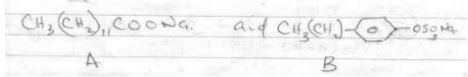CHEMISTRY
PAPER 1
FORM 4 MID TERM 2
INSTRUCTIONS
- Answer all questions
-
- Distinguish between allotropy and isomerism (2mks)
- Draw and name an isomer of butane (1mks)
-
- State grahams law of diffusion(1mks)
- 60cm3 of oxygen gas diffused through a porous portion in 50 seconds. How long would it take of sulphur (IV) oxide gas to diffuse through the same portion under same conditions? (S=32.0, 0-16.0)(3mks)
-
- Both iodine and astatine belong to the same group in the periodic table.name the group(1mks)
- Astatine is below iodine in the group how does their boiling points compare Explains?(2mks)
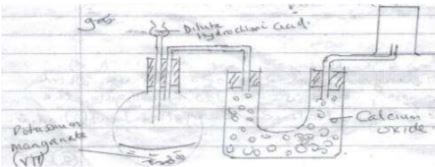
- A mixture contains iron (ii) chloride, zinc oxide and potassium chloride. Describe how each of the substances can be obtained from the mixture(3mks)
-
- what is a fuel(1mks)
- Other than heat content what other two factors should be considered when choosing a fuel (1mks)
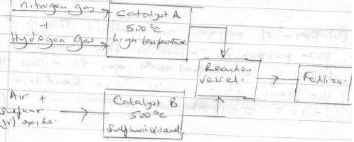
- The following is a flow chart representing the manufacture of fertilizer
- Identify (2mks)
- Catalyst A
- Catalyst B
- Give one source of hydrogen gas(1mks)
- Write down the formula of fertilizers formed(1mks)
- Identify (2mks)
- Water from kerugoya is suspected to contain sulphate ions.
- Describe how the presence of sulphate ions in the water can be shown(2mks)
- State one advantage of drinking hard water rather than soft water (1mks)
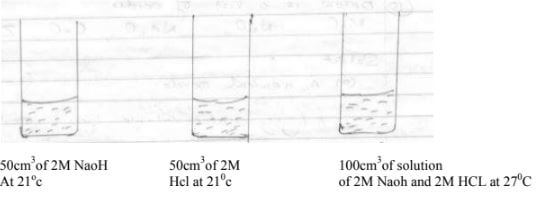
- The diagram below represents neutralization process carried out by a student
Calculate the molar heat of neutralization for the reaction that takes place (take specific heat capacity -4.2kg 'k and density of solution=1g/cm)(3mks) - The table below shows relative atomic masses and percentage abundance of the isotopes Li, L2 of element L
Relative atomic masses %age of abundance L1
L262.93
64.9369.09
30.91
Calculate the relative atomic mass of L - Below is a list of oxides
MgO, N2O, Na2O, CaO, . Select- Neutral oxide (1mks)
- Highly water soluble basic oxide(1mks)
- An amphoteric oxide (1mks)
-
- state Charles law(1mks)
- A certain gas occupied 4.2dm at 27°C and 2 atmosphere pressure. At what pressure will it be halved if the temperature then was 127°C (3mks)
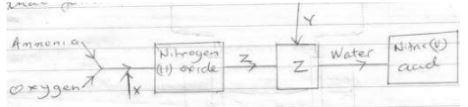
- Study the chart below for manufacture of nitric(v)acid and answer the questions that follows
- Name:(3mks)
X
Y
Z - Give the equation for the reaction in which nitrogen (ii) oxide is formed(1mk)
- State one use of nitric (v) acid(1 mk)
- Name:(3mks)
- Starting with calcium oxide describe how a solid sample of calcium carbonate can be prepared (3mks)
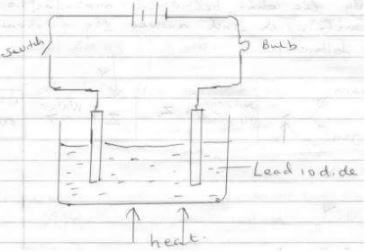
- The diagram below shows set up which was used by a student to investigate the effect of electricity on molten lead(ii) iodide
- Identify cathode and anode (1mks)
- Why does solid lead (m) iodide not allow the passage of electricity(1mks)
- Write equations to show reaction taking place
- At cathode
- At anode (2mks)
- The diagram below shows a set up used by a student to prepare and collect chlorine gas
- Identify with reasons two mistakes in the set up(3mks)
- Give another set of reagents that can be used to prepare chlorine gas (1mks
- Given below are pH values of different solutions P, Q, and S. study it and answer the questions that follows
Solution pH P 1 Q 7 S 14 - Which two solution would react together to give a pH. of 7.0 (1mks)
- Which solution is likely to be sodium chloride solution (1mks)
- What is the color of phenolphthalein when added to solutions (1mks)
- The table below gives properties of four substances Substances
Substances Melting
PointBoiling
PointsElectrical conductivity Solid Liquid A 1083 2567 Good Good B -182 -164 Poor Poor C 1723 2230 Poor Good D 993 1695 Poor Poor
State with reasons which of the above is- An ionic compound (1mks)
- Metallic structure (1mks)
- A giant atomic structure(1mks)
- Metal X displaced Z from its salt solution and metal Y displaced metal W from its salt solution. It was also observed that W and Y reacted with steam but X and Z did not. The reactivity of the metals starting with the most reactive is (2mks)
- Two detergents And B represented as
- Which of the detergent is suitable for washing using water containing magnesium sulphate(2mks)
- Give one disadvantages of continuous use of the detergent you have choose in (a) above (1mks)
- A compound was found to contain 48.7% carbon 8.1% hydrogen while the rest was oxygen by mass. If the relative molecular mass of the compound is 148 determine its molecular formula(C=12, H=1, O=16)(3mks)
-
- What is a saturated solution(1mks)
- 28gms of a saturated solution of the salt at 25°c yielded 7gms of solid when evaporated to dryness. Find the solubility of the salt at 25°C(3mks)
-
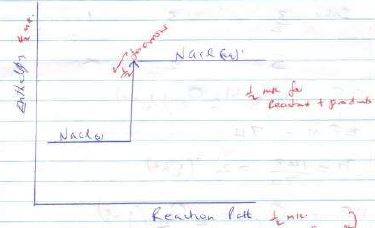
- The lattice energy for sodium chloride is 781kj/moa while the hydration energy is 774kjmol-1 calculate the enthalpy of solution of sodium chloride(2mks)
- Draw an energy level diagram when 1 mole of sodium chloride dissolves in water. (2mks)
- A form two student during an experiment left a container containing concentrated sulphiric (vi) acid exposed in air for two days. After two days he observed the level of acid had risen
- Why did the level of acid in the container rise (2mks?)
- How is this useful in the laboratory (1mks)
- A clean knife is left in the open ovemight and found to be coated with a reddish brown substances
- Write down the chemical formula of reddish brown substances (1mks)
- Give one condition necessary for the brown substance to be formed (1 mks)
- Suggest two methods that can be used to prevent formation of the reddish brown substances on the knife (2mks)
MARKING SCHEME
-
- allotropy is the existence of an element in different forms but same physical state while isomerism is existence of a compound with same molecular fromular but different sturctural formular.
(both must be correct)
- allotropy is the existence of an element in different forms but same physical state while isomerism is existence of a compound with same molecular fromular but different sturctural formular.
-
- The rate of diffusion of a gas is inversely proportional to the square root of its density.
50/T2=√ 32/√ 64
2500/T2 = 32/64
T22=2500 x 64/32=5000
T2=70.7 seconds
- The rate of diffusion of a gas is inversely proportional to the square root of its density.
-
- Halogens
- Astatine has a higher than Iodine. Astatine has a larger atomic radius than Iodine Hnece forces of attraction.
- Heat the mixture iron(iii)chloride sublimes and collect on cooler parts of apparatus. Add water to the remaining mixture potassium chloride dissolves off Zinc(ii)oxide as residue. Evaporate filtrate to obtain potassium chloride.
-
- A fuel is any substance which when heated produces heat energy.
- Availability, ease of storage, environental pollution.
-
-
- Finely divided iron/platinum
- Vanadium (v) oxide
- Cracking of alkanes/ passing steam over heated coke.
- (NH4)SO4
-
-
- Take a sample of water add Barium nitrate followed by a few drops of dilute HCl. A white ppt that persists on addition of HCl. Presence of SO42- in the water.
- Ca2+ ions are good for teeth and bone formation
- ΔT=27-21=6k
ΔH=100/1000 x 42 x 6 = 2.52KJ
1000cm3 of HCl → 2M
50cm3 of HCl = 2/1000 x 50= 0.1moles
0.1 moles → 2.52KJ
1 mole → 2.52/0.1 x 1
25.2KJ/mol - RAM = 62.93 x 69.09 + 64.93 x 30.91
100
= 4347.33 + 2006.99
100
=63.55 -
- N2O
- Na2O
- ZnO
-
- The volume of a fixed mass of gas is directly proportional to the absolute temperature at constant pressure.
- 2 x 4.2/300 = p2 x 2.1/400
D2= 2 x 42 x 400
300
=5.33 atmospheres
-
- X- air
Y-Water
Z-NO2 - 4NH3(g) + SO2(g) → 4NO(g) + 6H2O(g) + Heat
-
- Manufacture of nitrogen fertilizer
- dyes
- Explosives
- Picking of metals
- X- air
- Add dilute nitric acid to form calcium nitrate. Add aqueous sodium carbonate/potassium carbonate to precipitate calcium carbonate filter off calcium carbonate and dry it between filter papers.
-
- Anode (shown)
Cathode(shown) - Solid lead(ii) iodide does not contain mobile ions
- Cathode Pb2+ + 2e- → Pb(s)
Anode 2I(aq)- → I2(s) + 2e-
- Anode (shown)
-
- Method of collection - chlorine is denser than air and should be collected by downward delivery
Drying agent - Calcium oxide would react with chlorine -
- Bleaching powder CaOCl2 + dil HCl
Manganese(iv) oxed + dil HCl - Sodium Chloride, manganese(iv) oxide and conc sulphuric acid.
- Bleaching powder CaOCl2 + dil HCl
- Method of collection - chlorine is denser than air and should be collected by downward delivery
-
- P and Q
- Q
- Pink/ red
-
- C - high melting point and boiling point and good conductor of electricity in liquid form
- A- A high melting point and boiling point conduct electricity in solid and liquid form
- D- high melting point but poor conductor in liquid and solid form.
-
- B
- Pollute environment - it is non-bio degradable.
-
C H O 48.7 8.1 43.2 48.7/12 8.1/1 43.2/16 4.058 8.1 2.7 1.5 3 1 3/2 3 1 3 6 2 EF C3H6O2
EFM=74
n=148/74=2
MF= C6H12O4 -
- A solution that cannot dissolve any more solute at a particular temperature
- Mass of water in solution = 21gms
21gms of water → 7gms
100gms →?
7/21 x 100= 33.33 gms per 100g of water
- ΔH solution = ΔH + ΔHHydration
=781 + -774= 7KJ
-
- Conc Sulphuric(vi) acid in hugroscopic and absorb water causing rise in level
- It is used as a drying agent
-
- Fe2O3 x H2O - Hydrated iron(iii) oxide
- Moisture, air
- Painting, oiling, galvanizing, anodizing
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - Form 4 Mid Term 2 Exams 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students