A form three student from Anestar High School used the set up drawn below in an attempt to prepare dry carbon IV oxide .Study it carefully and use it to answer the questions that follows.
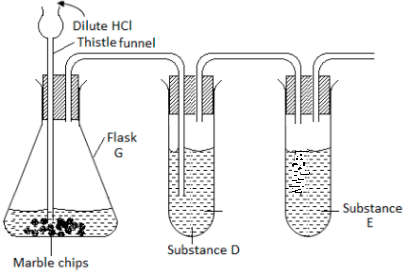
- Identify two faults in the set up of the apparatus.
- Complete the diagram to show how dry carbon IV oxide gas can be collected.
- Name substances D and E and give their purposes in the above set up
- Substance D.
Purpose - Substance E.
Purpose
- Write the equation of the reaction in the flask G.
- State and explain the observation made when dilute sulphuric acid is used in place of hydrochloric acid.