Figure 14 shows a setup that can be used to determine the specific latent heat of vaporisation of water. A beaker containing some water was placed on a weighing balance and an immersion heater rated 500 W immersed in the water.
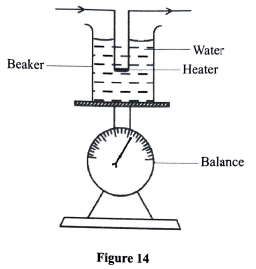
The water was then heated until it boiled. When the water started boiling, the initial the balance was reading on noted and the stop watch started immediately. The final was then noted reading on the balance after a time t seconds.
- State how the mass of steam can be measured using this setup.
- Write down an expression for the heat supplied by the heater.
- Determine the specific latent heat of vaporisation of water.