- Name another gas which is used with oxygen in welding [1 Mk]
-
- write the electronic configuration of calcium (atomic number 20) and magnesium (atomic number 12)
Calcium [½ Mk]
Magnesium [½ Mk] - Why is calcium more reactive than magnesium? [2 Mks]
- write the electronic configuration of calcium (atomic number 20) and magnesium (atomic number 12)
- The table below shows the relative atomic masses and the percentage abundance of the isotopes T1 and T2 of element T
Calculate the relative atomic mass of element T [3 mks]RAM % abundance T1 62.93 69.09 T2 64.93 30.91 - The diagram below is a set-up for the laboratory preparation of oxygen gas.
- Name solid P. [1 mk]
- Write an equation for the reaction that takes place in the conical flask [1 mk]
- Give two commercial uses of oxygen [2 mks]
- State two reasons why hydrogen is not commonly used as a fuel [2 mks]
- The figure shows a set-up by a form three student to prepare a certain gas
- Write an equation for the formation of gas K [1 mk]
- Give one use of gas K in the industries [1 mk]
- Give one use of the resulting solution after the metal has reacted [1 mk]
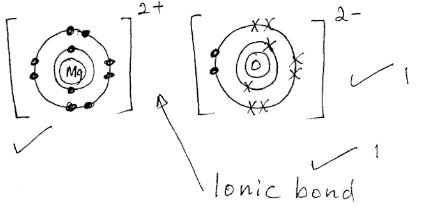
- Draw a dot and cross diagram showing the bonding in a molecule of calcium oxide. Name the type of bond. [3 mks]
- When 0.288g of an oxide of metal M was reduced using suitable reducing agent, 0.256 of pure metal was formed. Determine the empirical formula of the oxide of the metal M. [M=64 O=16] [4 mks]
- X+ is an ion with electronic configuration 2,8,8. Identify element X [1 mk]
- 20g of solid sodium hydroxide were dissolved in distilled water and made to 400cm3. 30cm3 of this solution required 27cm3 of dilute sulphuric (iv) acid for complete reaction. [Na=23 O=16 H=1]
Determine- Moles of sodium hydroxide contained in 30cm3 of solution [2 mks]
- Moles of sulphuric (iv) acid that reacted [2 mks]
- Concentration of the sulphuric (iv) acid in moles per litre [2 mks]
- The diagram shows the structures of two allotropes of carbon. Study them and answer the questions that follow.
- Name allotrope A and B [2 mks]
- Give two uses of allotrope B [2 mks]
- Which allotrope conducts electricity? Explain. [2 mks]
- An oxide of element F has the formulaF2O5
- Determine the oxidation state of F. [1 mk]
- In which group of the periodic table is element F? [1 mk]
- Explain how you would obtain solid sodium carbonate from a mixture of lead II carbonate and sodium carbonate. [3 mks]
- Give two properties of aluminum that makes it very suitable for making cooking utensils [2 mks]
- Write down an ionic equation for the reaction between dilute hydrochloric acid and calcium carbonate [3 mks]
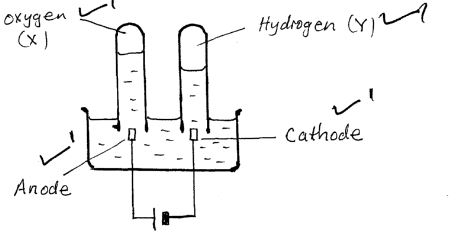
- The diagram shows electric current passing through dilute sulphuric (iv)acid
- On the diagram identify the cathode and the anode [2 mks]
- Identify substances X and Y [2 mks]
- State and explain the change in mass that occur when following substances are separately heated in open crucibles [4 mks]
- Copper metal
- Copper II nitrate
- The diagram below represents a paper chromatograph for three brands of juices suspected to contain banned food colourings
The result showed presence of banned food colourings in L and M only- On the diagram
- Circle the spots which show the banned colourings [2 mks]
- Show the solvent front [1 mk]
- On the same diagram indicate and label the baseline [1 mk]
- On the diagram
- Determine the number of sodium ions contained in 25cm3 of 0.5M sodium carbonate solution [a=6.023 x 1023] [3 mks]
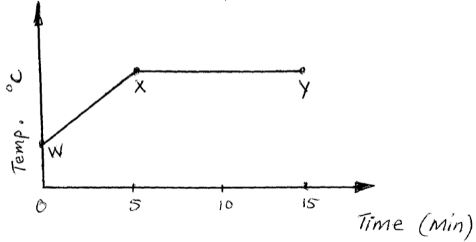
- The graph below shows a curve obtained when water at 20℃ was heated for 15 mins.
- What happens to the water molecules between points W and X [1 mk]
- In which part of the curve does a change of state occur? [1 mk]
- Explain why the temperature does not rise between points X and Y [1 mk]
- Write down the formula of the following compounds
- Potassium manganate VII [1mk]
- Aluminium oxide [1mk]
- Iron III chloride [1mk]
- Write balanced equations for the following reactions
- Reaction between sodium and excess oxygen [1mk]
- Reaction between Nitric Acid and Copper Carbonate [1mk]
- Reaction between Zinc and hydrochloric acid [1mk]
- Heated Silver nitrate reaction [1mk]
- The diagram shows PH values for several substances.
Choose the likely PH value for,- Dilute Hydrochloric acid [1mk]
- Calcium hydroxide [1mk]
- Sodium hydroxide [1mk]
- Lemon juice [1mk]
- Briefly outline how you would obtain ethanol from a mixture of ethanol and water. [3mks]
-
- What is rust? [1mk]
- Give two advantages of rusting.

Marking Scheme
- Name another gas which is used with oxygen in welding [1 Mk]
- Hydrogen/Acetylene or Ethyne
-
- write the electronic configuration of calcium (atomic number 20) and magnesium (atomic number 12)
Calcium [½ Mk]
2, 8, 8, 2
Magnesium [½ Mk]
2, 8, 2 - Why is calcium more reactive than magnesium? [2 Mks]
- The two valency electrons of calcium are further away from the nucleus, therefore not strongly held hence readily lost/donated
- write the electronic configuration of calcium (atomic number 20) and magnesium (atomic number 12)
- The table below shows the relative atomic masses and the percentage abundance of the isotopes T1 and T2 of element T
Calculate the relative atomic mass of element T [3 mks]RAM % abundance T1 62.93 69.09 T2 64.93 30.91
R.A.M = 62.93 × 69.09 + 64.93 × 30.91
100 100
= 4347.8337 + 2006.9863
100
= 6354.82
100
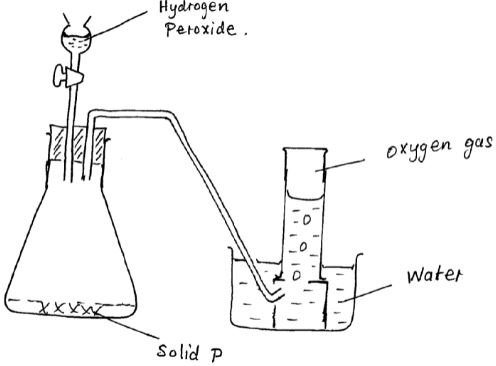
= 63.55 - The diagram below is a set-up for the laboratory preparation of oxygen gas.
- Name solid P. [1 mk]
- Manganese IV Oxide
- Write an equation for the reaction that takes place in the conical flask [1 mk]
MNO2
2H2O2(l)2H2O(l) +O2(g) // 2H2O2(l) → 2H2O(l) +O2(g)
- Give two commercial uses of oxygen [2 mks]
- Welding
- Rocket fuel
- Hospital, ICU
- Steel making
- Deep sea diving
- Name solid P. [1 mk]
- State two reasons why hydrogen is not commonly used as a fuel [2 mks]
- Expensive to produce
- Explosive
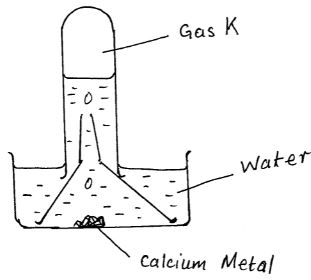
- The figure shows a set-up by a form three student to prepare a certain gas
- Write an equation for the formation of gas K [1 mk]
Ca(s) +2H2O(l) → Ca(OH)2(aq) + H2(g) - Give one use of gas K in the industries [1 mk]
- Welding
- Manufacture of HCL acid
- Hardening of vegetable oil into margarine
- Manufacture of Ammonia gas
- Give one use of the resulting solution after the metal has reacted [1 mk]
- Test for Carbon IV oxide
- Write an equation for the formation of gas K [1 mk]
- Draw a dot and cross diagram showing the bonding in a molecule of calcium oxide. Name the type of bond. [3 mks]
- When 0.288g of an oxide of metal M was reduced using suitable reducing agent, 0.256 of pure metal was formed. Determine the empirical formula of the oxide of the metal M. [M=64 O=16] [4 mks]
Element M O
Mass 0.256 0.032
RAM 64 16
Moles 0.256 0.032
64 16
0.004 0.002
Ratio of 0.004 0.002
moles 0.002 0.002
2 1
Empirical Formula = M2O - X+ is an ion with electronic configuration 2,8,8. Identify element X [1 mk]
- Potassium
- 20g of solid sodium hydroxide were dissolved in distilled water and made to 400cm3. 30cm3 of this solution required 27cm3 of dilute sulphuric (iv) acid for complete reaction. [Na=23 O=16 H=1]
Determine- Moles of sodium hydroxide contained in 30cm3 of solution [2 mks]
Molar mass of NaOH= 40g
20/40 = 0.5M
0.5 = 400cm3
? = 1000cm3
0.5 × 1000
400
=1.25M
1.25 = 1000cm3
? =30cm3
1.25 × 30
1000
=0.0375moles - Moles of sulphuric (iv) acid that reacted [2 mks]
2NaOH(aq) + H2SO4(aq) → Na2SO4(aq) + 2H2O(l)
Mol of H2SO4 = Mol NaOH Mole ration in equation above is 1:2
2
=0.0375
2
Mol of H2SO4 =0.01875moles - Concentration of the sulphuric (iv) acid in moles per litre [2 mks]
0.01875 = 27cm3
? =1000cm3
1000 × 0.01875
27
=0.6944M
- Moles of sodium hydroxide contained in 30cm3 of solution [2 mks]
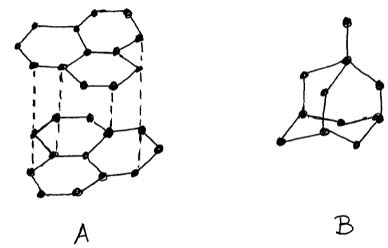
- The diagram shows the structures of two allotropes of carbon. Study them and answer the questions that follow.
- Name allotrope A and B [2 mks]
- A-Graphite
- B-Diamond
- Give two uses of allotrope B [2 mks]
- Tips of drills
- Jewellery
- Glass cutter
- Making bearing
- Padlocks
- Which allotrope conducts electricity? Explain. [2 mks]
- A/Graphite-The fourth electron of each carbon is unbounded hence free to conduct electricity
- Name allotrope A and B [2 mks]
- An oxide of element F has the formula F2O5
- Determine the oxidation state of F. [1 mk]
F2O5 = O
2F + 5(−2) = 0
2F+−10=0
2F = 0+10
2 2
F= +5 - In which group of the periodic table is element F? [1 mk]
- Group V
- Determine the oxidation state of F. [1 mk]
- Explain how you would obtain solid sodium carbonate from a mixture of lead II carbonate and sodium carbonate. [3 mks]
- Put the mixture in water and stir. Sodium carbonate dissolves while lead carbonate does not. Filter to remove lead carbonate as a residue. Evaporate the filtrate to saturation and allow to cool for crystallization of sodium carbonate to take place
- Give two properties of aluminum that makes it very suitable for making cooking utensils [2 mks]
- Good thermal conductivity.
- malleability.
- light weight
- high melting point
- Write down an ionic equation for the reaction between dilute hydrochloric acid and calcium carbonate [3 mks]
2H+Cl−(aq)+ Ca2+CO2−3(s) → Ca2+Cl−2(aq) + CO2(g) +H20(l)
2H+(aq) + CO2−3(s) → CO2(g) +H2O(l) - The diagram shows electric current passing through dilute sulphuric (iv)acid
- On the diagram identify the cathode and the anode [2 mks]
- Identify substances X and Y [2 mks]
- State and explain the change in mass that occur when following substances are separately heated in open crucibles [4 mks]
- Copper metal
- Mass increase-Copper metals combines with oxygen forming copper oxide
- Copper II nitrate
- Mass reduction-Copper II Nitrate is decomposed by heat forming copper oxide, Nitrogen IV Oxide and oxygen gases, the gases escape.
- Copper metal
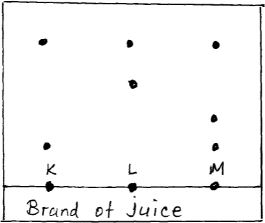
- The diagram below represents a paper chromatograph for three brands of juices suspected to contain banned food colourings
The result showed presence of banned food colourings in L and M only- On the diagram
- Circle the spots which show the banned colourings [2 mks]
- Show the solvent front [1 mk]
- On the same diagram indicate and label the baseline [1 mk]
- On the diagram
- Determine the number of sodium ions contained in 25cm3 of 0.5M sodium carbonate solution [a=6.023 x 1023] [3 mks]
Moles of Na2CO3 = 25 × 0.5
1000
=0.0125
Na2CO3(aq) → 2Na+(aq) + CO2−3(aq)
Moles of Na+ = 2 x 0.0125
= 0.025
Number of Na+ = 0.025 x 6.023 x 1023
= 1.5058 x 1022 - The graph below shows a curve obtained when water at 20℃ was heated for 15 mins.
- What happens to the water molecules between points W and X [1 mk]
- Water molecules gain energy and move faster
- In which part of the curve does a change of state occur? [1 mk]
- XY
- Explain why the temperature does not rise between points X and Y [1 mk]
- Supplied energy is used to break bonds between water molecules in liquid state and become free molecules in gaseous state
- What happens to the water molecules between points W and X [1 mk]
- Write down the formula of the following compounds
- Potassium manganate VII [1mk]
KMnO4 - Aluminium oxide [1mk]
Al2O3 - Iron III chloride [1mk]
FeCl3
- Potassium manganate VII [1mk]
- Write balanced equations for the following reactions
- Reaction between sodium and excess oxygen [1mk]
2Na(s) + O2(g) → Na2O2(g) - Reaction between Nitric Acid and Copper Carbonate [1mk]
2HNO3(aq) + CuCO3(s) → CuNO3(aq) +CO2(g) + H2O(l) - Reaction between Zinc and hydrochloric acid [1mk]
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g) - Heated Silver nitrate reaction [1mk]
2AgNO3(s)2Ag(s) + 2NO2(g) + O2(g)
- Reaction between sodium and excess oxygen [1mk]
- The diagram shows PH values for several substances.
Choose the likely PH value for,- Dilute Hydrochloric acid [1mk]
- 2
- Calcium hydroxide [1mk]
- 9
- Sodium hydroxide [1mk]
- 13
- Lemon juice [1mk]
- 5
- Dilute Hydrochloric acid [1mk]
- Briefly outline how you would obtain ethanol from a mixture of ethanol and water. [3mks]
- Heat the mixture in a fractional distillation apparatus maintaining the temperature at the boiling point of ethanol which is lower than that of water. Ethanol will distill over, leaving water behind.
-
- What is rust? [1mk]
- Hydrated Iron III oxide
- Give two advantages of rusting.
- Enable paint to stick properly on steel articles
- Assist the environment to get rid of scrap metal objects.
- What is rust? [1mk]
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - Form 3 Mid Term 1 Exams 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students