QUESTIONS
- You are provided with
- 4.5g of solid A in a boiling tube
- Solution B, 0.06M acidified potassium manganite (VII)
You are required to determine.- The solubility of solid A at different temperatures
- The number of moles of water of crystallization of solid A
Procedure 1
- Using a burette, add 4cm3 of distilled water to solid A in a boiling tube. Heat the mixture while string with a thermometer to about 70%. When all the solids bas dissolved allow the solution to cool while stirring with the thermometer. Note the temperature at which crystals of solid a first appear. Record this temperature in table 1.
- Using a burette add 2cm3 of distilled water to the content of the boiling tube. Warm the mixture while stirring the thermometer until all the solid dissolve Allow the mixture to cool while stirring. Note and record the temperature at which crystals of solid A first appears.
- Repeat procedure (b) two more times and record the temperature in table 1. Return the content of boiling tube for use in procedure.
-
- Complete the table calculating the solubility of solid A at different temperatures. The solubility of a substance is the mass of that substance that dissolves in 100cm3(100g) of water at a particular temperature. (6mks)
Table 1
Volume of water in the boiling tube (cm3) Temperature at which crystals of solid A first appear (°C) Solubility of solid A (g/100g of water) 4 6 8 10 - On the grid provided plot a graph of the solubility of solid A (vertical axis) against temperature. (3mks)
- Using your graph determine the temperature at which 100g of solid A would dissolve in 100cm3 of water. (1mk)
- Using your graph determine the solubility of solid A at 55°C. (1 mk).
- Complete the table calculating the solubility of solid A at different temperatures. The solubility of a substance is the mass of that substance that dissolves in 100cm3(100g) of water at a particular temperature. (6mks)
Procedure 2
(e)
- Transfer the content of the boiling tube into about 250ml volumetric flask. Rinse both the boiling tube and thermometer with distilled water and add it to the volumetric flask. Add more distilled water to make up to the mark. Label this solution A. Fill the burette with solution B using a pipette, place 25.0cm3 of solution A Into a conical flask. Warm the mixture to about 60°C. Titrate this hot solution A with solution B, until a permanent pink colour persist. Record your readings in table 2. Repeat the titration more times and complete table 2. (4mks)
Table 2
I II III Final burette reading (cm3) Initial burette reading (cm3) Volume of solution B used (cm3) - Calculate the
- Average volume of solution B used.
- Number of moles of potassium amangate (VII) used.
- Number of moles of A in 25cm3 of solution A given that 2moles of potassium manganite (VII) reacts completely with 5 moles
of A. -
- Relative formula mass of A.
- The formula of a has the formula of D.XH2O. Determine the value of x in the formula, given that the relative formula mass of D is 90. (O=16, H=1)
(2mks)
2. You are provided with solid FAS, FA6, and FA7. Carry out the following tests and write your observations and inferences in the spaces provided.
- Place all solid FA5 in the boiling tube. Add about 10cm3 of distilled water and shake until all the solid dissolves. Label this as solution FA5.
- To about 2cm3 of solution FA5 in a test tube, add 2M sodium hydroxide solution drop wise until in excess.
Observation Inferences - To about 2cm3 of solution FA5 (n a test tube, add 2M ammonium. hydroxide solution dropwise until in excess.
Observation Inferences - To about 2cm3 of solution FA5 in the test tube, add 4 drops of 2M sulphuric (VI) acid.
Observation Inferences - To about 2cm3 of solution FA5 in a test tube, add 2 drops of potassium lodide solution.
Observation Inferences
- To about 2cm3 of solution FA5 in a test tube, add 2M sodium hydroxide solution drop wise until in excess.
- Place solid FA7 into boiling tube. Add about 10cm of distilled water and shake well. Label this as solution FA7. Use this solution for the following tests.
- Place about 2cm3 of solution FA7 in a test tube and place the universal indicator paper provided into the solution hence determine its pH.
Observation Inferences - To about 2cm3 of solution FA7 made in (ii) above, add 3 drops of acidified.. potassium manganite(VII) solution.
Observation Inferences - To the remaining solution FA& in the boiling tube, add the other half of solid FA6.
Observation Inferences
- Place about 2cm3 of solution FA7 in a test tube and place the universal indicator paper provided into the solution hence determine its pH.
MARKING SCHEME
- You are provided with
- 4.5g of solid A in a boiling tube
- Solution B, 0.06M acidified potassium manganite (VII)
You are required to determine.- The solubility of solid A at different temperatures
- The number of moles of water of crystallization of solid A
Procedure 1
- Using a burette, add 4cm3 of distilled water to solid A in a boiling tube. Heat the mixture while string with a thermometer to about 70%. When all the solids bas dissolved allow the solution to cool while stirring with the thermometer. Note the temperature at which crystals of solid a first appear. Record this temperature in table 1.
- Using a burette add 2cm3 of distilled water to the content of the boiling tube. Warm the mixture while stirring the thermometer until all the solid dissolve Allow the mixture to cool while stirring. Note and record the temperature at which crystals of solid A first appears.
- Repeat procedure (b) two more times and record the temperature in table 1. Return the content of boiling tube for use in procedure.
-
- Complete the table calculating the solubility of solid A at different temperatures. The solubility of a substance is the mass of that substance that dissolves in 100cm3(100g) of water at a particular temperature. (6mks)
Table 1
Volume of water in the boiling tube (cm3) Temperature at which crystals of solid A first appear (°C) Solubility of solid A (g/100g of water) 4 67.0 - 69 112.5 6 75 8 56.25 10 45 - On the grid provided plot a graph of the solubility of solid A (vertical axis) against temperature. (3mks)
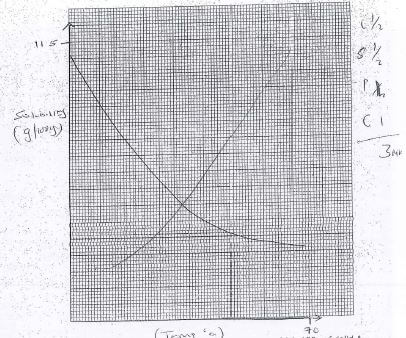
- Using your graph determine the temperature at which 100g of solid A would dissolve in 100cm3 of water. (1mk)
evidence 1/2 mk
value 1/2 mk
Reject for wrong shape of the graph - Using your graph determine the solubility of solid A at 55°C. (1 mk).
evidence 1/2 mk
value 1/2 mk
Reject for reading wrong graph
- Complete the table calculating the solubility of solid A at different temperatures. The solubility of a substance is the mass of that substance that dissolves in 100cm3(100g) of water at a particular temperature. (6mks)
Procedure 2
(e)
- Transfer the content of the boiling tube into about 250ml volumetric flask. Rinse both the boiling tube and thermometer with distilled water and add it to the volumetric flask. Add more distilled water to make up to the mark. Label this solution A. Fill the burette with solution B using a pipette, place 25.0cm3 of solution A Into a conical flask. Warm the mixture to about 60°C. Titrate this hot solution A with solution B, until a permanent pink colour persist. Record your readings in table 2. Repeat the titration more times and complete table 2. (4mks)
Table 2
s.v 250 cm I II III Final burette reading (cm3) Initial burette reading (cm3) Volume of solution B used (cm3) - Calculate the
- Average volume of solution B used.
show workings - Number of moles of potassium amangate (VII) used.
0.06 x A.V = ans
1000 - Number of moles of A in 25cm3 of solution A given that 2moles of potassium manganite (VII) reacts completely with 5 moles
of A. -
- Relative formula mass of A.
molarity = ans c x 1000 = ans
250 - The formula of a has the formula of D.XH2O. Determine the value of x in the formula, given that the relative formula mass of D is 90. (O=16, H=1)
(2mks)
x = (ans d(i) - 90) = ans
18
- Relative formula mass of A.
- Average volume of solution B used.
2. You are provided with solid FAS, FA6, and FA7. Carry out the following tests and write your observations and inferences in the spaces provided.
- Place all solid FA5 in the boiling tube. Add about 10cm3 of distilled water and shake until all the solid dissolves. Label this as solution FA5.
- To about 2cm3 of solution FA5 in a test tube, add 2M sodium hydroxide solution drop wise until in excess.
Observation Inferences white ppt
soluble in excess to form a colourless solutionZn2+, Pb2+, Al3+ present - To about 2cm3 of solution FA5 (n a test tube, add 2M ammonium. hydroxide solution dropwise until in excess.
Observation Inferences white ppt Pb2+, Al3+ present - To about 2cm3 of solution FA5 in the test tube, add 4 drops of 2M sulphuric (VI) acid.
Observation Inferences white ppt Pb2+present - To about 2cm3 of solution FA5 in a test tube, add 2 drops of potassium lodide solution.
Observation Inferences Yellow ppt Pb2+ ions present
- To about 2cm3 of solution FA5 in a test tube, add 2M sodium hydroxide solution drop wise until in excess.
- Place solid FA7 into boiling tube. Add about 10cm of distilled water and shake well. Label this as solution FA7. Use this solution for the following tests.
- Place about 2cm3 of solution FA7 in a test tube and place the universal indicator paper provided into the solution hence determine its pH.
Observation Inferences pH 4 or 5 or 6 weakly acidic solution - To about 2cm3 of solution FA7 made in (ii) above, add 3 drops of acidified.. potassium manganite(VII) solution.
Observation Inferences purple colour of H+/KMnO4 changes to colourless - To the remaining solution FA& in the boiling tube, add the other half of solid FA6.
Observation Inferences Effervescence O
||
H+, H3O+, R-COH
- Place about 2cm3 of solution FA7 in a test tube and place the universal indicator paper provided into the solution hence determine its pH.
Download Chemistry Paper 3 Questions and Answers - Mangu High School Mock Exams 2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students
