CHEMISTRY
FORM 1
MID TERM 2
INSTRUCTIONS:
- Answer all the questions
-
- What is Chemistry? {1mk}
- Define the following terms as used in chemistry;
- Matters {1mk}
- Mixture {1mk}
- Explain how you would distinguish a solid from a liquid {2mks}
-
- what is a drug {1mk}
- State two long term effects of drug abuse to the user {2mks}
- A form one student went to the school clinic and was prescribed malarial drugs to take 2 x3
- Explain how the student was supposed to take the drugs {2mks}
- Supposing the student took the drugs at 7.00a.m in the morning. Calculate the other hours of the day when he is expected to take the other drugs {2mks}
- State three ways in which chemistry has helped improve living standards in the society {3mks}
-
- State any four difference between luminous and non-luminous flame {4mks}
-
- Other than Bunsen burner name two other apparatus that are used in heating substances in the laboratory {2mks]
- Most of the laboratory apparatus are made of glass. Give three reasons {3mks}
- Name the apparatus used to measure the following;
- Accurate volume of liquids {three apparatus } {3mks}
- Amount of solid [one] apparatus {1mk}
- Temperature of boiling water [one]apparatus {1mk}
- Putting off flames not in use is one of the safety rules of laboratory to avoid injuries. List four other safety rules applied {4mks
- Draw and label a non-luminous flame {4mks}
-
- Name three major parts of bursen burner {3mks}
- State the functions of each of the part named in {a} above {3mks}
- The diagrams below are some common laboratory apparatus. Name each apparatus and state its use{8mks}
Apparatus Name Use i ii iii iv -
- What is a flame {1mk}
- Wooden splint W and Y were placed in different zones of a bursen burner flame. The diagram below shows the observations that were made
- State the zone of the flame that made
- the observation for W {2mks}
- the observation for Y
- Explain the difference between W and Y {2mks}
- Identify the most ideal flame used in the experiment above {1mk}
- State the zone of the flame that made
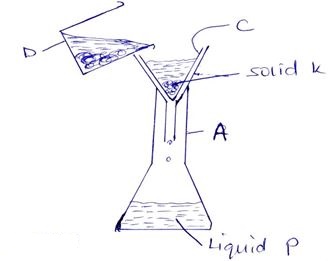
- Study the set-up shown below and answer the questions that follow;
- Name:
- Apparatus A {3mks}
- Apparatus C
- Apparatus D
- Name the method of separation shown above {1mk}
-
- Distinguish between a filtrate and residue {2mks}
- Identify them from the set-up above {2mks}
- Why is it possible to separate the mixture above using the method named in {b} above{1mk}
- Name:
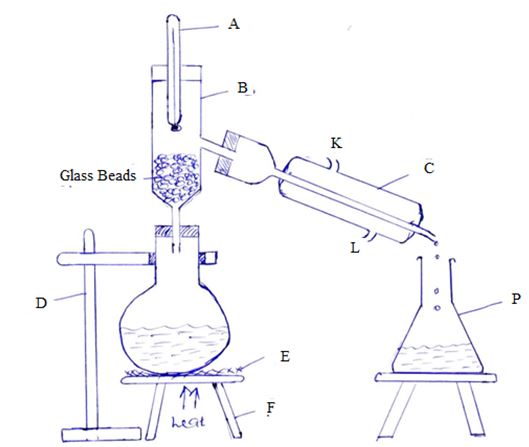
- The set-up below was used to separate a mixture of liquid M and N with boiling points of 680C and 780 respectively by the use of method K
- Name the method K {1mk}
- Name the apparatus {5mks}
- A
- B
- C
- D
- F
- State two properties of liquid M and N that makes them possible to separate by method K shown above {2mks}
- State one function of glass beads {1mk}
- Which letter represent;
- Water outlet in apparatus C {1mk}
- Water inlet in apparatus C {1mk}
- What is the effect of interchanging the water inlet and water outlet in apparatus C {1mk}
- What general name is given to the liquid collected in apparatus p {1mk}
- Give an example of two liquids that can be separated by method K {1mk}
MARKING SCHEME
-
- chemistry is the study of the structure, properties and composition of matter and the changes that matter undergoes. √1
-
- Matter is anything that has mass and occupies space. √1
- Mixture consists of two or more substances mixed together with the individual components forming the mixture retaining their physical and chemical properties. √1
-
- A solid has definite shape that is not easily changed while a liquid take the shape of the container. √1
- Solids have no ability to flow while liquids are able to flow. √1
-
- A drug is any substance natural or manufactured which when used alters the way the body functions. √1
-
- Addiction √1
- Dependency √1
- mental disorder
Any two correct
-
- 243 =8hrs√1
The student is to take 2tablets after every 8hrs. √1 - 7:00hrs
8:00
15:00hrs √1
8:00
23:00hrs √1
- 243 =8hrs√1
-
- manufacture of drugs to fight diseases. √1
- Food production to fight hunger. √1
- Manufacture of detergents. √1\
-
Luminous Non- Luminous Flame is not noisy Flame is noisy √1 Yellow flame Blue flame √1 Produces soot Does not produce soot √1 Large and wavy Small and steady √1
Any 4 correct -
-
- portable burner √1
- gas stove √1
- candle
Any 2 correct
-
- Easy to clean √1
- for easy making of observation since the apparatus are transparent √1
- do not react with most reagents used in the laboratory √
-
- Burette √1
Syringe √1
Pippete √1
Volumetric flask √1 - Weighing balance/ electrical balance √1
- Thermometer √1
- Burette √1
-
-
- Never smell gases directly instead waft the gas towards the nose with your hand.
- Incase of accident such as fire calmly walk out
- Label all the chemicals you are using to avoid confussion
- Always consult your teacher before trying any experiment to avoid accidents.
Any 4 correct
-
-
-
- Base √1
- Chimney √1
- Collar √1
-
- Base – provides support √1
- Chimney- ensures that gas and air mix before they start burning at the upper end of the chimney. √1
- Collar- helps to regulate the amount of air entering the chimney. √1
- a flame is a mass of burning gases √1
-
-
Apparatus Name Use i Round bottomed flask Used when heating liquid substances because heat is supplied uniformly
so that the flask does not crack as it expands. √1ii Thistle funnel Used for delivering liquid substances iii Pippete Used to measure accurate volume iv Deflagrating Spoon Used for holding substances being burned -
-
-
- almost colorless √1
- pale blue zone √1
- W is partly burnt because the wooden splint was placed on the cooler regions of e flame √1 while Y was burnt uniformly because the flame at that region is hotter. √1
- Non- Luminous flame √1
-
-
-
-
- A- conical flask √1
- C- filter paper √1
- D- Beaker √1
- Filtration √1
-
- Filtrate is the liquid that passes through the filter paper √1 while residue is the solid that remains on the filter paper when filtering. √1
- Filtrate- Liquid P √1
- Residue- solid K √1
- They are immiscible substances √1
-
-
- Fractional Distillation √1
-
- Thermometer √1
- Fractionating Column √1
- Lie- big condenser √1
- Clamp and stand √1
- Tripod stand √1
-
- Are miscible liquids √1
- Different but close boiling points √1
- To increase the surface area for the condensation of vapour coming from the distillation flask. √1
-
- K √1
- L √1
- If the water inlet and outlet are interchanged condensation would not occur efficiently or effectively. √1
- Distillate √1
- Ethanol and water, propanone and ethanol, propanol and water. √1
Any 2 correct
Download Chemistry Questions and Answers - Form 1 Mid Term 2 Exams 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students