In a certain factory, iron is extracted from the haematite ore using the blast furnace as shown in Figure 1. The other raw materials are coke, limestone and air. The melting and boiling points of iron are 1535°C and 3000 °C, respectively.
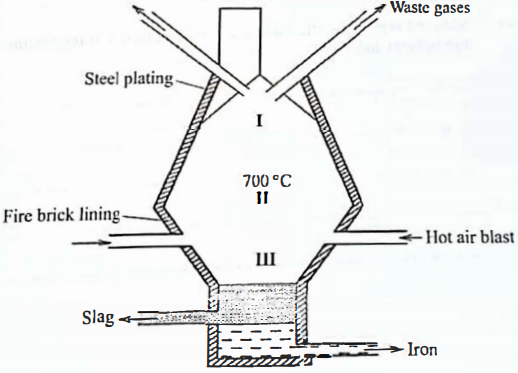
Figure 1
- State how the temperature in region I compares with that in region II. Give a reason.
- The main reducing agent in the furnace is carbon(II) oxide formed by the reaction
CO2(g) + C(s) →2C0g)
Write two equations to show how carbon(IV) oxide is formed in the furnace. - Suggest a value for the temperature in region III. Give a reason.
- Name the main component in the slag.
- State one role that slag plays in the blast furnace.
- The iron produced in the blast furnace is brittle due to presence of impurities
- Name the main impurity in this iron
- State one use of this iron
- Recycling is one method used to reduce production costs. State and explain the by products that can be recycled in this factory.