INSTRUCTION TO CANDIDATES
- Answer all the questions in the spaces provided.
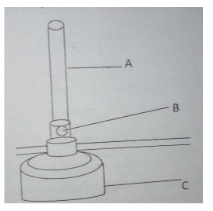
- The diagram below shows parts of the Bunsen burner.
- Name the parts labelled (1 mark).
- A
- B
- Give one use of the part labelled B (1 mark).
- Name the parts labelled (1 mark).
- Name two apparatus that are used to measure exact volumes (2 marks)
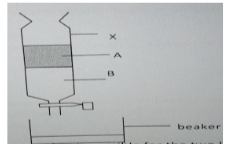
- The diagram below represents a method of separation used to separate two liquids , A and B,
Use it to answer the questions that follow.- Name the method of separation shown above (1 mark).
- Name two properties that makes it possible for the two liquids to be separated (2 marks).
- Give one alternative method that may be used to separate the two liquids (1 mark).
- In an experiment, two pieces of iron sheet were wrapped in each case with zinc and copper metal sheets as shown below. They were left in the open for some months.
State and explain the observations made in the experiment I and II (3 marks). - Given zinc oxide, dilute nitric (v) acid and sodium carbonate solution. Briefly describe how you can prepare zinc carbonate (3 marks).
- State two solids that may be heated to obtain oxygen gas as the only gas (2 marks).
- Study the information given below and answer the questions that follow.
Formula of the compound. NaCl MgCl2 Al2Cl6 SiCl4 PCl3 SCl2 B.P (°C ) 1470 1420 sublimes 60 75 60 M.P( °C ) 800 710 At 800°C −70 90 −80 - Give two chlorides that are liquid at room temperature. Give a reason for the answer (2 marks)
- Which two chlorides would remain in liquid state for the highest temperature range. Explain (2 marks).
- State one use of NaCl (1 mark).
- Copper (II) oxide reacts with zinc metal as shown below.
CuO(s) + Zn(s) → Cu(s) + ZnO(s)
Identify the substance that has been:- Reduced (1 mark)
- Oxidised (1 mark)
- An element B burns in air to form an oxide of B which dissolve in water to form a solution that turns blue litmus paper red. (B is not the actual symbol of the element).
Is B a metal or a non-metal? Give a reason for your answer (2 marks). - Study the diagram below and answer the questions that follow.
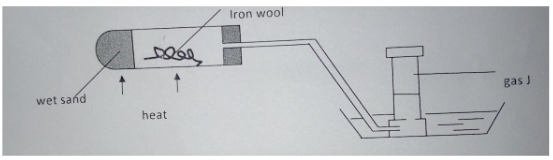
- Name gas J (1 mark).
- Explain why it is important to heat the wet sand before heating the iron wool (1 mark).
- State one observation made in the combustion tube as heating is carried out (1 mark).
- State two differences between permanent and temporary changes (2 marks).
- The table below give some properties of substances I, J and K. Study it and answer the questions that follow.
Substance Melting Point (°C) Solubility in water Electrical conductivity in: Solid state Molten state I 1063 Insoluble Conducts Conducts J 113 Insoluble Doesn't Doesn't K 402 Sparingly soluble. Doesn't Conducts and it is decomposed - Suggest the type of structure in:
- I (1 mark).
- K (1 mark).
- Explain why molten K is decomposed by current but I is not decomposed (1 mark).
- Suggest the type of structure in:
- Solution R,S and T have pH values shown in the table below.
Solution pH R 1.0 S 6.5 T 8.0 - What do you deduce about the nature of solution R? (1 mark).
- Identify two solutions that will react to form a neutral solution (1 mark).
- State and explain the changes in mass that occur when zinc metal is heated in an open crucible (2 marks).
- The pHof the soil sample was found to be 6.0. An agricultural officer recommended the addition of lime(calcium oxide). State two functions of lime in the soil (2 marks).
-
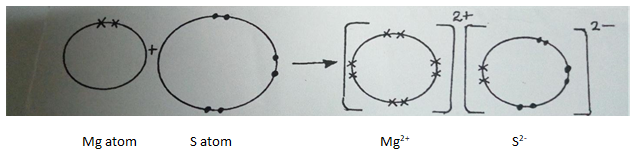
- Using a dot(.) and cross(x) to represent the outer most electrons, draw diagrams to show the bonding in magnesium sulphide (2 marks). (Mg=12,S=16).
- State the structure of the above compound (1 mark).
- Give two properties of substances with the above structure (2 marks).
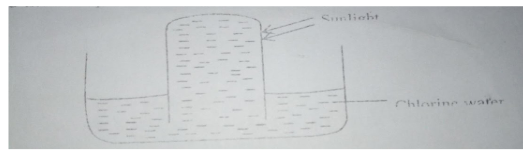
- In an experiment a test tube full of chlorine water was inverted in chlorine water as shown in the diagram and the set up left in the sunlight for one day.
After one day a gas was found to have collected in the test tube.- Identify the gas (1 mark).
- State the observations made when a blue litmus paper is dipped in chlorine water (1 mark).
- The elements shown in the table below (not actual symbols) belong to a certain family of metals in the periodic table. Study the information and answer the questions that follow.
Element Atomic size (nm) S 0.160 T 0.180 V 0.930 - Define the term ionization energy (1 mark).
- Which element is likely to have the highest ionization energy. Explain (2 marks).
- State two applications of solvent extraction (2 marks).
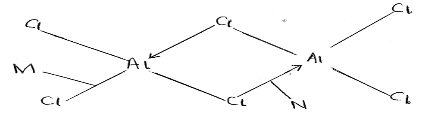
- The diagram below represents the structure of aluminum chloride.
- Identify the bonds labeled M and N (2 marks).
- What is the difference between bonds M and N (1 mark).
- From the following list of compound; Zinc oxide, Potassium carbonate, solid carbon (Iv) oxide, nitric acid, iron(III)chloride, sodium chloride.
- Identify two substances that sublime (1 mark).
- Identify two substances that react to form salt and water only (1 mark).
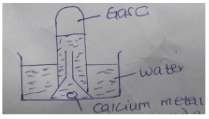
- The figure below is a set up used to investigate the reaction of calcium with water.
- State the observation made in the water (1 mark).
- Identify gas C (1 marks).
- State one laboratory application of the solution formed in the reaction (1 mark).
-
- State two types of salts (1 mark).
- Name the following processes;
- When anhydrous calcium chloride is left in an open beaker overnight a solution was formed (1 mark).
- When sodium carbonate decahydrate crystals are left in an open beaker for some days it turned into a powder (1 mark).
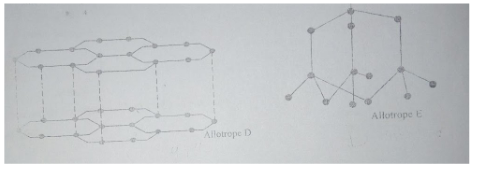
- The following diagrams show the structures of two allotropes of carbon. Study them and answer the questions that follow.
- Name the allotrope D and E(2 marks).
- Which allotrope does not conduct electricity? Explain (2 marks).
- Iron(III)chloride can be prepared in the laboratory by passing dry chlorine gas over hot steel wool.
- Name the above method of preparing salts (1 mark).
- Why should we prepare the salt in a dry environment? (1 mark)
- A solution of iron(III)chloride in water changes a blue litmus paper to red. Explain (1 mark).
- Metal S removes oxygen combined with P. Q reacts with an oxide of R but not with an oxide of P. P reacts with cold water but Q does not.
- Which is the most reactive metal? (1 mark).
- Which is the least reactive metal? (1 mark).
- Arrange the metals in order of reactivity starting with the most reactive to the least reactive (1 mark).
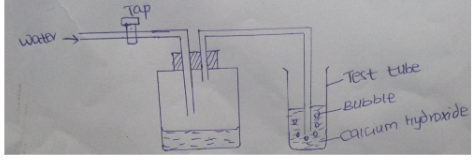
- The figure represents a set up of apparatus that was used to demonstrate the existence of a component of air.
- What is the purpose of water from the tap? (1mark).
- Other than bubbles, give one other observation made in the test tube (1 mark).
- Name a gas that was not absorbed by the calcium hydroxide. (1mk)
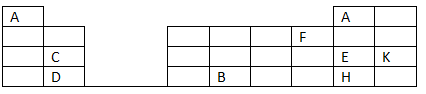
- The grid below is part of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements.
- Element A fits in two groups. Explain (2 marks).
-
- Name two elements that can form ions with a charge of -1. Explain your answer (2 marks).
- What type of structure would the oxide of B have? (1 mark).
- How does the reactivity of H compare with that of E? Explain. (2 marks)
- What name is given to the group of elements to which C and D belong? (1 mark).
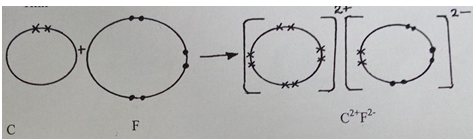
- Write the formula of the compound formed when elements C and F react (1 mark).
- Draw dot (.) and cross (x) diagram to represent the compound formed in (e) above (2 marks).
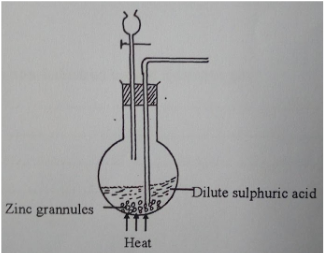
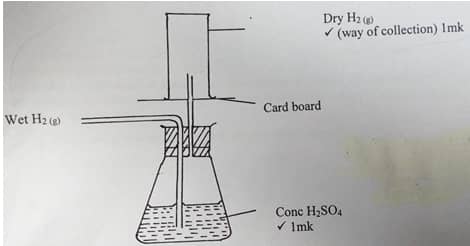
- A student set up the arrangement below to prepare and collect dry hydrogen gas.
- State the catalyst that is used in preparation of hydrogen gas (1 mark).
- Identify two errors from the section of the arrangement shown above (2 marks).
- Complete the diagram to show how dry hydrogen gas can be collected (2 marks).
- Explain the effect of hydrogen gas on a wet red litmus paper (1 mark).
- Write a balanced chemical equation for the reaction that takes place when hydrogen gas is burnt in air.
(1 mark). - State one property of hydrogen gas that makes it to be used in balloons (1 mark).
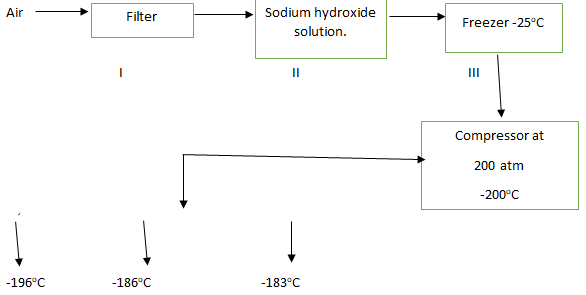
- Study the flow chart below and answer the questions that follow.
- Name the substances removed in steps I,II and III (3 marks).
- Name the gases obtained with respect to their boiling points (3 marks).
- -196°C
- -186°C
- -183°C

MARKING SCHEME
-
-
- A chimney (1MK)
- B Air hole
- Allows air to enter the chimney (1mk)
-
-
- Volumetric flask (2mks)
- Syringes
- Pipettes
- Burettes
-
- Use of a separating funnel (1MK)
-
- Different densities ((2MKS)
- Are immiscible
- Decantation/Use of dropper(1MK)
-
- No rusting occurred. Zinc is above iron in the reactivity series hence it reacts with moisture and air instead of iron
- Rusting occurred. Iron is more reactive than copper and hence Iron reacts with moisture and oxygen to form rust. (3mks)
- Add excess zinc oxide to dilute nitric (v) acid. Filter to get Zinc nitrate solution as filtrate and ZnO as residue. To the filtrate add sodium carbonate solution. Filter to obtain Zinc carbonate as residue. Wash with distilled water and dry between filter papers. (3mks)
-
- Potassium manganate (VII) (2mks)
- Sodium nitrate
- Potassium nitrate
-
- SiCl4 and SCl2 .Their melting points are below the room temperature.
- NaCl and MgCl2 (2mks)
There is a high difference between their M.P and B.P. -
- Used as a food additive.
- Brine (conc NaCl) is a source of hydrogen and chlorine.
- Manufacture of soapy and detergents
-
- CUO (1mk)
- Zn (1MK)
- Non-metal. Non-Metal oxides dissolve in water to form acidic solutions(2MKS)
-
- Hydrogen (1MK)
- To drive out air in the tube (1MK)
- Iron fillings glow red hot (1MK)
Black solid formed
-
Permanent Temporary (i) New substances are formed No new substances are formed (ii) The change is irreversible The change is reversible (iii)There is Change in mass Mass of substance does not change -
-
- I Giant metallic structure (1mk)
- K Giant Ionic structure(1MK)
- K is a compound while I is an element.(1MK)
-
-
- strongly acidic(1MK)
- R and T / S and T (1MK)
- The mass of zinc metal will increase. Zinc combines with the oxygen from the air to form zinc oxide. (2MKS)
-
- To raise the pH of the soil/Reduces acidity (2MKS)
- To add nutrients to the soil
-
- (2mks)
- Giant ionic structure (1MK)
-
- They do not conduct electricity in the solid state but conduct in molten state or aqueous solutions.(2MKS)
- Most of the substances dissolve in water.
- They have high melting and boiling points.
- (2mks)
-
- Oxygen gas (1mk)
- Turns red then white (1mk)
-
- Minimum energy required to remove an electron(s) from the outermost energy level of an atom in gaseous state (1mk)
- S.It has the smallest atomic size hence the outermostelectrons are strongly attracted to the nucleus hence more energy required to remove electron(s)
- Extraction of (2mks)
- Some herbal medicines from plants
- Natural dyes from plants
- Caffeine from tea and coffee
- Oil from nuts and seeds
- In dry cleaning to remove dirt.
-
-
- M covalent bond (2mks)
- N Dative/co-ordinate
- Covalent bond involves equal sharing ofelectrons donated by both species. In dative the shared pair is donated by one atom.
-
-
- Solid carbon (IV) oxide and iron (III) chloride (1mk)
- Zinc oxide and nitric(V) acid (1mk)
-
- White suspension (1mk)
- Hydrogen gas (1mk)
- To test for the presence of carbon (IV) oxide (1mk)
-
-
- Normal salts (1mk)
- Acid salts
- Basic salts
- Double salts
-
- Deliquescence
- Efflorescence
-
-
-
- D Graphite(2mks)
- E Diamond
- E. Has no delocalized electrons since it uses all its valence electrons in bonding (2mks)
-
-
- direct synthesis (1mk)
- Iron(III) Chloride reacts with water vapour (1mk)
- Iron (III) chloride is hydrolysed by water to form hydrochloric acid
-
- S (1mk)
- R (1mk)
- S,P,Q,R (1mk)
-
- To drive out air andbubble it through the calcium hydroxide solution (1mk)
- White precipitate is formed (1mk)
- Oxygen, Nitrogen and noble gases (1mk)
-
- A fits in group I because it has one electron in the outermost energy level. It also fits in group (VII) because it requires one electron like group (VII) elements to be stable (2mks)
-
- A,E and H (2mks)
- They gain one electron to be stable
- Giant atomic structure (1mk)
- A,E and H (2mks)
- E is more reactive than H (2mk)
- They both react by gain of an electron .However E has few energy levels making the nuclear pull stronger than in H
- Alkaline earth metals (1mk)
- CF (1mk)
-
-
- Copper (II) Sulphate crystals (1mk)
- The outlet delivery tube should not be dipped into the zinc/ dilute sulphuric acid mixture in the round bottomed flask.
Use of heat is not required (2mk) -
- Has no effect since hydrogen gas is neutral (1mk)
- 2H2(g) + O2(g) → 2H2O(g) (1mk)
- Lighter than air (1mk)
-
-
- I dust (3mks)
- II Carbon (IV) Oxide
- III Water
-
- -196°C Nitrogen gas
- -186°C Argon
- -183°C Oxygen (3mks)
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Questions and Answers - Form 2 Term 3 Opener Exams 2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students