The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters are not actual symbols of the elements.
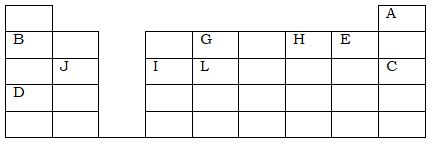
- What name is given to the family of elements to which A and C belong?
- Write the formula of the sulphate of element D.
- Which letter represents the most reactive;
- Metal
- Non-metal
- Name the bond formed when B and H react. Explain your answer.
- Select one element that belongs to period 4.
- Explain why the ionic radius of element E is bigger than the atomic radius.
- The electron configuration of a divalent anion of element N is 2.8.8. Indicate the position of element N on the periodic table above.
- The oxide of G has a lower melting point than the oxide of L. Explain.
- How do the atomic radii of I and C compare. Explain.
- Explain the trend in the 1st ionization energies of the elements J, I and L.