CHEMISTRY
FORM 2
MID TERM
TERM 1
INSTRUCTIONS
- Answer all the questions
- Define the following:
- Element (1mk)
- Ion (1mk)
-
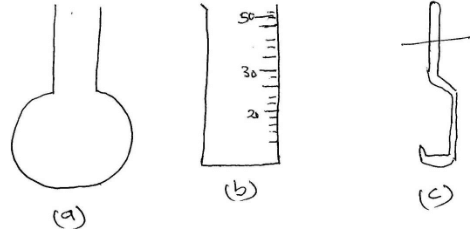
- Identify the following apparatus and give a use for each (3mks)
- Name another apparatus that can be used in place of (b) (1mk)
- Identify the following apparatus and give a use for each (3mks)
- Give four reasons why most apparatus are made of glass (4mks)
- Define the following terms
- Isotope (1mk)
- Ionization energy (1mk)
- Electron affinity (1mk)
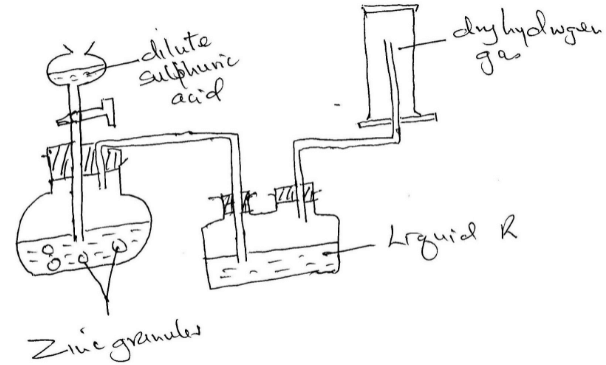
- Hydrogen gas was prepared in the lab. Using the following set up
- Write an equation for the reaction taking place and balance it (2mks)
- Name the method used to collect the gas and give a property of hydrogen that enables it to be collected through the method. (2mks)
- Name liquid R and state its function in the set up (2mks)
Liquid R:
Function: - Explain why it is not advisable to use sodium metal in place of zinc metal (2mks)
- State two uses of hydrogen gas (2mks)
- What will happen to the pH of the solution in the beaker after one day? Give an explanation.(2mks)
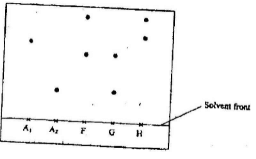
- Samples of urine from three participants F, G and H at an international sports meeting were spotted onto a chromatography paper alongside two from illegal drugs A1 and A2. A chromatogram was run using methanol. The figure below shows the chromatogram.
- Identify the athlete who had used an illegal drug (1mk)
- Which drug is more soluble in methanol? (1mk)
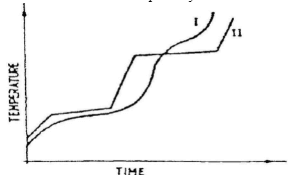
- The curve below represents the variation of temperature with time when pure and impure samples of a solid were heated separately.
Which curve shows the variation in temperature for the pure solid? Explain. (2mks) - In an experiment, a test-tube full of chlorine water was inverted in chlorine water as shown in the diagram below and the set up left in sunlight for one day.
After one day, a gas was found to have collected in the test-tube- Identify the gas (1mks)
- How can the above gas be tested? (2mks)
- The table below shows some properties and electronic arrangements of common ions of elements represented by letters P to X. Study the information in the table and answer the questions that follow
Element Ion Electron arrangement Atomic radius Ionic radius P P2+ 2,8,8 0.197 0.099 Q Q- 2,8 0.072 0.136 R R+ 2,8,8 0.231 0.133 S S3+ 2,8 0.143 0.050 T T2+ 2,8,8 0.133 0.074 U U2+ 2,8 0.160 0.065 V V+ 2,8 0.186 0.095 W W+ 2 0.152 0.060 X X- 2,8,8 0.099 0.181 - Give the atomic numbers of the elements P and Q (2mks)
P -
Q – - Select the most reactive metallic element (1mk)
- Select 3 elements that belong to the same group of periodic table (2mks)
- Select 3 elements that would react with cold water to evolve hydrogen gas (1mk)
- Why is the ionic radius of element X larger than its atomic radius? (1mk)
- Write an equation of the reaction between element S and Oxygen (2mks)
- Give the atomic numbers of the elements P and Q (2mks)
- Moist iron wool was inverted over water. The set up was left to stand for 2 days
- Explain whether rusting is a physical or chemical reaction (2mks)
- Write an expression using X and Y to show the percentage of Oxygen (2mks)
- What would be the effect of using a larger piece of iron wool? Explain. (2mks)
- State two similarities between rusting and combustion (2mks)
- Observe the equation below
Fe2O3(s) + CO(g) → Fe(s) + CO2(g)- Balance the equation (1mk)
- Select the following from the above equation
Oxidizing agent (1mk)
Reducing agent (1mk) - State two situations where redox reactions are applied in industry (2mks)
- Carbon (IV) sublimes at -78ºC. It is called dry ice
- Why is it called dry ice? (1mk)
- It is used for keeping ice cream cold. Why is it preferred to ordinary ice? (2mks)
- Name two other substances that behave as dry ice (2mks)
- Give an industrial application of sublimation (1mk)
-
- Element X has two isotopes. Two thirds of 3316X and one-third 3016X. What is the relative mass of element X?
- An element, A, has 30 protons and 35 neutrons. What is (2mks)
- The mass number of element A?
- The charge on the most stable ion of element A?
- An element B consists of three isotopes of mass, 28, 29 and 30 and percentage abundances of 92.2, 4.7 and 3.1 respectively. Show that the relative atomic mass of element is 28.11 (4mks)
- Elements X and Y have atomic numbers 11 and 17 respectively. Which one of the elements is a metal? Give a reason for your answer. (2mks)
- The table below shows the atomic numbers of four elements W,X,Y and Z
Which two elements belong to the same group? (2mksElement W X Y Z Atomic number 20 17 19 9 - Two elements M and N have atomic numbers 17 and 20 respectively. Write the formula of the compound formed when M and N react. (1mk)
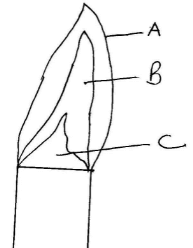
- The following diagram represents a non-luminous flame of the Bunsen burner
- Name the parts of the flame labeled A, B and C (3mks)
- Which of the parts in (a) above is the hottest? (1mk)
- A non-luminous flame is preferred for heating. Explain (2mks)
-
- Name the other type of flame produced by a Bunsen burner (1mk)
- Under what conditions does the Bunsen burner produce the flame in d(i)? (1mk)
- Define the following terms as used in medicine
- Drug (1mk)
- Prescription (1mk)
- Dosage (1mk)
- Drug abuse (1mk)
- Balance the following chemical equations
- Mg + O2 → MgO (1mk)
- Mg + N2 → Mg3N2 (1mk)
- Al + HCl → AlCl3 + H2 (1mk)
- C3H8 + O2 → CO2 + H20 (1mk)
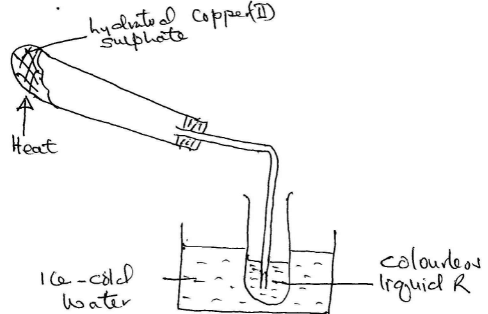
- Hydrated Copper (II) Sulphate is heated in a boiling tube as shown.
- State the colour of Copper (II) Sulphate before and after heating? (1mk)
- Explain why the boiling tube was slanted (1mk)
- How can the purity of the colourless liquid be confirmed? (1mk)
- Name another substance that can undergo the same change as hydrated Copper (II) Sulphate (1mk)
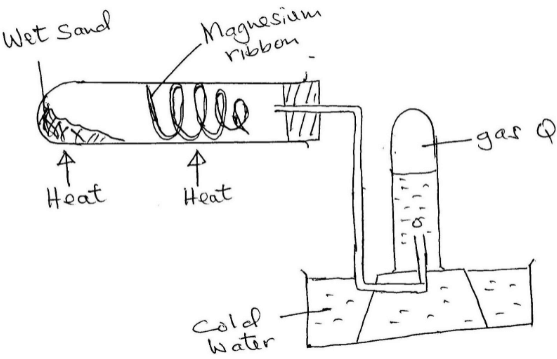
- A Magnesium ribbon was cleaned with steel wool and used in the following set up. Wet sand was heated before Magnesium ribbon.
- Explain the following:
- Sand was heated first before heating Magnesium ribbon (1mk)
- Magnesium ribbon was cleaned with steel wool (1mk)
- Name gas R (1mk)
- Write an equation for the reaction taking place in the combustion tube (1mk)
- Name the method used to collect gas R (1mk)
- Explain the following:
MARKING SCHEME
- Define the following:
- Element (1mk)
- Pure substance which cannot be split into simpler substance by chemical means
- Pure substance which cannot be split into simpler substance by chemical means
- Ion (1mk)
- Charged atom(s)
- Element (1mk)
-
- Identify the following apparatus and give a use for each (3mks)
- - round bottomed flask – heating substances (½mk)
- - Measuring cylinder/Measuring volume of liquids (½mk)
- - Spatula/Scooping solids/chemicals from containers (½mk)
- Name another apparatus that can be used in place of (b) (1mk)
- Burette/syringe/beaker (1mk any one correct)
(Reject apparatus that can’t measure accurate volumes)
- Burette/syringe/beaker (1mk any one correct)
- Identify the following apparatus and give a use for each (3mks)
- Give four reasons why most apparatus are made of glass (4mks)
- Easy to clean
- Transport
- Unreactive
- Modelled into many shapes
- Recyclable (Any 4 correct 1mk each)
- Define the following terms
- Isotope (1mk) - Atoms of same element having different number of neutrons hence different mass number (1mk)
- Ionization energy (1mk) - Energy required to remove an electron from an atom to form an ion in gaseous state (1mk)
- Electron affinity (1mk) - Energy required to capture/gain an electron by an atom to form an ion in gaseous state
- Isotope (1mk) - Atoms of same element having different number of neutrons hence different mass number (1mk)
- Hydrogen gas was prepared in the lab. Using the following set up
- Write an equation for the reaction taking place and balance it (2mks)
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
Wrong formula = no mark
Symbols ½
Balance ½ - Name the method used to collect the gas and give a property of hydrogen that enables it to be collected through the method. (2mks)
- Upward delivery/downward displacement of air (1mk)
- It is less denser than air (1mk)
- Name liquid R and state its function in the set up (2mks)
Liquid R: - Conc. Sulphuric (VI) acid (1mk)
Function: - For drying hydrogen (1mk) - Explain why it is not advisable to use sodium metal in place of zinc metal (2mks)
- Reaction would be explosive/dangerous because sodium is very reactive (1mk)
- Reaction would be explosive/dangerous because sodium is very reactive (1mk)
- State two uses of hydrogen gas (2mks)
- Manufacture of hydrochloric acid
- Manufacture of ammonia
- Hydrogenation of oils to form fats
- Weather balloons (rej. Air balloon)
- In oxy-hydrogen flame for welding
- As rocket fuel
- As fuel cells (Any 2 correct, a mark each)
- What will happen to the pH of the solution in the beaker after one day? Give an explanation.(2mks)
- It goes down.
- The solution becomes more acidic.
- It goes down.
- Write an equation for the reaction taking place and balance it (2mks)
- Samples of urine from three participants F, G and H at an international sports meeting were spotted onto a chromatography paper alongside two from illegal drugs A1 and A2. A chromatogram was run using methanol. The figure below shows the chromatogram.
- Identify the athlete who had used an illegal drug (1mk)
- G(1mk)
- G(1mk)
- Which drug is more soluble in methanol? (1mk)
- From A1 (1mk)
- From A1 (1mk)
- Identify the athlete who had used an illegal drug (1mk)
- The curve below represents the variation of temperature with time when pure and impure samples of a solid were heated separately.
Which curve shows the variation in temperature for the pure solid? Explain. (2mks)- I – does not have definite Mpt and Bpt (1mk)
- I – does not have definite Mpt and Bpt (1mk)
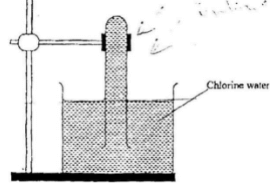
- In an experiment, a test-tube full of chlorine water was inverted in chlorine water as shown in the diagram below and the set up left in sunlight for one day.
After one day, a gas was found to have collected in the test-tube- Identify the gas (1mks)
- Oxygen (1mk)
- Oxygen (1mk)
- How can the above gas be tested? (2mks)
- Introduce a glowing splint into the gas (1mk)
- It will relight (1mk)
- Identify the gas (1mks)
- The table below shows some properties and electronic arrangements of common ions of elements represented by letters P to X. Study the information in the table and answer the questions that follow
Element Ion Electron arrangement Atomic radius Ionic radius P P2+ 2,8,8 0.197 0.099 Q Q- 2,8 0.072 0.136 R R+ 2,8,8 0.231 0.133 S S3+ 2,8 0.143 0.050 T T2+ 2,8,8 0.133 0.074 U U2+ 2,8 0.160 0.065 V V+ 2,8 0.186 0.095 W W+ 2 0.152 0.060 X X- 2,8,8 0.099 0.181 - Give the atomic numbers of the elements P and Q (2mks)
P - 20 (1mk)
Q –9 (1mk) - Select the most reactive metallic element (1mk)
R - Select 3 elements that belong to the same group of periodic table (2mks)
- P,T,U All 3 = 2mk/2 = 1mk/Otherwise no mark
- P,T,U All 3 = 2mk/2 = 1mk/Otherwise no mark
- Select 3 elements that would react with cold water to evolve hydrogen gas (1mk)
- P, R, T, U, V (Any 3 = 1mk)
- P, R, T, U, V (Any 3 = 1mk)
- Why is the ionic radius of element X larger than its atomic radius? (1mk)
- Form ions by gaining electrons ½mk/ Since it is a non-metal/resulting in electron-electron ½mk repulsion
- Form ions by gaining electrons ½mk/ Since it is a non-metal/resulting in electron-electron ½mk repulsion
- Write an equation of the reaction between element S and Oxygen (2mks)
- 4S + 3O2 → 2S2O3
- 4S + 3O2 → 2S2O3
- Give the atomic numbers of the elements P and Q (2mks)
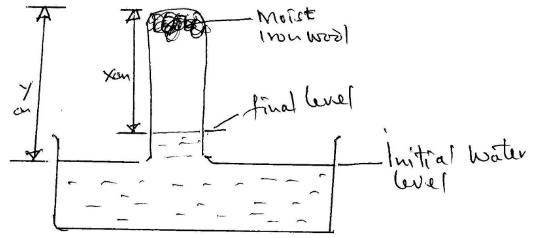
- Moist iron wool was inverted over water. The set up was left to stand for 2 days
- Explain whether rusting is a physical or chemical reaction (2mks)
- Chemical change 1mk/new substance formed
- Chemical change 1mk/new substance formed
- Write an expression using X and Y to show the percentage of Oxygen (2mks)
Oxygen = Y – X x 100
Y
1mk for x – y
1mk for %age - What would be the effect of using a larger piece of iron wool? Explain. (2mks)
- No change 1mk/ since % of oxygen is the same OR Vol. of O2 is fixed 1mk
- No change 1mk/ since % of oxygen is the same OR Vol. of O2 is fixed 1mk
- State two similarities between rusting and combustion (2mks)
- New substance formed
- Mass increases
- Heat change involved (Any 2 correct = 1mk each)
- Explain whether rusting is a physical or chemical reaction (2mks)
- Observe the equation below
Fe2O3(s) + CO(g) → Fe(s) + CO2(g)- Balance the equation (1mk)
Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO2 (1mk) - Select the following from the above equation
Oxidizing agent (1mk)- Fe2O3
Reducing agent (1mk)- CO - State two situations where redox reactions are applied in industry (2mks)
- Extraction of metals e.g. Iron 1mk each
- Purification of metals e.g. Iron
(Any other correct)
- Balance the equation (1mk)
- Carbon (IV) sublimes at -78oC. It is called dry ice
- Why is it called dry ice? (1mk)
- Sublimes without leaving a liquid
- Sublimes without leaving a liquid
- It is used for keeping ice cream cold. Why is it preferred to ordinary ice? (2mks)
- Leaves no liquid to spoil cream
- Takes longer to sublime
- Has a wider sublimation temp.
- Name two other substances that behave as dry ice (2mks)
- Iodine Iron III chloride naphthalene (1mk)
- Iodine Iron III chloride naphthalene (1mk)
- Give an industrial application of sublimation (1mk)
- Extraction of Zinc metal (any correct = 1mk)
- Why is it called dry ice? (1mk)
-
- Element X has two isotopes. Two thirds of 3316X and one-third 3016X. What is the relative mass of element X?
2 x 33 + 1 x 30 Total 4mks
3 3 - An element, A, has 30 protons and 35 neutrons. What is (2mks)
- The mass number of element A?
30 + 35 = 65 (1mk) - The charge on the most stable ion of element A?
A2+ or +2 (1mk)
- The mass number of element A?
- An element B consists of three isotopes of mass, 28, 29 and 30 and percentage abundances of 92.2, 4.7 and 3.1 respectively. Show that the relative atomic mass of element is 28.11 (4mks)
- 92.2 x 28 + 4.7 x 29 + 3.1 x 30 = R.A.M
100 100 100
25.816 + 1.363 + 0.93 = 28.11
Show percentages = 2mks
Work outs = 1mk
Ans. = 1mk
- 92.2 x 28 + 4.7 x 29 + 3.1 x 30 = R.A.M
- Elements X and Y have atomic numbers 11 and 17 respectively. Which one of the elements is a metal? Give a reason for your answer. (2mks)
- Y / has 7 outermost electrons showing it is in group 7/a halogen (1mk)
- Y / has 7 outermost electrons showing it is in group 7/a halogen (1mk)
- The table below shows the atomic numbers of four elements W,X,Y and Z
Which two elements belong to the same group? (2mks)Element W X Y Z Atomic number 20 17 19 9 - X and Z (1mk)
- X and Z (1mk)
- Two elements M and N have atomic numbers 17 and 20 respectively. Write the formula of the compound formed when M and N react. (1mk)
- NM2 (1mk)
- NM2 (1mk)
- Element X has two isotopes. Two thirds of 3316X and one-third 3016X. What is the relative mass of element X?
- The following diagram represents a non-luminous flame of the Bunsen burner
- Name the parts of the flame labeled A, B and C (3mks)
- Pale blue zone (1mk)
- Green blue zone (1mk)
- Almost colourless zone (1mk)
- Which of the parts in (a) above is the hottest? (1mk)
- A 1mk
- A 1mk
- A non-luminous flame is preferred for heating. Explain (2mks)
- It is very hot/hotter than luminous does not produce soot (Any correct 1mk each)
- It is very hot/hotter than luminous does not produce soot (Any correct 1mk each)
-
- Name the other type of flame produced by a Bunsen burner (1mk)
- Luminous flame
- Luminous flame
- Under what conditions does the Bunsen burner produce the flame in d(i)? (1mk)
- When air hole is closed
- When air hole is closed
- Name the other type of flame produced by a Bunsen burner (1mk)
- Define the following terms as used in medicine
- Drug (1mk)
- Any substance, natural or manufactured which when used alters body functions (1mk)
- Any substance, natural or manufactured which when used alters body functions (1mk)
- Prescription (1mk)
- giving written instructions by a qualified medical officer giving details on type
of drugs and how they should be used (1mk)
- giving written instructions by a qualified medical officer giving details on type
- Dosage (1mk)
- amount of drug/medicine to be taken at a time or regularly on a period of time
- amount of drug/medicine to be taken at a time or regularly on a period of time
- Drug abuse (1mk)
- Use of a drug for a use other than what it is meant for/under prescription or over prescription (1mk)
- Use of a drug for a use other than what it is meant for/under prescription or over prescription (1mk)
- Drug (1mk)
- Name the parts of the flame labeled A, B and C (3mks)
- Balance the following chemical equations
- Mg + O2 → MgO (1mk)
2Mg(s) + O2(g)→2MgO(s) (1mk) - Mg + N2 → Mg3N2 (1mk)
3Mg(s)+ N2(g) →Mg3N2(s) (1mk) - Al + HCl → AlCl3 + H2 (1mk)
2Al + 6HCl(aq)→2AlCl3(aq) + 3H2(aq) (1mk) - C3H8 + O2 → CO2 + H20 (1mk)
C3H8(g) + O2(g)→3CO2(g) + 4H20(l) (1mk)
- Mg + O2 → MgO (1mk)
- Hydrated Copper (II) Sulphate is heated in a boiling tube as shown.
- State the colour of Copper (II) Sulphate before and after heating? (1mk)
- Before – blue ½ mk/ after – white ½ mk
- Before – blue ½ mk/ after – white ½ mk
- Explain why the boiling tube was slanted (1mk)
- Avoid boiling tube cracking due to condensed water flowing back (1mk)
- Avoid boiling tube cracking due to condensed water flowing back (1mk)
- How can the purity of the colourless liquid be confirmed? (1mk)
- Test its Bpt/Mpt/density (any 1 correct = 1mk)
- Test its Bpt/Mpt/density (any 1 correct = 1mk)
- Name another substance that can undergo the same change as hydrated Copper (II) Sulphate (1mk)
- Any hydrated slat (1mk)
- Any hydrated slat (1mk)
- State the colour of Copper (II) Sulphate before and after heating? (1mk)
- A Magnesium ribbon was cleaned with steel wool and used in the following set up. Wet sand was heated before Magnesium ribbon.
- Explain the following:
- Sand was heated first before heating Magnesium ribbon (1mk)
- to generate steam for – driving air ½ mk from apparatus; react with Magnesium ½ mk
- to generate steam for – driving air ½ mk from apparatus; react with Magnesium ½ mk
- Magnesium ribbon was cleaned with steel wool (1mk)
- Remove layer of Magnesium oxide to allow Magnesium to combine with steam (1mk)
- Remove layer of Magnesium oxide to allow Magnesium to combine with steam (1mk)
- Sand was heated first before heating Magnesium ribbon (1mk)
- Name gas R (1mk)
- Hydrogen (1mk)
- Hydrogen (1mk)
- Write an equation for the reaction taking place in the combustion tube (1mk)
- Mg(s) + H2O(g)→MgO(s) + H2(g) (1mk)
- Mg(s) + H2O(g)→MgO(s) + H2(g) (1mk)
- Name the method used to collect gas R (1mk)
- Over water method (1mk)
- Explain the following:
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Questions and Answers - Form 2 Term 1 Mid Term Exams.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students