Marks)
- What is the role of concentrated sulphuric (VI) acid in Chamber A? (1 Mark)
- Name two catalysts that can be used in the Catalytic Chamber B. (2 Marks)
- State two roles of the heat exchanger (2 Marks)
- Describe the test for sulphite anion, SO32- (2 Marks)
- Explain the observation made when a few drops of concentrated sulphuric (VI) acid are added to crystals of hydrated copper (II) sulphate. Explain your answer. (2 Marks)
- The diagram below shows part of the Frasch process, used for the extraction of sulphur. Use it to answer the questions that follow.
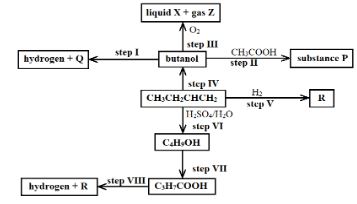
- Study the reaction scheme below and answer the questions the follow:
- What is the distinguishing physical property of Substance P? (1 Mark)
- Identify a suitable reagent that can be used in Step I. (1 Mark)
- Describe a chemical test on how C3H7COOH can be distinguished from C4H9OH. (2 Marks)
- Write an equation for the reaction that takes place in Step III (1 Mark)
- Name the types of reaction that occur in steps II, III, V, and VII (2 Marks)
- If 7.4g of butanol completely underwent Step III, determine the volume of gas Z produced at s.t.p. (MGV = 22.4 litres, C = 12, H = 1, O = 16) (3 Marks)
- Write an equation for the reaction between R and one mole of fluorine gas (1 Mark)
- Describe a chemical test for liquid X (2 Marks)
Marking Scheme
-
-
- Identify the most reactive non-metal. Explain (2 Marks)
- Element I. It has the smallest atom with the highest effective nuclear force of attraction.
[1 mk statement; 1 mk explanation]
- Element I. It has the smallest atom with the highest effective nuclear force of attraction.
- What is the name given to the family of elements to which I and J belong? (1 Mark)
- Halogens Reject: Group VII
- Using dots (•) and crosses (×) to represent electrons, show bonding in the compound formed between C and H. (2 Marks)
-
- How does the atomic radius of F compare with that of I? Explain. (2 Marks)
- F is larger than I. F experiences a lower effective nuclear force of attraction on its energy levels hence more loosely held // F has fewer protons for the same number of energy levels as I hence its energy levels are more loosely held.
- Identify the most reactive non-metal. Explain (2 Marks)
- Study the table below and answer the questions that follow
Substance M N O P Q R Melting Point (°C) 801 1356 -101 26 -39 113 Boiling Point (°C) 1410 2850 -36 154 457 445 Electrical conductivity in solid state Poor Poor Poor Poor Good Poor Electrical conductivity in molten state Good Poor Poor Poor Good Poor - Explain why substance M is a good conductor of electricity in the molten state but not in the solid state. (2 Marks)
- It has a giant ionic structure. Its ions are held by strong ionic bonds in the solid state but these are weakened in the molten state to allow their movement.
- What is the most likely structure and bond in substance N? Explain. (2 Marks)
- Structure Giant covalent//Giant atomic Bond Covalent
- It has high melting point and boiling point and does not conduct electricity in the solid and in the molten state
[½mk each for structure and bond; 1mk for explanation]
- Identify, with a reason, a substance that exists as a liquid at room temperature. (2 Marks)
- Q. Its melting point is below room temperature and its boiling point above room temperature.
- Explain why substance M is a good conductor of electricity in the molten state but not in the solid state. (2 Marks)
-
- What name is given to different forms of an element which exist in the same physical state? (1 Mark)
- Allotropes
- Name two crystalline forms of carbon (1 Mark)
- Diamond Graphite
- What name is given to different forms of an element which exist in the same physical state? (1 Mark)
- The figure below is part of a setup used to prepare and collect dry carbon (II) oxide from carbon (IV) oxide.
- Complete the diagram to show how dry carbon (II) oxide gas is collected. (1 Mark)
- Identify:
- Substance U and state its use
- Concentrated sodium hydroxide solution // Concentrated potassium hydroxide solution
- Drying agent Y
- Concentrated sulphuric (VI) acid
- Substance U and state its use
- Write a chemical equation for the reaction which takes place in the combustion tube (1 Mark)
- CO2 (g) + C (s) 2CO (g)
- Carbon (II) oxide is a major environmental pollutant.
- Give one major source of carbon (II) oxide in the atmosphere (1 Mark)
- Automobile exhaust fumes // Industrial emissions
- Explain how carbon (II) oxide causes poisoning (1 Mark)
- It forms stable carboxyhaemoglobin that prevents oxygen from reaching body cells to result in suffocation
- Give one major source of carbon (II) oxide in the atmosphere (1 Mark)
- State one use of carbon (II) oxide (1 Mark)
- Used as a reducing agent in the extraction of metals from their ores
- Used in the industrial production of methanol
- A mixture of carbon (II) oxide and hydrogen is used as a fuel (water gas)
[the candidate’s first answer for 1 mk]
- Write an equation for the formation of water gas. (1 Mark)
- C (s) + H2O (g) CO (g) + H2 (g)
[rules for writing chemical equations apply]
- C (s) + H2O (g) CO (g) + H2 (g)
- Explain why sodium hydroxide solution is not used in testing for carbon (IV) oxide gas, while calcium hydroxide is preferably used. (2 Marks)
- Sodium hydroxide reacts with carbon (IV) oxide to form soluble sodium carbonate hence no change in physical appearance; while calcium hydroxide reacts with carbon (IV) oxide to form a white precipitate which can be used to detect the reaction.
- Complete the diagram to show how dry carbon (II) oxide gas is collected. (1 Mark)
-
-
- Study the following energy cycle diagram and then answer the questions that follow.
- Name the enthalpy change represented by ∆H2. (1 Mark)
- Heat of formation of carbon (II) oxide
- Use the following information to calculate the value of ∆H1 for 144g of graphite. (2 Marks)
- ∆H2 = -110 kJ/mole ∆H3 = -283kJ/mole
ΔH2 = ΔH1 - ΔH3
- ∆H2 = -110 kJ/mole ∆H3 = -283kJ/mole
- Name the enthalpy change represented by ∆H2. (1 Mark)
- The following are thermochemical equations for molar enthalpies of combustion for some substances. Study them and answer the questions that follow.
C4H10 (g) + 13/2O2 (g) ? 4CO2 (g) + 5H2O ∆Hθc = -2877kJ/mole
C (s) + O2 (g) ? CO2 (g) ∆Hθc = -399kJ/mole
H2 (g) + ½O2 (g) ? H2O (l) ∆Hθc = -286kJ/mole- What is molar enthalpy of combustion of a substance? (1 Mark)
- This is the enthalpy change that occurs when one mole of a substance is completely burnt in oxygen.
- Calculate the molar enthalpy of formation of butane (C4H10) using the information given above. (3 Marks)
-
from cycle diagram
ΔHf = 4ΔHcc + 5ΔHCH2 - ΔHCC4H10
= 4(-399) + 5(-286) - (-2877)
= -149KJ/mole
-
- What is molar enthalpy of combustion of a substance? (1 Mark)
- The following results were obtained in an experiment, to determine the heat of neutralization of 25cm3 of 2M sodium hydroxide solution, using 25cm3 of hydrochloric acid:
Initial temperature of acid = 25.0°C
Initial temperature of alkali = 26.0°C
Final temperature of mixture of acid + alkali = 38.5°C
Density of solution =1g/cm3
Specific heat capacity of solution =4.2 J/g/- Define molar heat of neutralization (1 Mark)
- This is the enthalpy change that occurs when an acid and a base react to form one mole of water.
- Write an ionic equation for the neutralization reaction involving hydrochloric acid and sodium hydroxide solution. (1 Mark)
- H+(aq) + OH-(aq) → H2O (l)
- Calculate:
The enthalpy change during this experiment. (2 Marks)- ∆T = 38.5 – () = 13 mass = volume = 25 + 25 = 50g
∆H = mc∆T = 50g x 4.2 J/g/K x 13K = -2730J - The molar enthalpy of neutralization for this reaction (2 Marks)
- Moles H2O
Moles H2O = moles NaOH
If 1000cm3 base contains 2 moles
Then 25cm3 base contains = 0.05 moles
Molar heat of neutral
Since 0.05 moles of base evolves -2730 J
1 mole of base evolves = = - 54600J/mole = 54.6kJ/mole
- ∆T = 38.5 – () = 13 mass = volume = 25 + 25 = 50g
- Define molar heat of neutralization (1 Mark)
- Study the following energy cycle diagram and then answer the questions that follow.
-
- Below is a simplified diagram of the Down’s Cell, used for the manufacture of sodium. Study it and answer the questions that follow.
- What material is the anode made of? Give the reason why that material is used. (2 Marks)
- Carbon. It is inert and will therefore not react with the chlorine.
[1mk statement; 1mk explanation]
- Carbon. It is inert and will therefore not react with the chlorine.
- What precaution is taken to prevent chlorine and sodium from re-combining? (1 Mark)
- The anode is surrounded by wire meshing to prevent chlorine from coming in contact with the hot sodium metal.
- Write an ionic equation for the reaction in which chlorine gas is formed (1 Mark
- 2Cl (l) → Cl2 (g) + 2e-
- What material is the anode made of? Give the reason why that material is used. (2 Marks)
- In the Downs process, (used for manufacture of sodium), a certain salt is added to lower the melting point of sodium chloride from about 800oC to about 600oC.
- Name the salt that is added. (1 Mark)
- Magnesium chloride
- State why it is necessary to lower the temperature in b) above (1 Mark)
- To lower the amount of energy required to maintain the electrolyte in the molten state [hence lower production cost].
- Name the salt that is added. (1 Mark)
- Explain why aqueous sodium chloride is not suitable as an electrolyte for the manufacture of sodium in the Down’s Process. (2 Marks)
- H+ is preferentially discharged, and not Na+ at the anode since Na+ (aq) requires a high potential.
The Na formed will also react explosively with water.
- H+ is preferentially discharged, and not Na+ at the anode since Na+ (aq) requires a high potential.
- Sodium metal reacts with air to form two oxides. Give the formulae of the two oxides (1 Mark)
- Na2O Na2O2 Reject: Na3N Reject names written in words.
- State two uses of sodium (2 Marks)
- The yellow glow of sodium is used in making fog-lights and street lamps
- Sodium chloride is used as a food additive
- It is used as a reducing agent in the extraction of titanium from its ore
[1st two of the candidate’s answers]
- Below is a simplified diagram of the Down’s Cell, used for the manufacture of sodium. Study it and answer the questions that follow.
-
- The diagram below shows part of the Frasch process, used for the extraction of sulphur. Use it to answer the questions that follow.
- Identify X
- molten sulphur and water (1 Mark)
- Why is it necessary to use superheated water and hot compressed air in this process? (2 Marks)
- Superheated water melts the sulphur deposits while hot compressed air increases pressure in the sulphur deposit to push the molten sulphur up the middle concentric pipe.
- State two physical properties of sulphur that makes it possible for it to be extracted by this method. (2 Marks)
- It does not does not dissolve in water
- It has a low melting point // It melts easily
- Identify X
- The diagram below shows part of the process in the manufacture of sulphuric (VI) acid. Study it and use it to answer the questions that follow.
- Give two reasons why air is referred to as a mixture (2 Marks)
- Its components can be separated by physical means
- It does not have a specific melting and boiling point.
- What is the role of concentrated sulphuric (VI) acid in Chamber A? (1 Mark)
- It dries the gas produced // It acts as a drying agent.
- Name two catalysts that can be used in the Catalytic Chamber B. (2 Marks)
- Vanadium (V) oxide
- Platinum
- State two roles of the heat exchanger (2 Marks)
- It preheats the mixture of sulphur (IV) oxide and air before being taken to the catalytic chamber
- It cools down the sulphur (VI) oxide prepared in the catalytic chamber
- Describe the test for sulphite anion, SO32- (2 Marks)
- Add a few drops of barium nitrate solution to a small of the test substance. Add 1cm3 of hydrochloric acid to the resultant mixture. If a white precipitate that dissolves on addition of hydrochloric acid is observed, it indicates that sulphite ions are present.
[marking points are underlined, each scores ½ mark]
- Add a few drops of barium nitrate solution to a small of the test substance. Add 1cm3 of hydrochloric acid to the resultant mixture. If a white precipitate that dissolves on addition of hydrochloric acid is observed, it indicates that sulphite ions are present.
- Explain the observation made when a few drops of concentrated sulphuric (VI) acid are added to crystals of hydrated copper (II) sulphate. Explain your answer. (2 Marks)
- The blue crystals turn to white. Blue hydrated copper (II) sulphate loses its water of crystallization and changes to white powder.
- Give two reasons why air is referred to as a mixture (2 Marks)
- The diagram below shows part of the Frasch process, used for the extraction of sulphur. Use it to answer the questions that follow.
- Study the reaction scheme below and answer the questions the follow:
- What is the distinguishing physical property of Substance P? (1 Mark)
- It has a pleasant smell Reject: It has a sweet smell
- Identify a suitable reagent that can be used in Step I. (1 Mark)
- Sodium metal // Potassium metal
- Describe a chemical test on how C3H7COOH can be distinguished from C4H9OH. (2 Marks)
- Put samples of the C3H7COOH and C4H9OH in separate test tubes. Add a spatula of sodium carbonate in both test tubes. Effervescence of a colourless gas is observed in the test tube containing C3H7COOH while no effervescence is observed in the test tube containing C4H9OH.
- Write an equation for the reaction that takes place in Step III (1 Mark)
- C4H7OH (l) + 6O2 (g) → 4CO2 (g) + 4H2O (l)
[rules for writing chemical equations apply; state symbols may only be ignored if they have not been put completely; if one is stated the all MUST be included]
- C4H7OH (l) + 6O2 (g) → 4CO2 (g) + 4H2O (l)
- Name the types of reaction that occur in steps II, III, V, and VII (2 Marks)
- II addition reaction III combustion reaction
- V addition reaction VII oxidation
[hydrogenation, esterification etc are considered as processes and NOT types of reaction hence cannot answer the question of ‘type of reaction’]
- If 7.4g of butanol completely underwent Step III, determine the volume of gas Z produced at s.t.p. (MGV = 22.4 litres, C = 12, H = 1, O = 16) (3 Marks)
- moles butanol
R.M.M= 4(12) + 7(1) + 16 + 1 = 72
if 72g butanol is 2 mole
then 7.4g will be
7.4 x 2 = 0.1028 moles (4sf)
72
moles CO2
butanol: CO2
1:4
0.1028 : 0.1028 x 4
1
= 0.4112 moles
volumes CO2
1 moles = 22.4l
1 mole = 22.4l
0.4112 mole
0.4112 x 22.4
1
=9.211L
- moles butanol
- Write an equation for the reaction between R and one mole of fluorine gas (1 Mark)
- C4H10 (g) + F2 (g) → C4H9F (g) + HF (g)
- Describe a chemical test for liquid X (2 Marks)
- Add a few drops of liquid X to white anhydrous copper (II) sulphate powder. The white powder turns to blue crystals
OR
Add a few drops of liquid X to blue anhydrous cobalt (II) chloride powder. The blue powder turns to pink crystals.
- Add a few drops of liquid X to white anhydrous copper (II) sulphate powder. The white powder turns to blue crystals
- What is the distinguishing physical property of Substance P? (1 Mark)
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - KCSE 2022 Mock Exams Set 2.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students