- State three differences between luminous and non-luminous flame. (3mks)
- The diagram below shows a non–luminous flame of the Bunsen burner
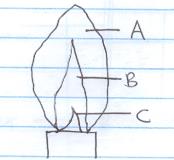
Name the parts of the flame labelled (3mks)
A
B
C
-
- What is a drug? (1mk)
- State two long- term effects of drug abuse (2mks)
- Putting off flames after use is one of the safety rules in the laboratory. State three other safety rules. (3 marks)
- Name four career opportunities open to a chemist? (4mk)
- The diagram below represents a paper chromatogram for three brands of juices suspected to contain banned food colourings?
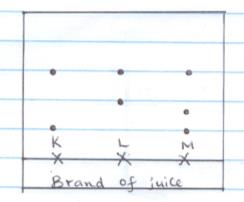
The results showed the presence of banned food colourings L and M only.
On the same diagram:- Circle the spots which show the banned food colouring? (2mks)
- Show the solvent front (1mk )
- The diagram below shows then relationship between the physical state of matter. Study it and answer the questions that follow
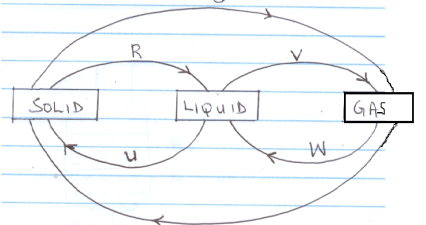
- Identify the following processes (2 mks)
R
V
W
U - Name two substances which can undergo the processes represented by S and I (2mks)
- Identify the following processes (2 mks)
- The set up below was used to determine the melting point of naphthalene.
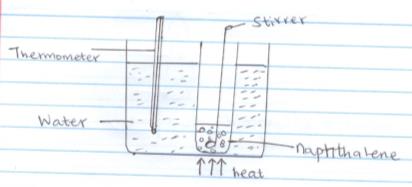
- State one precaution which should be taken into consideration when carrying out this experiment (1mk)
- State the use of the following in this experiment
- Thermometer (1mk)
- Stirrer (1mk)
- Boiling water (1mk)
-
- The apparatus shown below is commonly used in chemistry laboratory. Give its name and state one use. (2mks)

- Draw and name any two apparatus for measuring volume (2mks)
- The apparatus shown below is commonly used in chemistry laboratory. Give its name and state one use. (2mks)
- In an experiment to separate a mixture of two liquids A and B, a student set up the apparatus as shown below
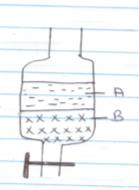
- Name the apparatus (1mk)
- Which liquid is denser (1mk
- Which other method can be used to separate the two liquids? (1mk)
-
- What is a mixture? (1mk)
- State an example for each of the following categories of mixtures. (3mks)
- Solid-Solid
- Solid-Liquid
- Liquid- Liquid
-
- Define a conductor (1mk)
- Categorise each of the following substances as either conductor or non-conductors of electricity. (2mks)
- Aluminium
- Rubber eraser
-
- State two skills acquired in the study of chemistry (2mk)
- Give two industries where the knowledge of chemistry is applied. (2mks)
- Study the diagram below and answer the questions that follow
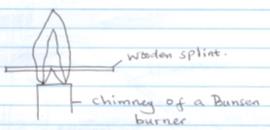
- State the condition under which the flame above is produced (1mk)
- A wooden splint was introduced into the flame as shown in the diagram. Draw a sketch showing the observation made on the splint and explain the observation made (3mk)
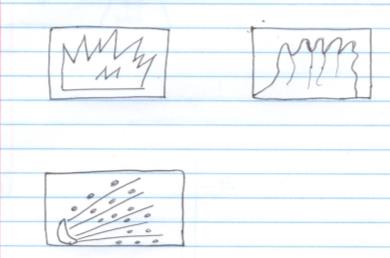
- The following symbols had been put on containers which certain chemicals identify the symbol and explain its meaning. (6marks)
- A student wanted to extract oil from cashew nuts. He first crushed the nuts then added propanone a little at a time while still crushing. He allowed it to settle, decanted the resulting solution then exposed it to sunlight for propanone to evaporate.
- Why was it necessary to crush the cashew nuts before adding propanone ( 1mk)
- Why was propanone preferred to water in the experiment (1mk)
- Name the method of extraction described above. (1mk)
- Study the diagram below and answer the questions that follow. It shows the heating curve for naphthalene.
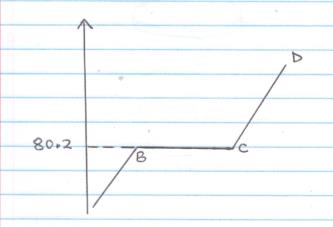
- Explain the shape of the graph at region BC (2mk)
- State whether the naphthalene used was pure or impure. Give a reason for your answer (2mk)
- Explain the behaviour of naphthalene particles at region AB (2mk)
-
- Differentiate between a temporary physical change and a temporary chemical change (1mk
- State two observations made when copper (ii) sulphate crystals are heated in a test tube (2mk)
- Is heating of cobalt (ii) chloride crystals a chemical change or a physical change (1mk)
- A student set up the following experiment to carry out fractional distillation
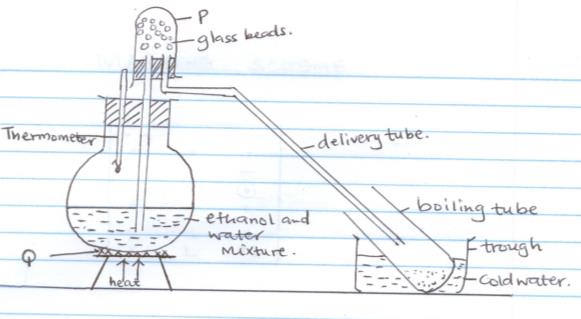
- Name the apparatus labelled
- P (1mk)
- Q (1mk)
- Write down one error that was made in setting up the apparatus. (1mk)
- What is the role of glass beads in fractional distillation (1mk)
- What other apparatus would you use to improve the role played by the cold water?
- Name the apparatus labelled
- What do you understand by the term over the counter (OTC) drugs (2mk)
- State why the following laboratory rules should be followed
- Never wear loose clothing in the laboratory (1mk)
- When heating a substance in a test tube or boiling tube never let the open end face you or your neighbour (1mk)
-
- Define matter (1mk)
- List the states of matter (1mk)
- The diagram below shows a Bunsen burner.
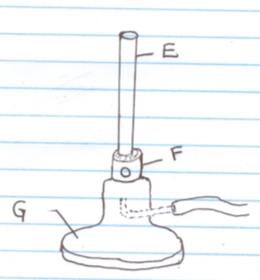
- Name the parts labelled
E
F
G - What is the role of the part labelled F? (1mk)
- Name the parts labelled
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download CHEMISTRY - Form 1 End of Term 1 2019 Examinations.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

