Instructions
- Write your name, admission number and class in the spaces provided above.
- Sign and write the date of examination in the spaces provided above.
- Answer all the questions in the spaces provided in the question paper.
- All working must be clearly shown where necessary.
- This paper consists of 11 printed pages. Confirm this and that no questions are missing.

QUESTIONS
- The grid below is part of a periodic table. Study it and answer the questions that follow. The letters are not the actual symbols of the elements
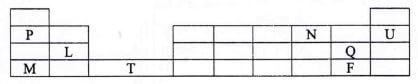
- Give
- The electron arrangement of an ion
P...................(1 mark)
N.................(1 mark) - The formula of the compound formed between elements L and Q (1 mark)
- The electron arrangement of an ion
- Which is the most reactive metallic element shown in the table? Explain (2 marks)
- What is the name given to the group of elements to which Q and F belong? (1 mark)
- Compare the atomic radius of P and N (2 marks)
- How do the ionization of P and M compare? Explain (2 marks)
- What name is given to the elements to which element 'T belong. (1 mark)
- Give
- The diagram below represents the extraction of Susiphur by Frasch process.
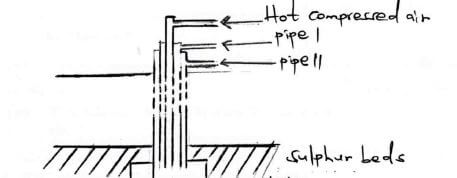
-
- Name the substance that passes through pipe (2 marks)
(i).......
(ii)...... - What is the purpose of hot compressed air in iis process (1 mark)
- Name the substance that passes through pipe (2 marks)
- Below is a flow diagram for an industrial process. Study it and answer the questions below.
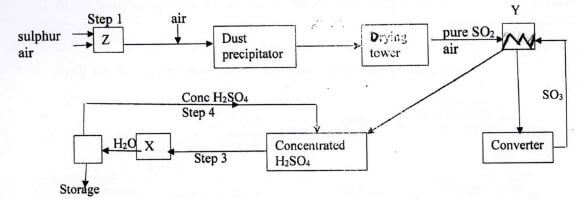
- Identify the name of the chambers labelled(2 marks)
X............
Y.............. - State any two conditions in the converter (1 mark)
- Explain why the gases are passed through;
- Dust precipitator and drying tower (1 mark)
- Chamber Y (1 mark)
- Write balanced chemical equation for reactions in;
Step 1 (1 mark)
Step 3(1 mark)
- Identify the name of the chambers labelled(2 marks)
- In an experiment, hydrogen sulphide gas was mixed with moist SO2 gas.
- What two observations are made in the boiling tube.(2 marks)
- Write an equation for the reaction taking place in the flask (1 mark)
-
- Study the flow-chart below and use to answer the questions that follow.
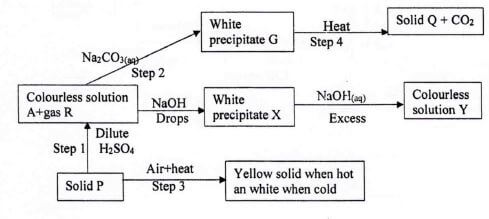
- Identify the
- White precipitate X (1 mark)
- Substance P (1 mark)
- Write an ionic equation for the formation of white precipitate G (1 mark)
- Write an ionic equation for the reaction in step 3 (1 mark)
- Name the ions present in colourless solution Y (1 mark)
- Give two uses of gas R (2 marks)
- Identify the
-
- Study the flow chart below and answer the questions that follow.
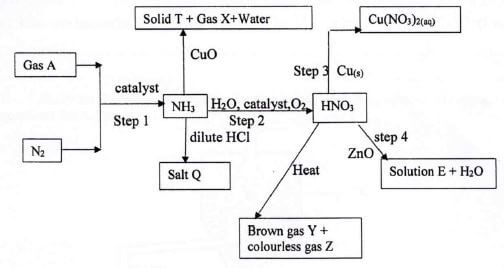
- Identify the following substances;
- Gas X (1 mark)
- Solution E (1 mark)
- Salt Q (1 mark)
- Give the 3 equations for the reactions that take place in step 2. (3 marks)
- Name the catalyst used in;(2 marks)
Step 1
Step 2 - State the process taking place in step I (1 mark)
- Give one industrial use of HNO3 (1 mark)
- Identify the following substances;
- Study the figure below for industrial preparation of HCL(aq) and use it to answer the questions that follow.
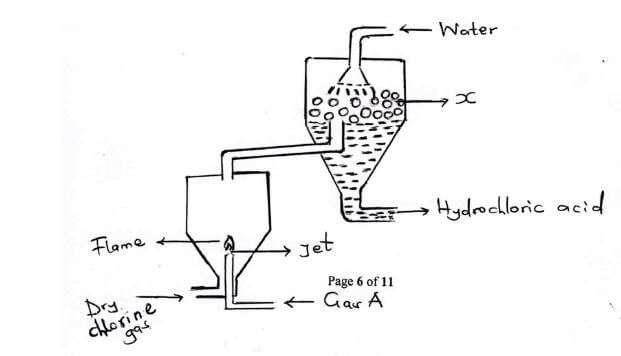
- State two sources of Gas A (1 mark)
- A small amount of gas A is allowed to burn in excess chlorine through a jet. Give a reason (1 mark)
-
- Name part X and give a reason for its presence (2 marks)
- Write a balanced chemical equation for the reaction taking place over part A. (1 mark)
- State one use of HCL (1 mark)
- Study the flow chart below and answer the questions that follow.
- The flow chart below is used to manufacture a detergent. Study and use to answer the questions that follow.
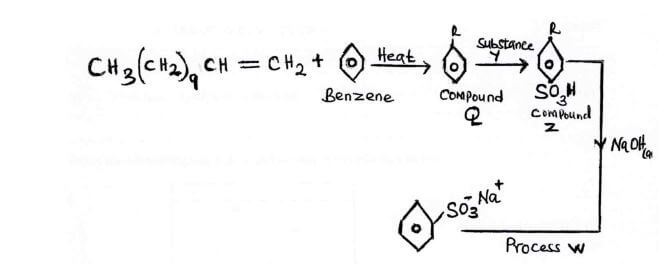
- Name components
- Q (1 mark)
- Z (1 mark)
-
- Identify reagent Y (1 mark)
- Process W (1 mark)
-
- name the type of detergent above (1 marks)
- state the disadvantage of the detergent above (1 mark)
- Give a reason for adding tetraxophosphate (v) to the detergents. (1 mark)
-
- Give two examples of natural polymers. (2 marks)
- What type of polymer is terylene (1 mark)
- Name components
- Study the chromatogram below and answer questions that follow.
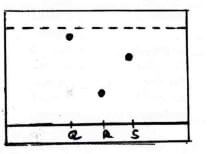
- Which is the
- Most soluble dye (1 mark)
- Most absorbed dye (1 mark)
- Possible solvent that can be used in the process above. (1 mark)
- On the diagram, show the chromatogram of T which is a mixture of R and S. (1 mark)
- State two applications of chromatography (2 marks)
- I In a different experiment, Fatuma a student in your school wanted to extract oil from simsim seeds in the laboratory.
- Which method of mixture separation would she use? (1 mark)
- Describe the method stated above as used in the experiment (3 marks)
- Which is the
-
- The table below gives some properties of substances A, B, C and D. Study it and answer the questions that follow.
Substance Electrical conductivity Melting point (°C) Boiling point (°C) Solid Molten A Does not conduct Conducts 800 1420 B Conducts Conducts 650 1108 C Does not conduct Does not conduct 1701 2200 D Does not conduct Does not conduct 113 441 - What type of bonding exists in substances A and B? (2 marks)
A ..........
B ........... - Which substance is likely to be Sulphur? Explain (2 marks)
- What type of bonding exists in substances A and B? (2 marks)
-
- Distinguish between covalent bond and ionic bond (2 marks)
- Using dots (.) and crosses (x) to represent electrons, show bonding in the following compounds.
- H3O+ (1 mark)
- CO (1 mark)
- In terms of structure and bonding, explain the following observations.
- The melting point of aluminium is higher than that of sodium (2 marks)
- Graphite conducts electricity but diamond does not conduct electricity (2 marks)
- Below is a structure of aluminium chloride.
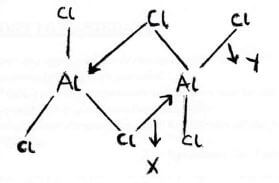
Name the bonds labelled x and y
X ................... (1/2 mark)
Y .................. (1/2 mark)
- The table below gives some properties of substances A, B, C and D. Study it and answer the questions that follow.
Download Chemistry Paper 2 Pre Mock Questions - Mokasa I Joint Examination July 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

