QUESTIONS
-
- The grid below represents part of the periodic table .Study it and answer the questions that follow .The letters
given do not represent the actual symbols of the elements.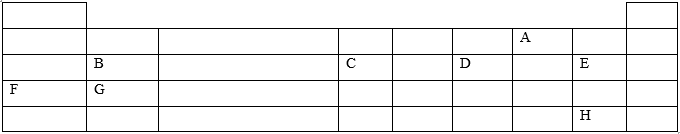
- Select the element that can form an ion with a charge of -2 .Explain your answer. (2 marks)
- What type of structure would the oxide of C have? Explain your answer. (1 mark)
- How does the reactivity of H compare with that of E? Explain your answer. (1 mark)
- 2.6 g of B reacts completely when heated with 2.42 litres of chlorine gas (Cl2) at s.t.p.
(1 mole of gas occupies 22.4 litres at s.t.p.)- Write a balanced equation for the reaction between B and chlorine gas. (1 mark)
- Determine the relative atomic mass of B. (2marks)
- Explain how you would expect the atomic radii of F and G to compare. (2 Marks)
- The table below shows physical properties of some substances .Use the information on the table to answer the questions that follow.
Substance
Melting point (0c)
Boiling point(0C)
Electrical conductivity
Solid
Liquid
U
1083
2595
Good
Good
V
801
1413
Poor
Good
W
5.5
80.1
Poor
Poor
X
-114.8
-84.9
Poor
Poor
Y
3550
4827
Poor
Poor
- Which substance is likely to be (with a reason)
- a metal. (1mark)
- a liquid at room temperature? (1 mark)
- Which substance is likely to have the following structures.
- Simple molecular (1 mark)
- Giant atomic (1 Mark)
- Which substance is likely to be (with a reason)
- The grid below represents part of the periodic table .Study it and answer the questions that follow .The letters
-
- The following diagram shows an incomplete set up that can be used to prepare and collect dry oxygen gas in the laboratory. Study it and answer the questions that follow
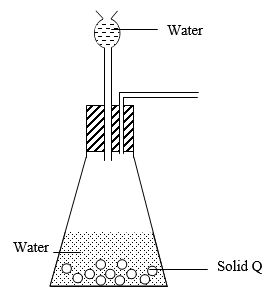
- Correct the mistakes in the set up (1 mark)
- Complete the diagram to show how a dry sample of the gas may be collected. (2 marks)
- Identify solid Q and give its formula (1 mark)
- Write an equation that produces oxygen gas in this experiment. (1 mark)
- Name another pair of chemicals that can be used to produce oxygen in this set up.
- A small amount of burning sulphur was lowered in a gas jar of oxygen.
- State one observation made. (1 mark)
- Write an equation for the reaction taking place. (1 mark)
- State the approximate pH of the solution when the product in b(ii) above is dissolved in water. Explain. (2 marks)
- The following diagram shows an incomplete set up that can be used to prepare and collect dry oxygen gas in the laboratory. Study it and answer the questions that follow
-
- In an experiment 10.6g of a mixture of a anhydrous Sodium Carbonate and Sodium chloride were dissolved in water to make 100cm3 of solution .25cm3 of this solution required 20cm3 of 1M Hydrochloric acid solution for complete neutralization.
- Calculate the number of moles of Hydrochloric acid used (1 mark)
- Which substance reacts with the Hydrochloric acid in this mixture? (1 Mark)
- Write an equation for the reaction that occurs. (1 Mark)
- Calculate the mass of Sodium Carbonate in 25 cm3n of this mixture. (1 Mark)
- Determine the moles of Sodium Carbonate in 100cm3 of the mixture. (1 Mark)
- What is the mass of sodium carbonate in the mixture? (2 Marks)
- Calculate the percentage of Sodium Chloride in the mixture. (2 Marks)
- A gaseous compound P contains 55% Carbon, 9.1 % Hydrogen and the rest is Oxygen.
- Determine the empirical formula of P.(C=12, H=1, O=16) (2 Marks)
- Given that 0.262g of P occupies a volume of 67cm3 at s.t.p
- Calculate the relative formula mass of P. (1 Mark)
- Determine the molecular formula of P. (1 Mark)
(Molar gas volume at s.t.p.=22.4 dm3)
- In an experiment 10.6g of a mixture of a anhydrous Sodium Carbonate and Sodium chloride were dissolved in water to make 100cm3 of solution .25cm3 of this solution required 20cm3 of 1M Hydrochloric acid solution for complete neutralization.
- The set up below was used to prepare hydrogen gas
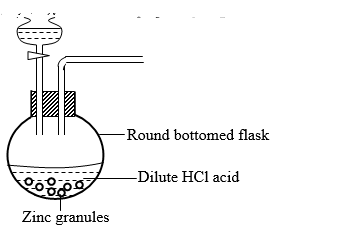
- Complete the diagram to show how dry sample of hydrogen can be collected. (2 Marks)
- Write an experiment for the reaction which takes place
- In the round bottomed flask. (1 Mark)
- When hydrogen gas burns in air. (1 Mark)
- State two industrial uses of hydrogen gas. (2 Marks)
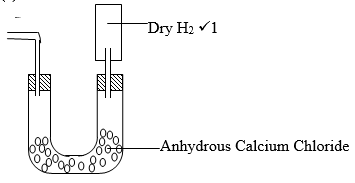
- Dry hydrogen gas passed over heated Copper (ii) oxide in the set up below.
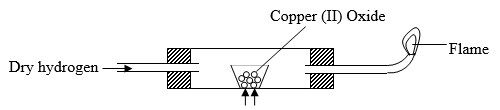
- State two observations made in the experiment. (2 Marks)
- What property of Hydrogen was being investigated. (1 Mark)
- Write an equation for the reaction which proves the property in (ii). (1 Mark)
- Why should hydrogen be passed for sometime through the combustion tube before being lit. (1 Mark )
- The flow diagram below shows a reaction scheme starting from propene.
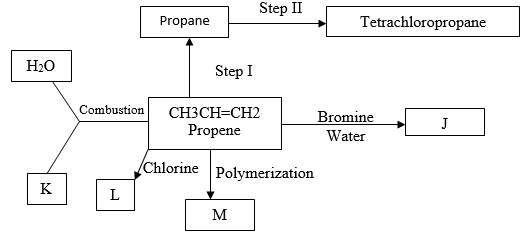
- Draw the structural formula of propene (1 Mark)
- State the reagent and condition necessary for step1 to occur
Reagent: (1 Mark)
Condition: (1 Mark) - Give the names of the following substances.
J-.............
K -............
L-.............
M-.............(2 Marks) - Write the equation for the reaction that produces substance L. (1 Mark)
- State the type of reaction that occurs in step I. (1 Mark)
- Write the equation for complete combustion of propene. (1 Mark)
-
- Give one use of substances M. (1 Mark)
- State the environmental effects of the continued use of plastics to the environment .Explain. (2 Marks)
-
- State one form of amorphous carbon other than charcoal and state its use. (2 Marks)
- The diagram below represents a ‘jiko’ when in use .Study it and answer the questions that follow.
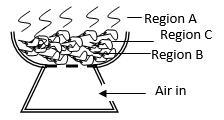
- Write equations for the reactions that occur in region
- B (1 mk)
- C (1 mk)
- Explain what happens in region A. (1 mk)
- Write equations for the reactions that occur in region
- The following flow chart represents the Solvay process used in the manufacture of sodium carbonate. Study it and answer the questions that follow.
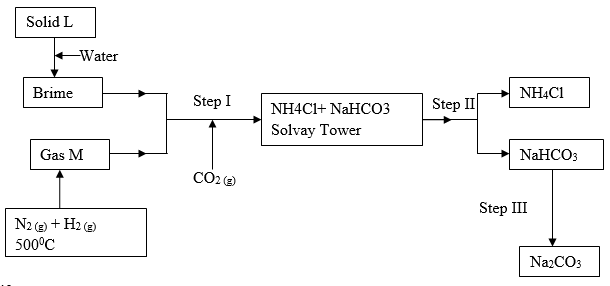
- Identify
- Solid L (½ mk)
- Gas M (½ mk)
- Name the processes that take place in steps
- (½ mk)
- (½ mk)
- Give the name of catalyst R. (1 Mark)
- Write an equation for the reaction that occur in steps
- (1 Mark)
- (1 Mark)
- Describe how carbon (IV) oxide for this process is obtained. (1 mk)
- State one commercial use of sodium carbonate (1 mk)
- Identify
-
-
- Name two reagents to prepare Hydrogen chloride in the laboratory. (1 Mark)
- State a suitable drying agent for hydrogen chloride gas (1 mk)
- Write an equation for the reaction that produces Hydrogen Chloride using the reagents named in a (i) above.(1 Mark)
- Describe a test for Hydrogen Chloride gas. (1 mk)
- Dry hydrogen gas was reacted with iron fillings using the set up below:-
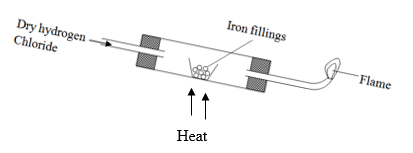
- Write occur in the combustion tube. (1 mk)
- Write an equation at the point of the flame. (1 Mark)
- 1.96 g of iron filling ,reacted completely with hydrogen chloride .Calculate the volume of Hydrogen gas produced.
(Molar gas volume at r.t.p. is 24 dm3, Fe=56.0) (2 Marks)
- One of the industrial uses of chlorine gas is manufacture of Hydrochloric acid.
- Give one source of chlorine gas. (1 Mark)
- Name another major raw material for this process and state its source. (2 Marks)
-
MARKING SCHEME
-
-
- A 1 It gains two electrons to acquire electron arrangement of a noble gas.1
- Giant ionic structure1 .it is formed through ionic bonding i.e. through transfer of electrons from C to oxygen.1
- E is more reactive than H.½ E has a higher effective nuclear charge than H and gains electrons more readily than H.½
- I B(S) +Cl2 (g) → BCL2 (S)1
II 2.62g of B reacts with 2.42 dm3 of Cl2
? of B will react with 22.4dm3 of Cl2
=22.4 x2.62g 1
2.42
. : RAM of B = 24.25 1 - F has a larger atomic radius than G.1
G has a higher effective nuclear charge than F and therefore its electrons are attracted very close towards the nucleus thus minimizing the atomic size.1
-
- I U. ½ It is a good conductor both in solid as well as in liquid form.½
II W. ½ Its melting point is below room temperature but its boiling point is above room temperature.½ - I X ¹
II Y ¹
- I U. ½ It is a good conductor both in solid as well as in liquid form.½
-
-
-
- The thistle funnel should be extended to the water in the conical flask.
-
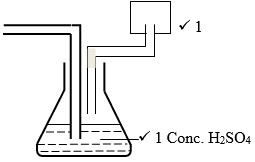
- Sodium Peroxide½ (Na2O2(S))½
- 2Na2 O2 (S) + 2H2O(l) → 4 NaOH (aq) +O2(g)¹
- Manganese (IV) Oxide and Hydrogen Peroxide.¹
-
- Continues to burn with a blue flame and a colourless gas with a irritating chocking smell ¹is formed.
- S(s) + O2 (g) → SO2 (g)¹
- PH =3.5¹
Dissolves in water to form dilute Sulphuric (IV) Acid which is a weak¹ acid.
-
-
-
- 1000cm3of HCl solution = 1 mole
20cm3of HCl solution = 20cm3 x 1 =0.02 moles¹
1000cm3 - Sodium carbonate = 0.02 moles
- 2HCl (aq) + Na2CO3 (aq) → 2NaCl (aq) + CO2 (g) +H2O (l) ¹
- Mole ratio Acid : Carbonate
2 : 1
25cm3 of Na2 CO3 solution → 0.020 moles¹
2
= 0.01 moles - If 25cm3of Na₂CO₃ solution = 0.010 moles ¹
100cm³ of Na₂ CO₃ soln = 100 cm³ x 0.01
25cm³
=0.04 moles ¹ - R.F.M of Na₂ CO₃ = (2 x 23) +(12 x 1)+ (16 x 3)¹
=106g
1 mole of Na₂CO₃ → 106 g
0.04 moles of Na₂ co₃ → 0.04 x 106 =4.24 g¹
1 - Mass of NaCl in mixture
= (10.6- 4.24)g¹
= 6.36g
% of NaCl =6.36g x 100 =60%¹
10.6
- 1000cm3of HCl solution = 1 mole
-
- Mass of oxygen
=100-(55 + 9.1) %
=100-64.1
=35.9%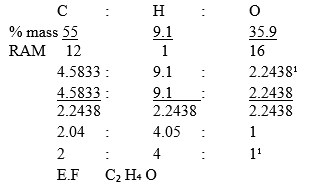
-
- 67cm³ of p → 0.262g
22400cm³ → 22400 x 0.262 g
67cm³
22400cm³ of p = 87.594¹
RFM of P =87.594. - E. F . M of P =(12 x2 )+(1 x 4)+(16x1)=44
n RFM/EFM=87.594/44 = 2
Molecular formula of P.
=(C₂ H₄ O )₂¹
=C₄ H₈ O₂
- 67cm³ of p → 0.262g
- Mass of oxygen
-
-
-
-
- Zn(s) +2HCl(aq) → ZnCl2(aq) +H2 (g)¹
- 2H₂ (g) + O₂ (g) → 2H₂ O(L) ¹
-
- Manufacture of hydrochloric acid
- Manufacture of ammonia gas by the Haber Process
- Hardening of oils in the manufacture of margarine. (any 2 @¹)
-
-
- Black Copper (II) Oxide turns to brown copper metal.¹
- Hydrogen burns with a blue flame forming a colorless liquid.¹
- Reduction /reducing agent.¹
- CuO(S) + H₂ (g) → Cu (s) +H₂O(l)¹
- To drive out air to prevent an explosion occurring.¹
-
-
-
-
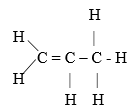
- Reagent : Hydrogen gas.¹
Conditions – Nickel catalyst /platinum
-temperature of 1800C-2000C¹ -
- 1, 2 – Bromo propan -1-ol ½
- K = Carbon (IV) oxide½
- L = 1,2- dichloropropane½
- M = Polypropane.½
- Conditions ;- U-V light /sunlight; excess chlorine.¹
-
- CH₃- CH= CH₂ +Cl₂ → CH₃ –CH Cl –CH₂ Cl¹
- Addition reaction ¹
-
- M- polypropene¹
- - Making buckets¹
- Making plastic chairs and tables. - - They lead to environmental pollution.¹
- They are non- biodegradable since bacteria cannot degrade it , it persists in the environment.
-
-
- Animal charcoal ¹ – Used to absorb colors in sugar industry.
Lamp black - Used to make printers ink /paint.
Coke - Used in fuel industries.¹ -
- Region B: CO₂(g) +C(s) 2CO(g)¹
Region C: C(g) + O₂ (g) CO₂ (g)¹ - In region A carbon (II) Oxide is oxidized by oxygen from air to form carbon (IV)oxide hence it burns producing a blue flame.¹
- Region B: CO₂(g) +C(s) 2CO(g)¹
-
- Solid L –sodium chloride (solid)½
Gas M –Ammonia gas.½ - Step (II) –filtration ½ step (III) –Heating /Thermal decomposition.½
- Finely divided iron catalyst.¹
- Step (I)
NaCl (aq) +NH₃ (g) +CO₂ (g) +H₂ O(l) → NH₄ Cl (aq) +Na H CO₃(s)¹
step III
heat
2NaHCO₃(S) → Na₂ CO₃(S) +CO₂(g) +H₂ O(L)¹ - Limestone (Calcium carbonate is heated to give carbon (iv) oxide.½
Recycling carbon (IV) oxide produced when Na H CO₃ decomposes.½
heat
CaCo₃(s) → CaO(s) + CO₂(g) - - Softening hard water¹
- Manufacture of glass
- Manufacture of soaps and detergents.
- In paper making process.
- In manufacture of sodium Hydroxide.
- Making textiles
- Used as a fixative.
- Solid L –sodium chloride (solid)½
- Animal charcoal ¹ – Used to absorb colors in sugar industry.
-
-
- Sodium chloride and concentrated Sulphuric (IV) acid.¹
- Concentrated sulphuric acid, Anhydrous Calcium chloride.¹
- NaCl(S) + H₂ SO₄(l) → Na H SO₄(aq) + HCl (aq)¹
- Introduce a glass rod dipped in ammonia solution into a gas jar containing Hydrogen chloride gas.
Formation of dense white fumes confirms presence of Hydrogen Chloride gas.
-
- 2HCl (g) + Fe (s) → FeCl₂(s) + H₂(g)¹
- 2H₂(g) + O₂(g) → 2H₂O(g)¹
- From the above equation (i)
56g of Fe produce – 24000cm³¹
1.96g ------------- produce ?
1.96 x 24000 = 840cm³¹
56
-
- From electrolysis of brine ¹
- Hydrogen¹
- From electrolysis of brine ¹
- from cracking of alkanes¹
-
Download Chemistry Paper 2 Questions and Answers - Form 3 End Term 3 2022 Exams.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

