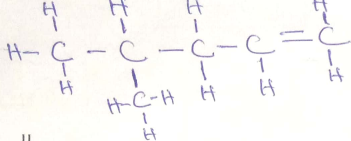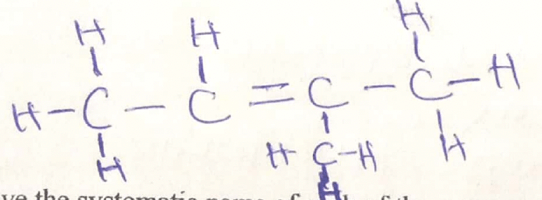INSTRUCTIONS TO CANDIDATES
- Answer all the questions in the spaces provided after each question.
- Mathematical tables and non-programmable electronic calculators may be used.
- ALL working must be clearly shown where necessary.
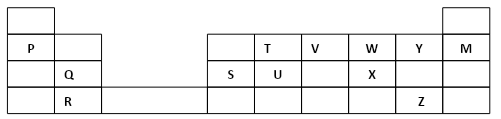
- The grid below forms part of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements
- Write the general name given to the element P belong. (1mark)
- An element N has an atomic number of 15. Write down its electronic arrangement and hence fix it in its right position on the grid above. (1mark)Electronic arrangement
- Compare the size of the atom of R and that of its ion. Explain your answer. (2mks)
- Give the formula of the compound formed between (1mark)
- P and W …………………………………………………………………………
- T and Y …………………………………………………………………………..
- Compare the melting points of element Q and S. Explain (2Mks)
- State the least reactive element in the grid. Give a reason for your answer (1mk)
- Give two advantages that element S has over element Q in making electric cables (2mks)
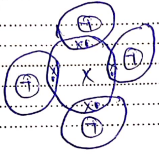
- Draw (a) dot (.) and cross (x) diagram to represent the bonding in compound formed between T and Y (2 marks)
-
- Study the table below and answer the questions that follow
Bond type bond energy kJmol-1
C-C 346
C = C 610
C-H 413
C-Br 280
Br-Br 193- Calculate the enthalpy change for the following reaction (3 marks) C2H4(g) + Br2(g)
C2H4Br2(g)
- Name the type of reaction that took place in (a) above. (1mark)
- Calculate the enthalpy change for the following reaction (3 marks) C2H4(g) + Br2(g)
- Butane C4H10 cannot be prepared directly from its elements but its standard heat of formation (
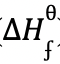 ) can be obtained indirectly.
) can be obtained indirectly.
The following heats of combustion are given.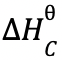 (Carbon) = -393kJ/mol
(Carbon) = -393kJ/mol 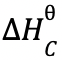 (Hydrogen) = -286kJ/mol
(Hydrogen) = -286kJ/mol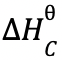 (Butane) =-2877kJ/mol
(Butane) =-2877kJ/mol- Draw an energy cycle diagram linking the heat of formation of butane with its heat of combustion and the heat of combustion of its constituents elements. (2mk)
- Calculate the heat of formation of butane
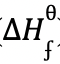 (C4H10) (2mks)
(C4H10) (2mks)
- Given that the lattice enthalpy of potassium chloride is +690kJ/mol and hydration enthalpies of K+ and Cl- are -322kJ and -364kJ respectively. Calculate the enthalpy of solution of potassium chloride. (3 mks)
- Study the table below and answer the questions that follow
- The diagram below represents the Haber process for the manufacture of ammonia. Study it and answer the questions that follow.
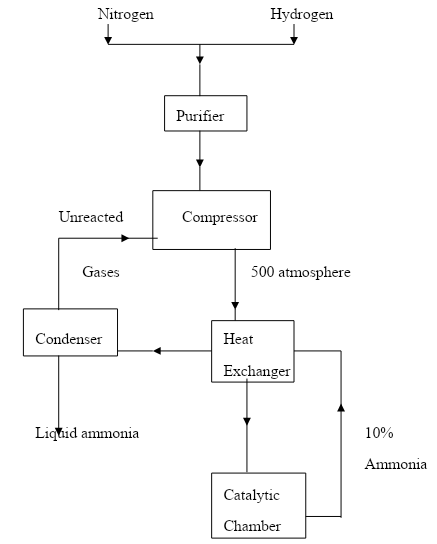
- Name any two impurities removed by the purifier. (1mark)
- The catalyst used in the process is finely divided iron. Why iron is finely divided? (1mk)
- In the Haber process the conversion of nitrogen and hydrogen into ammonia is only 10%.
The remaining unreacted gases are recycled. What is the advantage of this? (1mk) - A part from iron catalyst and pressure of 500 atmospheres, name any other condition required for this process. (1mk)
- Give any two uses of ammonia (1mk)
- In the manufacture of nitric (v) acid from ammonia and air, ammonia is catalytically oxidized to nitrogen (ii) oxide
- Name the catalyst used in this reaction. (1mk)
- Write a balanced chemical equation for the reaction between ammonia and air. (1mk)
- State one environmental problem likely to be faced in an area where nitric (v) acid manufacturing plant is located. (1mark)
-
- In the preparation of chlorine gas in a school laboratory, either manganese (IV) oxide or potassium manganate(VII) may be used on concentrated hydrochloric acid. State one advantage of potassium manganate (VII) over manganese (IV) oxide in this reaction.(1mark)
- State and explain what would be observed when dry litmus papers are dipped in a gas jar of chlorine.(1mark)
- Freshly prepared chlorine water bleaches but chlorine water exposed to sunlight for sometime does not bleach. Explain.(2marks)
- When preparing hydrogen chloride gas from sodium chloride and sulphuric (VI) acid, two conditions are necessary. State the conditions.(1mark)
- A label on the bottle containing Sulphuric (IV) acid has the following information
- Density = 1.836 g/cm3
- Percentage purity = 98%
- Relative formula mass = 98
- Calculate:
- The concentration of the acid (3 mks)
- The volume of concentrated sulphuric (IV) acid that should be diluted to produce 2 litres of 2 M Sulphiric (IV) acid (2 mks)
- A solution of sodium hydroxide was found to contain 12.4g/dm3 of sodium hydroxide. 25cm3 of this solution reacted with 15cm3 of a solution of sulphuric (VI) acid. (Na=23.0, H=1.0, S=32.0, O=16.0)
- Find the molarity of the sodium hydroxide solution. (1 mark)
- Calculate the number of moles of sodium hydroxide solution used. (1 mark)
- Calculate the number of moles of the acid used. (1 mark)
- Determine the concentration of the sulphuric (VI) acid solution in g/dm3. (3marks)
- Calculate:
-
- Define a saturated solution. (1 mark)
- The table below represent the solubilities of sodium nitrate and Sulphur (IV) oxide at different temperatures.
On the grid provided below, plot a graph of solubilities of sodium nitrate and Sulphur (IV) oxide against temperature. (4 marks)Temp(°C) 10 18 26 34 42 Solubility of Sodium Nitrate (g/100g of water) 20 29 40 53 68 Solubility of Sulphur (IV) Oxide (g/100g of water) 78 55 45 40 36
Using the graph;- Determine the solubility of Sulphur (IV) oxide at 16oC. (1 mark)
- The concentration, in moles per litre, of sodium nitrate at 16 oC. (assume density of solution is 1 g/cm3) (Na=23, 0=16, N=14). (3 marks)
- Mass of crystals formed when a solution of sodium hydroxide is cooled from 40oC to 26oC.
(2 marks) - What is the relationship between solubility of sodium nitrate and temperature? (1 mark)
- Give one advantage of hard water. (1 mark)
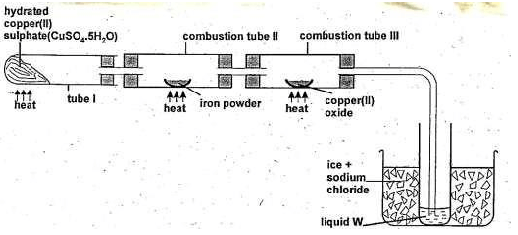
- The diagram below shows the apparatus for the preparation of gas A and investigate on its properties . Study it and answer the questions that follow.
-
- Name gas A. (1 mark)
- suggest property of gas A under investigation (1mark)
- Write chemical equations for the reactions in the;
- Boiling tube I (1 mark)
- Combustion tube II (1 mark)
-
- State and explain the observation made in
- Tube I. (1 mark)
- Combustion tube II (1 mark)
- State and explain the observation made in
-
- What is the use of hydrated copper (II) sulphate in the experiment? (1 mark)
- Name one other substance that comes out of tube III. (1 mark)
- Name liquid W. (1 mark)
- What is the role of sodium chloride in the ice (freezing mixture) (1 mark)
-
- Study the condensed formulae below and answer the questions that follow
-
- CH3CH (CH3) CH2CHCH2
- CH3CHCH(CH3)CH3
- Draw the structural formula of each of the compounds I and II. (2mks)
- Give the systematic name of each of the compounds represented by the formulae above
(2mks) - To which homologous series does the compound represented by I belong (1/2 mk)
- The flow chart below shows some reactions starting with a long chain alkane. Study it and answer the questions that follows.
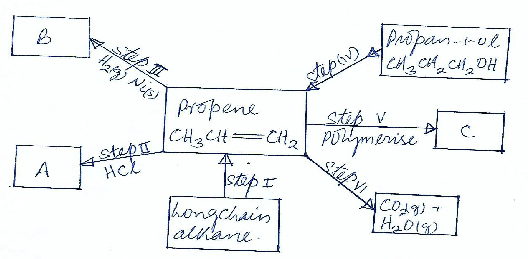
- Name substance. (11/2 mks)
- A___________________________
- B___________________________
- C____________________________
- What is the name given to the process represented by
- Step I_______________________ ( ½ mk)
- Step III______________________ ( ½ mk)
- Step IV______________________ ( ½ mk)
- Step VI______________________ (½ mk)
- Write down the chemical equation represented by the reaction in step VI (1mk)
- Name substance. (11/2 mks)
-
MARKING SCHEME
-
- Alkali metals
- 2.8.5
- R has a bigger atomic size kthan its ions because R looses electrons hence an energy level lost therefore making the size of its ion to be small than the atom.
-
- P2W
- TY4
- S has higher melting point than Q. This is because S has stronger metallic bond than. Q (3 electrons in outermost energy level requiring a lot of heat to break the bond in S than Q
- M. It is stable doesn't need to react with other elements to gain stability.
-
- It is ductile
- Does not corrode
- Has higher electrical conductivity.
-
-
-
- Bond breaking
C=C-1
C-H=4
Br-Br-1
610 + 4(413) + 193
= 2455
Bond formation
C-C=1× 346
C-Br - 2× 280
C-HF 4 × 413
= j2558
ΔH = 245J − 2558
= − 103kJmol−1 - Expthermic reaction/ Addition Reaction
- Bond breaking
-
-

- ΔH1 = ΔHF + ΔH2
ΔHF = ΔH1 − ΔH2
4(−393) + 5 (−286) −(−2877)
− 30002 + 2877
= − 125KJmol−1
-
- ΔHSoln = ΔHLatt + ΔHhyd
= 690 + 322 + −(364)
= 690 − 686
= +4kJmol−1
-
-
-
- Carbon(IV)Oxide
- Sulphur(IV) Oxide
- Dust particles
Any two ½mk each.
- To increase Its surface Area
- Reduces wastage
- Temperature of 1450-500ºC (No range)
-
- As nitrogenous fertilizer
- Softening of hard water
-
- Platinum - rhodium catalyst
- 4NH3(g) + 5O2(g) → 4NO + 6H2O(l)
- NO reacts nwith O2 to form NO2 which dissolve in moisture forming HNO3 falls as acid rain which causes death of sea life.
-
- Heat is not required.
- There would be no change in both litmus mpapers. Dry chlorine doesn't have acidic property hence do not bleach.
- Freshly prepared chlorine water has chloric(I) acid therefore bleaches. But when exposed to sunlight Chloric(I) acid decomposes into hydrochloric acid and oxygen gas is released.
-
- Heat
- The acid must be concentrated.
-
-
-
- Mass of acid in 1cm3 = 1.836g
Mass of acid in 1000cm3
1000 × 1.836 = 1836g
1
Mass of pure acid = 98/100 × 1836
= 1799.28g
M = Mass
RMM
= 1799.28
98
= 18.36M - M1V1 = M2V2
M1 = 18.36
V1 = ?
M2 = 2M
v2 = 2L
V1 = M₂V₂
M1
V1 = 2 × 2
18.36
= 0.21786L
- Mass of acid in 1cm3 = 1.836g
-
- g/L = Molarity × RFM
M = 12.4
40
= 0.31M - If 1000cm3 = 0.31
25cm3 = ?
25 × 0.31
1000
= 0.00775moles - 2NaOH + H2SO4 → Na2SO4 + H2O
Mole ratio
NaOH : H2SO4
2 1
0.00775 = 0.003875
2 - If 15cm3 = 0.003875
1000cm3 = ?
1000 × 0.003875
15
= 0.25833M
= 0.26M
RFM of H2SO4 = 98
g/L = M × R.F.M
= 0.26 × 98
= 25.32g/L
- g/L = Molarity × RFM
-
-
- A solution that cannot dissolve anymore solute at a particular temperature
-
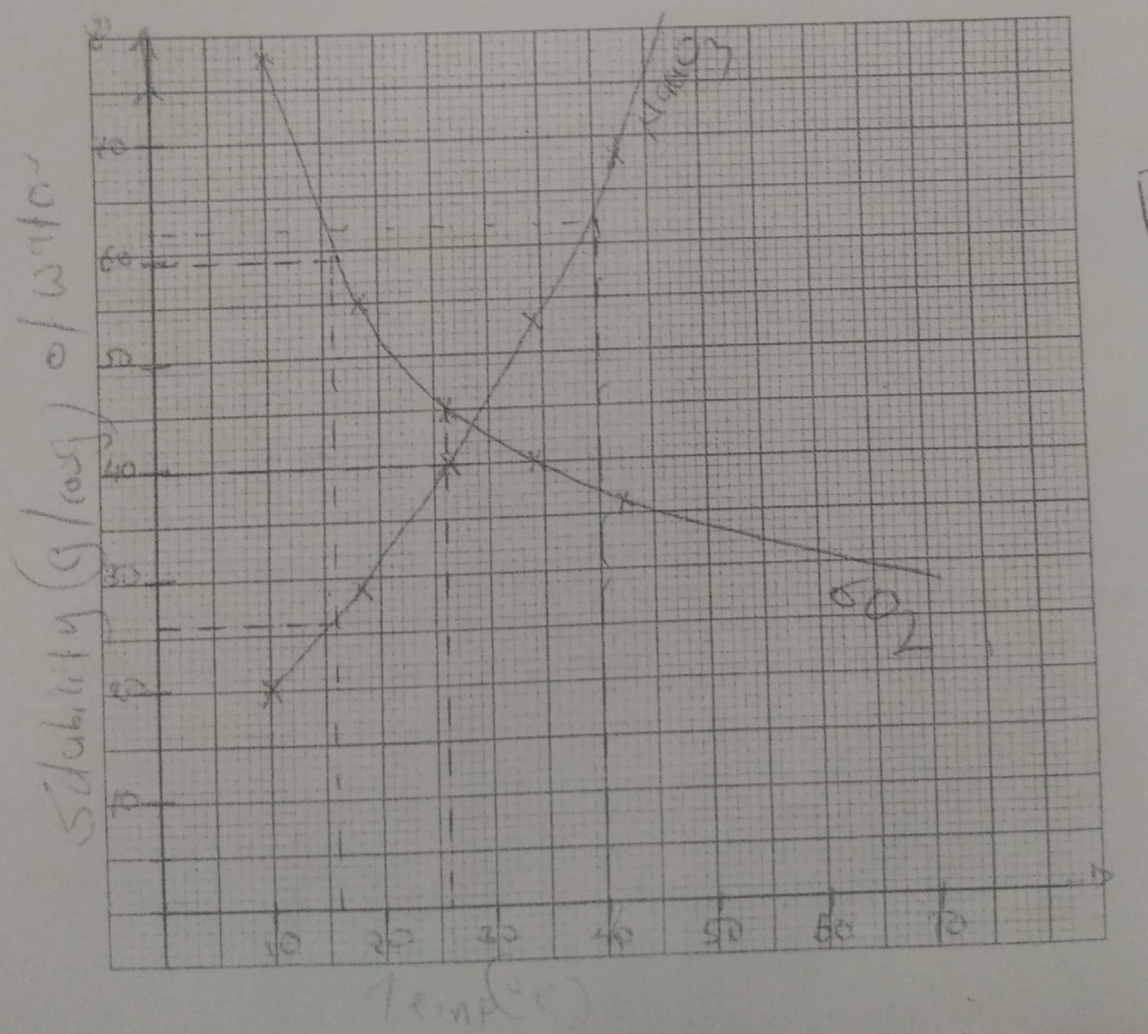
- 59g/100g ± 1 of water must be shown on graph
- At 16°C 26g/100g of water
100cm3 = 26g
1000cm3 = ?
1000 × 26
100
= 260
RFM = 23 + 14 + (16×3)
= 85g
M = 260/85 = 3.0588M - At 40°C = 62g/100gwater
At 26°C = 40g/100g water
Mass = 62 − 40
= 22g - As temperature increases, solubility of NaNO3 increases
-
- Used in brewing
- Provides Calcium - essential nutrient
- HCl is a strong acid, CH3COOH is a weak acid CH3COOH partially dissociates while HCl dissociates fully since some energy is used in fully ionising of CH3COOH
-
-
- Hydrogen
- Reducing property
-
- Heat
CuSO4.5H2CuSO4(s) + 5H2O(l)
- 3Fe(s) + H2O(l) → Fe3O4 + 4H2O(g)
- Heat
-
-
- Tube I
- Blue solid turns white/ colourless liquid is formed at cooler part of test tube
- CuSO4.5H2O losses water of crystallisation
- Combustion tube II
- Black solid turns brown. Copper (II) oxide reduced by H2(g) to copper metal.
- Tube I
-
-
- To provide steam
- Hydrogen
- Water
- Decrease freezing point of water
-
-
-
-
-
-
- 4 - methylpent -1- ene
- 2 - methylbut - 2- ene
- Alkenes
-
-
-
- A - 1, 2 - dichloropropane
- B - Propane
- C - Polythene/ polypropene
-
- Step I - Cracking
- Step III - Hydrogenation
- Step IV - Dehydration
- Step VI - Combustion
- 2C3H6(g) + 9O2(g) → 6CO2(g) + 6H2O(l)
-
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - Momaliche Pre Mock Exams 2023.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students