QUESTIONS
SECTION A (25 marks)
Answer all the questions in this section in the spaces provided.
- A micrometer screw gauge with a zero error –0.02mm was used to measure the diameter of a marble whose actual radius is 2.17mm. Determine the reading expected and hence draw the scale seen on the gauge (Pitch = 0.5mm). (2 marks)
- Explain the variation of density of water when its temperature falls from 10ºC to freezing point. (2 marks)
- The figure below shows two identical thermometers A and B placed near and at equal distances on either sides of a hot metal plate painted black on the side A and silvery on side B.
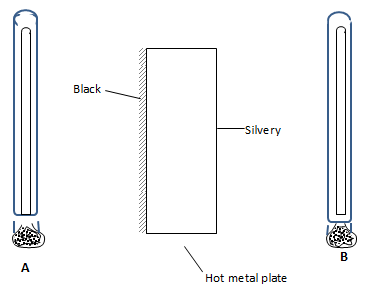
State and explain the observation made on the reading of the thermometers after some minutes. (2 marks) - A body is acted upon by a force of 10N towards the right hand side and 6N towards the left hand side. Determine the resultant force. (2marks)
- The figure below a uniform wooden plank of length 4m and weight 10N. The plank is held at equilibrium by a weight of 40N placed at one end as shown.
Determine the distance, d. (3marks)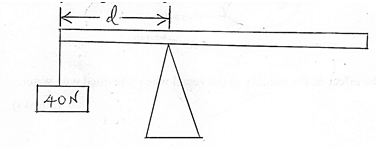
- The figure shows a non-viscous fluid that is not compressible moving through a pipe of varied cross-sectional area.
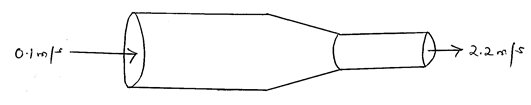
If the area of the narrower region is 0.05m2, determine the diameter of the wider region. (3marks) - State the S.I unit of luminous intensity. (1mark)
- In terms of intermolecular forces, explain the difference between liquid and gaseous state. (2marks)
- The figure shows a vessel resting on a horizontal bench.

State and explain the effect on the stability of the vessel when it is filled with water. (2marks) - The figure below shows a planet Venus orbiting the sun in a circular orbit at constant speed.
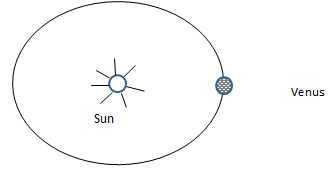
- State what provides the centripetal force. (1 mark)
- Indicate the direction of centripetal force on the planet. (1 mark)
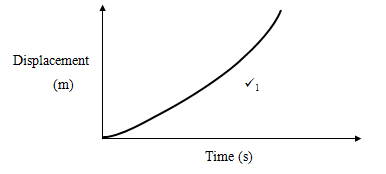
- On the axes below, sketch displacement time graph for an accelerating body. (1mark)
- The figure shows a capillary tube dipped in water.
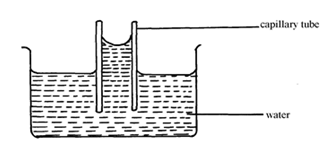
State two differences that will be observed when water is replaced with mercury in the set up above. (2 marks) - Water is known to boil at 100ºC. A student heated some water and noticed that it boiled at 101ºC. State two possible reasons for this observation. (2 marks)
SECTION B (55 marks)
Answer all the questions in this section in the spaces provided.
-
- State Archimedes’ principle. (1mark)
- The figure shows a hydrometer.
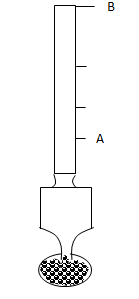
- State the reason why lead shots are fixed at the bottom of hydrometer. (1 mark)
- State the letter in the figure which represents a greater density. (1 mark)
- An ordinary hydrometer of mass 30g floats with 4cm of its stem out of the water. The area of the cross – section of the stem is 0.90 cm2. Taking density of water= 1.0g/ cm3.
Determine the:- mass of the water displaced. (1mark)
- Volume of water displaced. (1mark)
- Volume of stem above water. (2marks)
- Total volume of the hydrometer. (2marks)
- Length of stem above the surface when it floats in a liquid of relative density 1.5. (2marks)
- A balloon is filled with a gas which is lighter than air. It is observed to rise in air up to a certain height. State a reason why the balloon stops rising. (1 mark)
- The figure shows a bucket of water.
Explain why manufacturers prefer the shape shown above as a container for holding liquids such as water. (2marks)
-
- The figure shows a block hanging at rest from a ceiling by a piece of rope.
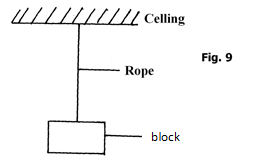
Use the figure to answer question 16 (a) and (b)- Both gravitational force and tensional force act on the system. State a reason for the presence of each force:
- Gravitational force. ( 1mark)
- Tensional force (1mark)
- Air resistance and normal reaction are absent (do not act on the system). State a reason for each.
- Air resistance. (1 mark)
- Normal reaction (1mark)
- Both gravitational force and tensional force act on the system. State a reason for the presence of each force:
- The figure shows a brick resting on an incline plane at an angle θ to the horizontal. The weight W and the frictional force, Fr are shown.
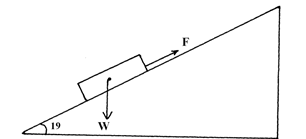
- On the same diagram show with an arrow one other force acting on the block and name the force. (1mark)
- A runway for an airport is designed such that the lowest acceleration rate for the plane is 3m/s2. The take off speed for the plane is 65m/s. Determine the minimum length for the runway. (2 marks)
- A motor cyclist wears a helmet lined on the inside with sponge. Explain how this minimizes injuries to the motorists head when involved in an accident. (2 marks)
- The figure shows a block hanging at rest from a ceiling by a piece of rope.
-
- The figure below shows masses A, B and C placed at different points on a rotating table
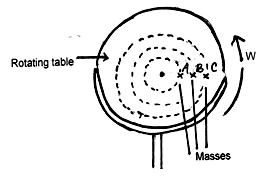
The angular velocity ω, of the table can be varied.- State two factors that determine whether a particular mass slides off the table or not. (2marks)
- It is found that the masses slide off at angular velocities ωA, ωB and ωC respectively. Arrange the values of ωA, ωB and ωC in decreasing order. (1mark)
- A block of mass 200g is placed on a frictionless rotating table while fixed to the centre of the table by a thin thread. The distance from the centre of the table to the block is 15cm. If the maximum tension the thread can withstand is 5.6N, determine the maximum angular velocity the table can attain before the thread cuts. (4marks)
- State pressure law of an ideal gas. (1 mark)
- When the temperature of a gas in a closed container is raised, the pressure of the gas increases. Explain how the molecules of the gas cause the increase in pressure. (2 marks)
- State one assumption for the experiments carried out to verify the gas laws.(1 mark)
- A constant mass of hydrogen gas occupies a volume of 4.0 cm3 at a pressure of 2.4 × 105 Pa and temperature of 15ºC. Determine its volume at a pressure of 1.6 × 105 Pa when the temperature is doubled. (3 marks)
- The figure below shows masses A, B and C placed at different points on a rotating table
-
- State what is meant by specific latent heat of vaporization. (1mark)
- In an experiment to determine the specific latent heat of vaporization of water, steam at 100ºC was passed into water contained in a well lagged copper calorimeter.
The following measurements were made:-
Mass of calorimeter = 50g
Initial mass of water = 70g
Initial temperature of water = 5ºC
Final mass of water + Calorimeter + condensed steam = 123g
Final temperature of mixture = 300C
Specific heat capacity of water = 4200JKg-1k-1
Specific heat capacity of copper = 390 JKg-1k-1- Determine the:-
- Mass of condensed steam (2marks)
- Heat gained by water and calorimeter. (2marks)
- Given that L is the specific latent of heat of vaporization of steam:
- Write an expression for the heat given out by steam. (1mark)
- Determine the value of L. (2marks)
- Determine the:-
- The figure shows two similar containers with equal amounts of methylated spirit at room temperature. A draught is blown over container A and their temperatures taken after sometime.
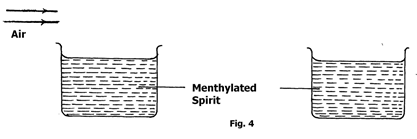
State and explain one that will have a lower temperature. (2marks)
-
- Two gear-wheels have 80 teeth and 20 teeth and they lock with each other. They are fastened on axles of equal diameter such that equal weight of 150N attached to the string around the axle will just raise 450N on the other axles.
Determine:- the mechanical advantage. (2marks)
- the velocity ratio. (2marks)
- The efficiency of this machine. (2marks)
- State any one possible way of increasing velocity ratio of wheel and axle. (1mark)
- A Loudspeaker is a transducer. Explain. (1mark)
- Two gear-wheels have 80 teeth and 20 teeth and they lock with each other. They are fastened on axles of equal diameter such that equal weight of 150N attached to the string around the axle will just raise 450N on the other axles.

MARKING SCHEME
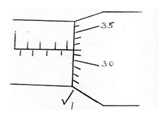
d = 2 x r
= 2 x 2 .17
= 4.34mm
Scale reading = Actual diameter – zero error
= 4.34 – 0.02
= 4.32mm-
- It contracts/reduce in volume upto 4ºC where its density in maximum.
- From 4ºC to 0ºC its volume increase thus the density reduces.
-
- Thermometer A shows a higher reading
- Black surface are good emitters of radiant heat.
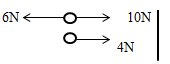
Resultant force = 10-6
= 4N towards the right hand side- Taking moments about the pivot
Sum of clockwise moments = same of anticlockwise moments
F1d1 = F2d2
40 x d = 10 x ( 2-d)
40d = 20 – 10d
40d + 10d = 20
50d = 20
d = 20/50=0.4m - A1V1 = A2V2
A1 x 0.1 = 0.05 x 2.2
A1 = 0.05 x 2.2
0.1
= 1.1m2
π(d/2)2 = 1.1
πd2 = 1.1
4
d2 = 1.1 x 4
π
d = 1.1 x 4
π
= 1.84m - Candela
- In liquids the forces are smaller and molecules are able to roll over one another while in gaseous state, the molecules are free to move because gases have the weakest forces of attraction.
-
- Leaward
- As water fills the versel, the centre of gravity rises and this lowers stability
- Gravitational force of attraction between the sum and versus
-
-
- Memory level inside the capillary tube is below the level outside
- Has a convex meniscus
-
- Presence of impurities
- Increase in pressure
-
- When a body/object is partially or wholly immersed in a fluid, it experiences an up thrust equal to the weight of the fluid displaced.
-
- To keep the hydrometer upright
- A
-
- 30g
- V = m/p = 30/1 = 30cm3
- V = 0.90 x 4
= 3.6cm3 - 30+3.6= 33.6cm3
- V=m/p = 30/1.5 = 20cm3
Volume of stem above = 33.6-20 = 13.6cm3
Length of stem above liquid = 13.6/0.9 = 15.11cm3
- When the sum of upthrust and air resistance balance with the weight of the gas and the balloon fabric
- The pressure at the base is reduced
This is because pressure in fluids is directly proportional to the depth of the fluid
-
-
-
- It has mass
- Since the rope is stretched
-
- Since the block is not moving relative to the surrounding / block is stationary / block at rest
- Block is not supported by any surface
-
-
- Normal reaction – correct direction
- V2 = u2 + 2as
652 = 02 + (2x3x5)
652 = 65
S = 704.2m
- This is to increase time for collision
This reduces the rate of change of momentum hence the impulsive force is reduced.
-
-
-
- Roughness/smoothness of surface
- Radius of path
- Angular velocity/speed (any two)
- F = mv2/5
5.6 = 0.2 x v2
0.15
v2 = 5.6 x 0.15
0.2
v2 = 4.2
v = 2.049m/s
v = rw
w = v/r
= 2.049
0.15
= 13.66 rad/s - pressure of a fixed mass of a gas is directly proportional to its absolute temperature provided volume is kept constant
- when a gas is heated molecules gain kinetic energy and move faster. If the volume of the container is constant, this increases the rate of Collins’s of the molecules with the walls of the container. The rate of change of momentum per unit area pressure increases.
- Size of the molecules is negligible
Intermolecular forces are negligible (any one) - PV/T = K
P1V1 = P2V2
T1 T2
2.4 x 105 x 4 = 1.6 x 105 x v2
T 2T
V2 = 2 x 2.4 x 4
1.6
= 12cm3
-
-
- Quantity of heat required to convert a unit mass of liquid to vapour at constant temperature.
-
-
- 1= 123 – (70 + 50)
= 123 – 120
= 3g - Total heat gained = 487.5 + 7350 = 7837.5J
Heat gained by water = MCΔθ
= 0.07 x 4200 x 25= 7350J
Heat gained by calorimeter = 0.05 x 390 x 25 = 487.5J
- 1= 123 – (70 + 50)
-
- 0.003L + 882 = 7837.5
0.003L = 6955.5
Heat lost by steam = mL + MCA
= 0.003L + (0.003L + (0.003 x 4200 x 70)
= 0.003L + 882 - L = 6955.5/0.003 = 2318500Jkg-1
- 0.003L + 882 = 7837.5
-
- Container A
The rate of evaporation is increased /more latent heat is drawn in A than in B.
-
-
- M.A. = L/E = 450/(150 ) = 3
- V.R = No.of teeth driven gear = 80/20
No.of teeth on drawing gear
= 4 - M = M.A/V.R X 100%
¾ x 100%
= 75%
- V.R = R/r (Radius of wheel)
(Radius of the axle)
Increase the radius of wheel (R)
Reduce the radius of the axle(r) (any one) - converts electrical energy to sound energy
-
Download Physics Paper 1 Questions and Answers - Kakamega Evaluation Mock Exams 2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students
