The diagram below shows a set-up that can be used to obtain nitrogen gas in an experiment.
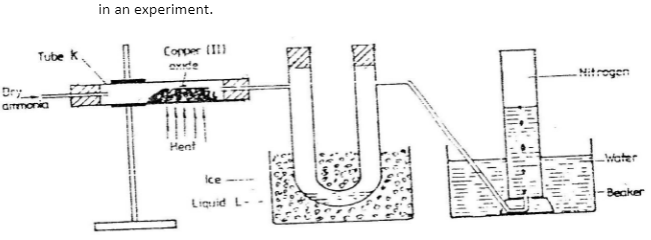
-
- Name liquid L
- What observation would be made in tube K after heating for some time?
- Write an equation for the reaction that took place in tube K.
- If 320cm3 of ammonia gas reacted completely with the copper?
Calculate:- Volume of nitrogen gas produced. (1mk)
- the mass of copper oxide that reacted (3mks)
(Cu = 63.5, O=16.O, one mole of gas occupies 24 litres at room temperature and pressure)
- At the end of experiment the PH of the water in the beaker was found to be about 2. Explain.