QUESTIONS
- The curve below shows variation of temperature with time when pure and impure samples of a solid were heated separately.
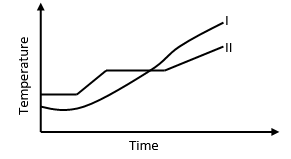
Explain the variation in temperature in curves I and II -
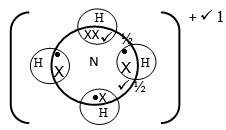
- Use (x) cross and dot (•) diagram to show the structure of ammonium ion (NH4+) (2 Marks)
- Name the bonds found in (NH4+) ion (1 Mark)
- A student passed ammonia gas through concentrated sulphuric (VI) acid to dry the gas and tried to collect the gas.
- State the problem he encountered and the reason why. (2 Marks)
- Write the equation for the reaction above. (1 Mark)
- Name the appropriate drying agent for ammonia gas. (1 Mark)
- A piece of chromatography paper was spotted with coloured inks obtained from pens labelled A to F. The diagram below shows the spots as they were developed.
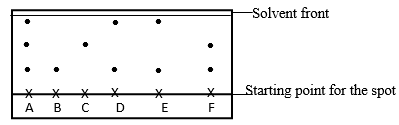
- Which two pens contained the same pigment? (1 Mark)
- Which pens contained only one pigment (1 Mark)
- According to the chromatogram, which pigments are present in the ink of pen No. F (1 Mark)
- A student found a colourless gas in a fume chamber and wanted to find out if it is was Hydrogen Chloride. He placed a drop of Silver Nitrate on a glass rod and placed it on the beaker as shown.
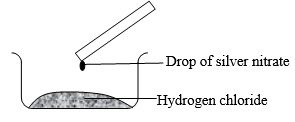
- State the observation made. (1 Mark)
- Write ionic equation for the reaction above (1 Mark)
-
- What are isotopes? (1 Mark)
- Determine the number of neutrons in
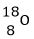 (1 Mark)
(1 Mark)
- A soil was found to have a PH of 5.0. An agricultural officer recommended the addition of Calcium Oxide to the soil. State two functions of Calcium Oxide in the soil (2 Marks)
- Name the compound below. (1 Mark)
- CH3CH2CH = CH2
- Draw and name other isomers of the compound above. (2 Marks)
- When magnesium is burnt in air it reacts with both oxygen and nitrogen gas giving a white ash. Write two equations for the reactions that take place. (2 Marks)
- In terms of structure and bonding explain the following.
- Melting point of aluminium is higher than that of sodium. (1 ½ Mark)
- Melting point of chlorine is lower than that of iodine. (1 ½ Mark)
- A compound contains 82.75% carbon and the rest is Hydrogen.
- Determine its empirical formula. (2 Marks)
- Determine the molecular formula if its molecular mass is 58. (1 Mark)
- Elements A, B and C have atomic numbers 9, 11 and 18 respectively.
- Which element can be used in electric light bulbs? (1mark)
- Which two elements react to form an ionic compound? (1 Mark)
- Write an equation for the reaction between element B and water? (1mark)
- A student heated copper (II) Nitrate as shown below.
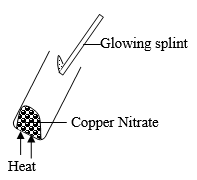
- State two observations made (2 Marks)
- Write an equation for the reaction taking place. (1 Mark)
-
- A form three student was given two gases C2H6 and C2H4. He added acidified Manganate (VII) to each solution. What observation was made? (2 Marks)
- State one use of C2H4 (1 Mark)
- A student carried out an experiment to determine the value of x in hydrated barium chloride. (BaCl2.xH2O). The following results were obtained.
Mass of crucible = 36.12g
Mass of crucible + barium chloride before heating = 41.00g
Mass of crucible + barium chloride after 1st heating = 40.42g
Mass of crucible + barium chloride after 2nd heating = 40.30g
Mass of crucible + barium chloride after 3rd heating = 40.30g- What mass of anhydrous barium chloride was formed in this experiment? (1 Mark)
- How much water of crystallization did the sample contain? (1 Mark)
- Calculate the mass of crystallization that could be obtained from one mole of hydrated barium
Chloride. (BaCl2 = 208; H2O = 18) (1 Mark)
-
- Explain why hot iron lowered in a gas jar containing chlorine continues to glow for some time. (1 Mark)
- Write the equation for the reaction above. (1mark)
-
- Candle wax mainly consists of two elements. Name the two elements (2 Marks)
- Name the products of burning candle wax in air. (1 Mark)
- Explain why anhydrous aluminium chloride is fairly soluble in organic solvent while anhydrous magnesium chloride is not soluble. (2 Marks)
-
- What are allotropes? (1 Mark)
- Name two allotropes of sulphur (2 Marks)
- Sodium carbonate reacts with dilute hydrochloric acid to liberate carbon (IV) oxide gas. Calculate the volume of carbon (IV) oxide measured at s.t.p which is evolved when 5.3g of sodium carbonate completely reacts with dil. Hydrochloric acid. (C = 12, O = 16, Na = 23)
(Molar gas volume at s.t.p = 22.4dm3) (3 Marks) - Write ionic equations for the following reactions.
- Ba(NO3)2(aq) + Na2CO3(aq) → BaCO3(s) + 2NaNO3(aq)
- CuSO4(aq) + Fe(s) → Cu(s) + FeSO4(aq)
- Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
- The diagram below represents a set up used to prepare and collect Nitric (V) acid in the laboratory.
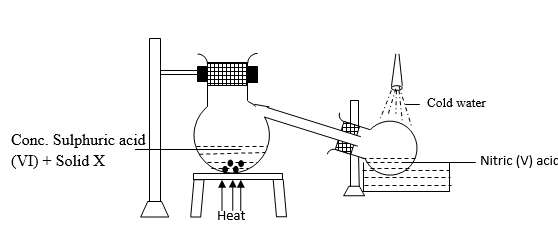
- Identify solid X (1 Mark)
- Write the equation for the reaction above (1 Mark)
- What is the appearance of the nitric acid prepared by this method and why? (1 Mark)
- Molten lead bromide was electrolysed using carbon electrodes.
- Name the products at the anode and cathode
- Anode (1 Mark)
- Cathode (1 Mark)
- State two applications of electrolysis (1 Mark)
- Name the products at the anode and cathode
- Nitrogen (I) oxide was passed over heated copper turnings as shown in the diagram below.
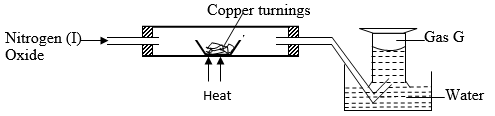
- State the observation made in the combustion tube. (1 Mark)
- Name gas G (1 Mark)
- Write the equation for the reaction at the combustion tube. (1 Mark)
- Give two reasons why hydrogen is not commonly used as a fuel. (2 Marks)
- When magnesium powder and carbon (II) oxide are heated in a crucible.
- State the observations made. (1 Mark)
- Write an equation for the reaction above. (1 Mark)
- A gas Y occupied a volume of 200cm3 at 20ºC and 1 atmosphere. Calculate the volume it will occupy at 40ºC and 2 atmospheres. (3 Marks)
- Study the table below and answer the questions that follow:-
Ion
Electron arrangement
X3+
2,8
Y2-
2,8
W+
2,8
- Write the electron arrangement of atoms of element X and Y. (2 Marks
X ________________
Y ________________ - Write the formula of the compound formed between Y and W. (1 Mark)
- In what state would you expect the compound above to conduct electricity? (1 Mark)
- Write the electron arrangement of atoms of element X and Y. (2 Marks
- Differentiate between the following apparatus.
- Pipette and a measuring cylinder. (1 Mark)
- Round bottomed flask and flat bottomed flask. (1 Mark)
-
- Distinguish between metallic and ionic bonding. (1 Mark)
- Draw a diagram to show bonding in sodium metal. (1 Mark)
MARKING SCHEME
-
- Is impure substance: melting point is lower ½ than pure substance where as boiling point is higher than ½ of pure substance.
- Pure substance ½; Both melting and boiling points are constant. ½
-
-
-
- Covalent bond ½
- Co-ordinate bond ½
-
-
-
- He did not collect any of the ammonia gas. 1
- Ammonia gas reacts with concentrated sulphuric acid 1
- 2NH3(g) + H2SO4(l) → (NH4)2SO4(aq) 1
- Anhydrous calcium oxide ½
-
-
- D and E 1
- B and C 1
- Pigment A, B, C, D and E 1
All other pigments other than F. 1
-
- A white solid of silver chloride was seen at the tip of the glass rod where the drop was. 1
- HCl(g) + AgNO3 → AgCl(s) + HNO3
Ag+(aq) + Cl-(aq) → AgCl(s) 1
-
- Isotopes; Are atoms of the same element with same number of protons but different numbers of neutrons. 1
- No. of neutrons.
18 – 8 = 10 ½
-
-
- Raises the PH of the soil. 1
- Lowers the acidity of the soil.
- Adds nutrients to the soil. 1
-
-
- But-1-ene 1
-
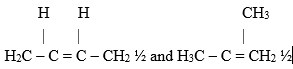
But – 2 – ene ½ 2-methylprop – 2 – ene ½
-
- 3Mg(s) + N2(g) → Mg3N2(s) 1
- 2Mg(s) + O2(g) → 2MgO(s) 1
-
- Aluminium has more ½ delocalised electrons in its structure than sodium which has one per atom ½ forming a stronger metallic bond ½.
- Iodine has a stronger molecules due to strength of van-der – waals forces ½ increasing down ½ the group compared to chlorine with weaker ½ van – der – waals forces.
-
- Empirical formula.
Elements : Carbon : Hydrogen
C : H
% Mass 82.75 : 17.25 ½
R.A.M 12 1
Moles 6.89 : 17.5 ½
Mole ratio 6.89 : 17.3
6.89 6.89
(1: 2.5)2 ½
E.F C2H5 ½ - M.F = (E.F) n
n = (M.F)/(F.M) ½ = 5.8/2.9 = 2
M.F. = C4H10 ½
- Empirical formula.
-
- Element C 1
- A and B 1
- 2B(s) + 2H2O(l) → 2BOH(aq) + H2(g) 1
-
-
- Brown fumes of a gas (NO2) was seen 1
- The glowing splint was relighted. 1
heat
- 2Cu(NO3)2 → 2CuO(s) + 4NO2(g) + O2(g)
-
-
- The gas C2H4 decolourised potassium 1 manganate (VII) where as C2H6 did not 1, but it remained purple in it.
-
- Making polythene bags. 1
- Ripening of fruits.
-
- Mass of anhydrous barium chloride formed = (40.30 – 36.12)g ½ = 4.18g ½
- Mass of water of crystallization = (41.00 – 40.30)g ½ = 0.70g ½
- Mass of water of crystallization contained in one mole of hydrated barium chloride = (20.8 x 0.7) g ½
4.18
= 34.8g ½
-
- Because the reaction is exothermic 1
- 3Cl2(g) + 2Fe(s) 2FeCl3(s) 1
-
- Carbon 1
Hydrogen 1 - Carbon (IV) Oxide ½
Water ½
- Carbon 1
- Aluminium chloride is made up of molecules ½ thus makes it soluble in organic solvents ½
Magnesium chloride is made up of ions ½ which makes it insoluble in organic solvents. ½ -
- Allotropes are different forms of the same element but in the same physical state 1
- Monoliclinic sulphur 1
Rhombic sulphur 1
- Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + CO2(g) + H2O(l)
5.3g
No. of moles of Na2CO3 = 5.3/106 = 0.05 moles ½
Mole ratio Na2CO3 : CO2
1 : 1 ½
Moles of CO2 produced is 0.05 ½
1 mole of CO2 occupies – 22.4dm3 ½
0.05 moles - ?
= 0.05 x 22.4
= 1.12dm3 of CO2 ½ -
- Ba2+(aq) + CO32-(aq) → BaCO3(s) 1
- Cu2+(aq) + Fe(s) → Cu(s) + Fe2+(aq) 1
- Zn(s) + 2H+(aq) → Zn2+(aq) + H2(g) 1
-
- X – Potassium Nitrate 1
- H2SO4 + KNO3(aq) → KHSO4(aq) + HNO3(aq) 1
- It is yellow because it has dissolved NO2 gas. 1
-
-
- Anode (+) – Bromine gas is produced. 1
- Cathode (-) – Lead metal is deposited.
-
- Electroplating
- Purification of metals
- Extraction of metals
(Any one) 1
-
-
- Brown copper metal changed to black copper (II) oxide.
- Gas G – Nitrogen gas 1
- N2O(g) + Cu(s) → CuO(s) + N2(g) 1
-
- Hydrogen gas when ignited in oxygen is explosive. 1
- Hydrogen gas is not commonly/readily available.
-
- A white solid ½ of magnesium oxide and ½ black specks of carbon are seen.
- CO2(g) + 2Mg(s) → 2MgO(s) + C(s) 1
-

V1 = 200cm3 V2 = ?
T1 = 293k ½ T2 = 313k ½ -
-
- X : 2,8,3 1
Y: 2,6 1
NB: Use commas not dots.
- X : 2,8,3 1
- W2Y 1
- In molten or aqueous state. 1
-
-
-
- Pipette for measuring and transferring fixed volumes. ½
- Measuring cylinder to measure approximate volumes of liquids ½
-
- Round-bottomed: Used where heating is required. ½
- Flat-bottomed flask used where no heating is required. ½
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - Form 3 End Term 3 2022 Exams.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

