CHEMISTRY
PAPER 3
FORM 4 MID TERM 2
INSTRUCTIONS
- Answer all questions
- QUESTION 1
You are provided with:- Solution Q 0.125M Hydrochloric acid solution
- N grammes of anhydrous sodium carbonate.
- Methylorange indicator
You are required to prepare a solution of sodium carbonate and then standardize it with hydrochloric acid solution Q.
Procedure - Transfer all the N grams of sodium carbonate into a 250 ml volumetric flask.
- Add 100cm3 of distilled water and shake till all the solid dissolves.
- Add more distilled water upto the 250 ml mark and label it solution P. using a measuring cylinder, transfer 50cm3 of solution P into a clean 250 ml beaker and add 50cm3 of distilled water.
- Stir well with a glass rod and label it solution T.
- pipette 25.0cm3 of solution T and place it into a conical flask, add 2 drops of methyl orange indicator and titrate with solution Q from the burette. Record your results in the table 1 below and repeat the titration part to complete the table below.
(3 mks)
I II III Final burette reading (cm3) Initial burette reading (cm3) Volume of Q used (cm3) - Calculate the average volume of solution Q used. (1 mk)
-
- Calculate the number of moles of solution Q used. (2 mks)
- Write an equation for the reaction between solution T and Q. (1 mk)
Calculate: - Number of moles of sodium carbonate solution in 25cm3 of solution T. (2 mks)
- Number of moles of sodium carbonate in 100cm3 of solution T. (2 mks)
- Number of moles of sodium carbonate in 50cm3 of the original solution P. (2 mks)
- Given that Na=23.0, C=12.0, O=16.0;
- The mass of sodium carbonate N grammes that were dissolved to make solution P. (1 mk)
- The concentration of sodium carbonate solution P in moles per litre. (2 mks)
- QUESTION 2
You are provided with:- 1 M Sulphuric (vi) acid solution D.
- 2 M Sodium hydroxide solution C.
You are required to determine the heat of neutralization of Sulphuric (vi) acid solution D.
Procedure - Measure 50cm3 of solution C and transfer into a 250cm3 plastic beaker provided. record the initial temperature of solution C.
- measure 10cm3 of sulphuric(vi)acid solution D and add it to solution C in the beaker. Stir gently with the thermometer to mix and record the highest temperature in the table below.
- Continue adding 10cm3 portions of solution D recording the temperature after each addition until 80cm3 of solution D has been added. (3 mks)
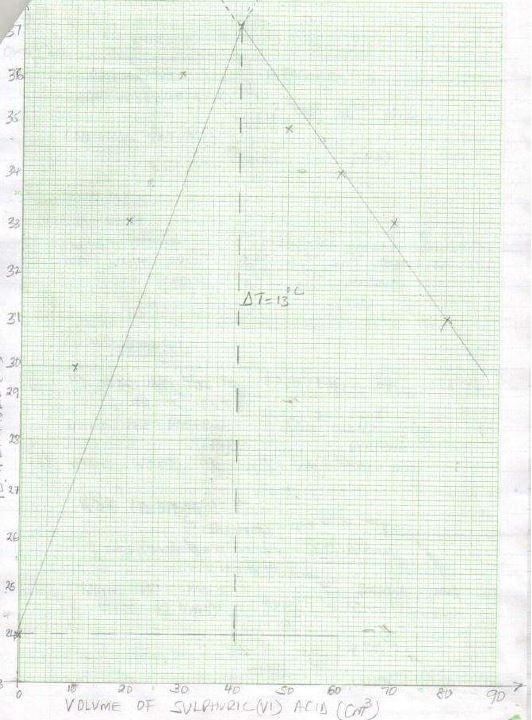
Total volume of solution D added (cm3) 0 10 20 30 40 50 60 70 80 Temperature of the mixture (ºC) - Plot a graph of temperature against volume of sulphuric(vi)acid solution D added. (3 mks)
- From your graph determine the maximum temperature change ΔT. (1/2 mk)
- From your graph determine the volume of sulphuric(vi)acid solution required for neutralization.(1/2 mk)
- Calculate the molar heat of neutralization of 1M sulphuric(vi)acid solution D. (Assume density of water = 1g/cm3, specific heat capacity = 4.2 Jg-1K-1) (3 mks)
-
- You are provided with solid A. carry out the following tests, record the observations and inferences.
- Place solid A in a boiling tube and add about 10cm3 of distilled water while shaking. Filter the solution and divide the filtrate into three portions. Keep the residue for part (b).
Observation Inference - To the first portion, add sodium hydroxide dropwise till in excess.
Observation Inference - To the second portion add 3 drops of barium nitrate solution.
Observation Inference - To the third portion add 3 drops of lead(ii)Nitrate solution
Observation Inference
- To the first portion, add sodium hydroxide dropwise till in excess.
- Place the residue in(a) above in a boiling tube. Add dilute nitric(v)acid while shaking till the solid just dissolves.Divide the solution into two portions.
Observation Inference - To the first portion add sodium hydroxide solution dropwise till in excess.
Observation Inference - To the second portion, add a few drops of ammonia solution then in excess.
Observation Inference
- To the first portion add sodium hydroxide solution dropwise till in excess.
- Place solid A in a boiling tube and add about 10cm3 of distilled water while shaking. Filter the solution and divide the filtrate into three portions. Keep the residue for part (b).
- You are provided with solid Z. carry out the tests below and record your observations and inferences. Place all of solid Z into a boiling tube. Add 10cm3 of distilled water and shake well. Divide the solution into three portions.
- Test the first portion with both blue and red litmus paper.
Observation Inference - To the second portion add 2 drops of acidified potassium manganate(vii) and shake well.
Observation Inference - To the third portion add a little sodium hydrogen carbonate.
Observation Inference
- Test the first portion with both blue and red litmus paper.
- You are provided with solid A. carry out the following tests, record the observations and inferences.
CONFIDENTIAL
In addition to the other common apparatus and reagents found in a chemistry laboratory, each candidate will require the following.
- About 100cm3 of 0.125M Hydrochloric acid labelled solution Q.
- Accurately weighed 2.65g of unhydrous sodium carbonate labeled solid N.
- 250ml volumetric flask.
- 100ml measuring cylinder.
- Distilled water
- 250 ml empty glass beaker
- Glass rod
- 1 label
- One burette (0-50ml)
- One 25.0 ml pipette
- Two conical flasks (250 ml)
- Methyl orange indicator
- Retort stand
- Pipette filler
- A white tile
- 6 dry test tubes
- 1 boiling tub
- One filter funnel
- 0-1100C thermometer
- 1 metallic spatula
- About 60cm3 of 1M Sulphuric(vi)acid labeled solution D.
- About 60cm3 of 2M sodium hydroxide solution labeled solution C.
- 250 ml plastic beaker empty.
- About 0.5g of solid Z.
- About 1.5g of solid A.
- 1 red and 1 blue litmus paper.
- About 0.2g of sodium hydrogen carbonate.
- 1 filter paper.
Access to:
- 2M NaOH with a dropper.
- 2M Ammonia solution with a dropper.
- 0.25M Barium nitrate solution with a dropper.
- 0.2M Lead (ii) nitrate solution with a dropper.
- 0.02M acidified Potassium Manganate (vii) with a dropper.
- 1M Nitric(v) acid.
NOTE:
- Solid A is a mixture of zinc carbonate and Sodium Sulphate in the ratio 1:1.
- Solid Z is oxalic acid.
- Solution Q is 0.125M Hydrochloric acid prepared by dissolving 10.75cm3 of concentrated hydrochloric acid in 1 litre.
- Solution D is 1M Sulphuric (vi) acid prepared by dissolving 55 cm3 of concentrated sulphuric (vi) acid in 1 litre of solution (Density of Sulphuric acid is 1.84 g/cm3).
- Solution C is 2M sodium hydroxide prepared by dissolving 80g of sodium hydroxide pellets in 1 litre of solution.
- 2 M Ammonia solution is prepared by dissolving 112cm3 of concentrated ammonia solution in 1 litre of solution.
- 0.25M Barium nitrate is prepared by dissolving 65g of Barium nitrate in 1 litre of solution.
- 0.2M lead(ii)nitrate is prepared by dissolving 66.2g of lead(ii)nitrate in a litre of solution.
- 1 M nitric acid is prepared by dissolving 64cm3 of concentrated Nitric(v) acid in 1 litre of solution.
- 0.02M acidified potassium manganate(vii) is prepared by dissolving 3.2g of the solid in 2.00cm3 of 2M sulphuric(vi) acid then diluting to 1 litre.
MARKING SCHEME
-
Table:- Award 1 mark for 3 titrations
- Award 1/2 mark for only 2 titrations
- Award 0 mk for 1 titration done
- Decimal - 1mk
- Accuracy 1mk
± 0.2cm3 award 1/2 mk
± o.1cm3 award 1mk
Total 3 mks- Average volume of solution Q= 20.0cm3
-
- moles of solution Q used
20 x 0.125 = 0.0025 moles
1000 - Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + CO2(g) + H2O(l)
- Moles of Na2CO3 in 25cm3 of solution T
1/2 x 0.0025 = 0.00125 - Moles of Na2CO3 in 100cm3 of solution T
25cm3 → 0.00125moles
then 100cm3 = 0.00125 x 100 = 0.005 moles
25 - Moles of Na2CO3 in 50cm3 of the original solution P
0.00moles → 100cm3 of T
→ 250cm3
250 x 0.005=0.025
50 - Mass of Na2CO3 in grammes
=0.025 x 106g= 2.65g - Concentration of Na2CO3 in solution P
2.65 →250cm3
? → 1000cm3
2.65 x 1000 = 10.6g/litre
250
Molarity= 10.6/106 = 0.1m
- moles of solution Q used
-
Total volume of solution D added (cm3) 0 10 20 30 40 50 60 70 80 Temperature of the mixture (ºC) 240. 30.0 33.0 36.0 37.0 35.0 34.0 33.0 31.0
CT 1 mk
D.P - 1mk
Temperature at 0 volume of solution D ± 2D- Graph of temperature against volume of sulphuric (vi) acid added during neutralization reaction
Labelled axis with units - 1mk
plotting - 1mk
Shape - 1mk
Total - 3mks - Temperature changes =- 13.0ºC
- Volume of H2SO4 required for neutralization 40cm3(1/2mk)
- Molar heat of neutralization
Heat change= MCθ
=(50 + 40) x 42 x 13J
=90 x 42 x 13J= -4914J
Moles of H2SO4 = 1 x 40= 0.04
1000
0.04 moles → -4914J
1mole → ?
-4.914 = 122.85KJ/mol
0.04
- Graph of temperature against volume of sulphuric (vi) acid added during neutralization reaction
-
-
-
Observation Inference - White residue
- Colourless filtrateCu2+, Fe2+, Fe3+ present -
Observation Inference No white ppt Pb2+, Al 3+. Zn2+ absent
or
K+ or Na+ present -
Observation Inference White ppt formed SO42-, SO32-, CO32- present
(any two) -
Observation Inference White ppt present SO42- present
-
-
Observation Inference Effervesence, produces colourless gas CO32- present -
Observation Inference White ppt which dissolves in excess Pb2+, Al3+, Zn2+ present -
Observation Inference White ppt that dissolves in excess Zn2+ present
-
-
-
-
Observation Inference Blue litmus turns to red,
red litmus remains redH+ present -
Observation Inference Purple acidified potassium manganate (vii) is decolourised | |
C = C
| |
-C≡C-
or
R-OH present -
Observation Inference Effervesence present H+ present
-
-
Download Chemistry Paper 3 Questions and Answers with Confidential - Form 4 Mid Term 2 Exams 2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students