
Introduction to Acids, Bases and Indicators
In a school laboratory:
- An acid may be defined as a substance that turns litmus red.
- A base may be defined as a substance that turns litmus blue.
Litmus is lichen found mainly in West Africa. It changes its colour depending on whether the solution it is in, is basic/alkaline or acidic. It is thus able to identify/show whether another substance is an acid, base or neutral. - An indicator is a substance that shows whether another substance is a base/alkaline, acid or neutral.
Common naturally occurring acids include:
| Name of acid | Occurrence |
| Citric acid | Found in ripe citrus fruits like passion fruit/oranges/lemon |
| Tartaric acid | Found in grapes/baking powder/health salts |
| Lactic acid | Found in sour milk |
| Ethanoic acid | Found in vinegar |
| Methanoic acid | Present in ants, bees stings |
| Carbonic acid | Used in preservation of fizzy drinks like coke, Lemonade, Fanta |
| Butanoic acid | Present in cheese |
| Tannic acid | Present in tea |
Most commonly used acids found in a school laboratory are not naturally occurring. They are manufactured.
They are called mineral acids.
Common mineral acids include:
| Name of mineral acid | Common use |
| Hydrochloric acid (HCl) | Used to clean/pickling surface of metals Is found in the stomach of mammals/human beings |
| Sulphuric(VI) acid (H2SO4) | Used as acid in car battery, making battery, making fertilizers |
| Nitric(V)acid (HNO3) | Used in making fertilizers and explosives |
Mineral acids are manufactured to very high concentration. They are corrosive (causes painful wounds on contact with the skin) and attack/reacts with garments/clothes/metals.
In a school laboratory, they are mainly used when added a lot of water. This is called diluting. Diluting ensures the concentration of the acid is safely low.
Bases are opposite of acids. Most bases do not dissolve in water. Bases which dissolve in water are called alkalis.
Common alkalis include:
| Name of alkali | Common uses |
| Sodium hydroxide (NaOH) | Making soaps and detergents |
| Potassium hydroxide(KOH) | Making soaps and detergents |
| Ammonia solution(NH4OH) | Making fertilizers, softening hard water |
Common bases (which are not alkali) include:
| Name of base | Common name |
| Magnesium oxide/hydroxide | Anti acid to treat indigestion |
| Calcium oxide | Making cement and neutralizing soil acidity |
Indicators are useful in identifying substances which look-alike.
An acid-base indicator is a substance used to identify whether another substance is alkaline or acidic. An acid-base indicator works by changing to different colors in neutral, acidic and alkaline solutions/dissolved in water.
Experiment: To prepare simple acid-base indicator
Procedure
-
- Place some flowers petals in a mortar.
- Crush them using a pestle.
- Add a little sand to assist in crushing.
- Add about 5cm3 of propanone/ethanol and carefully continue grinding.
- Add more 5cm3 of propanone/ethanol and continue until there is enough extract in the mortar.
- Filter the extract into a clean 100cm3 beaker.
- Place 5cm3 of filtered wood ash, soap solution, ammonia solution, sodium hydroxide, hydrochloric acid,
distilled water, sulphuric (VI) acid, sour milk, sodium chloride, toothpaste and calcium hydroxide into separate
test tubes. - Put about three drops of the extract in (a)to each test tube in (b). Record the observations made in each case.
Sample observations
| Solution mixture | Colour on adding indicator extract | Nature of solution |
| wood ash | green | Base/alkaline |
| soap solution | green | Basic/alkaline |
| ammonia solution | green | Basic/alkaline |
| sodium hydroxide | green | Basic/alkaline |
| hydrochloric acid | Red | Acidic |
| distilled water | orange | Neutral |
| sulphuric(VI)acid | Red | Acidic |
| sour milk | green | Basic/alkaline |
| sodium chloride | orange | Neutral |
| Toothpaste | green | Basic/alkaline |
| calcium hydroxide | green | Basic/alkaline |
| Lemon juice | Red | Acidic |
The plant extract is able to differentiate between solutions by their nature. It is changing to a similar colour for similar solutions.
- Since lemon juice is a known acid, then sulphuric (VI) and hydrochloric acids are similar in nature with lemon juice because the indicator shows similar colors. They are acidic in nature.
- Since sodium hydroxide is a known base/alkali, then the green colour of indicator shows an alkaline/basic solution.
- Since pure water is neutral, then the orange colour of indicator shows neutral solutions.
In a school laboratory, commercial indicators are used. A commercial indicator is cheap, readily available and easy to store. Common indicators include: Litmus, phenolphthalein, methyl orange, screened methyl orange, bromothymol blue.
Experiment: Using commercial indicators to determine acidic, basic/alkaline and neutral solutions
Procedure
- Place 5cm3 of the solutions in the table below.
- Add three drops of litmus solution to each solution.
- Repeat with phenolphthalein indicator, methyl orange, screened methyl orange and bromothymol blue.
Sample results
| Substance/Solution | Indicator Used | ||||
| Litmus | Phenolphthalein | Methyl orange | Screened methyl orange | Bromothymol blue | |
| wood ash | Blue | Pink | Yellow | Orange | Blue |
| soap solution | Blue | Pink | Yellow | Orange | Blue |
| ammonia solution | Blue | Pink | Yellow | Orange | Blue |
| sodium hydroxide | Blue | Pink | Yellow | Orange | Blue |
| hydrochloric acid | Red | Colourless | Red | Purple | Orange |
| distilled water | Colourless | Colourless | Red | Orange | Orange |
| sulphuric(VI)acid | Red | Colourless | Red | Purple | Orange |
| sour milk | Blue | Pink | Yellow | Orange | Blue |
| sodium chloride | Colourless | Colourless | Red | Orange | Orange |
| Toothpaste | Blue | Pink | Yellow | Orange | Blue |
| calcium hydroxide | Blue | Pink | Yellow | Orange | Blue |
| Lemon juice | Red | Colourless | Red | Purple | Orange |
From the table above, then the colour of indicators in different solution can be summarized.
| Indicator | Colour of indicator in | ||
| Acid | Base/alkali | Neutral | |
| Litmus paper/solution | Red | Blue | Colourless |
| Methyl orange | Red | Yellow | Red |
| Screened methyl orange | Purple | Orange | Orange |
| Phenolphthalein | Colourless | Purple | Colourless |
| Bromothymol blue | Orange | Blue | Orange |

The universal indicator
The universal indicator is a mixture of other indicator dyes. The indicator uses the pH scale. The pH scale shows the strength of bases and acids. The pH scale ranges from 1-14. These numbers are called pH values:
- pH values 1, 2, 3 shows a substance is strongly acid
- pH values 4, 5, 6 shows a substance is a weakly acid
- pH value 7 shows a substance is a neutral
- pH values 8, 9, 10, 11 shows a substance is a weak base/alkali.
- pH values 12, 13, 14 shows a substance is a strong base/alkali
The pH values are determined from a pH chart. The pH chart is a multicolored paper with each colour corresponding to a pH value. i.e
- red correspond to pH 1, 2, 3 showing strongly acidic solutions.
- Orange/ yellow correspond to pH 4, 5, 6 showing weakly acidic solutions.
- Green correspond to pH 7 showing neutral solutions.
- Blue correspond to pH 8, 9, 10, 11 showing weakly alkaline solutions.
- Purple / dark blue correspond to pH 12,13,14 showing strong alkalis.
The universal indicator is available as:
- Universal indicator paper / pH paper
- Universal indicator solution.
When determining the pH of a unknown solution using
- pH paper then the pH paper is dipped into the unknown solution. It changes/turn to a certain colour. The new colour is marched/compared to its corresponding one on the pH chart to get the pH value.
- universal indicator solution then about 3 drops of the universal indicator solution is added into about 5cm3 of the unknown solution in a test tube. It changes/turn to a certain colour. The new colour is marched/compared to its corresponding one on the pH chart to get the pH value.
Experiment: To determine the pH value of some solutions
- Place 5cm3 of filtered wood ash, soap solution, ammonia solution, sodium hydroxide, hydrochloric acid, distilled water, sulphuric (VI) acid, sour milk, sodium chloride, toothpaste and calcium hydroxide into separate test tubes.
- Put about three drops of universal indicator solution or dip a portion of a piece of pH paper into each.
- Record the observations made in each case.
- Compare the colour in each solution with the colors on the pH chart provided.
- Determine the pH value of each solution.
Sample observations
| Solution mixture | Colour on the pH paper/adding universal indicator | pH value | Nature of solution |
| wood ash | Blue | 8 | Weakly alkaline |
| soap solution | Blue | 8 | Weakly alkaline |
| ammonia solution | green | 8 | Weakly alkaline |
| sodium hydroxide | Purple | 14 | Strongly alkaline |
| hydrochloric acid | red | 1 | Strongly acidic |
| distilled water | green | 7 | Neutral |
| sulphuric(VI)acid | red | 1 | Strongly acidic |
| sour milk | blue | 9 | Weakly alkaline |
| sodium chloride | green | 7 | Neutral |
| toothpaste | Blue | 10 | Weakly alkaline |
| calcium hydroxide | Blue | 11 | Weakly alkaline |
| Lemon juice | Orange | 5 | Weakly acidic |
Note
- All the mineral acids Hydrochloric, sulphuric (VI) and nitric (V) acids are strong acids
- Two alkalis/soluble bases, sodium hydroxide and potassium hydroxide are strong bases/alkali. Ammonia solution is a weak base/alkali. All other bases are weakly alkaline.
- Pure/deionized water is a neutral solution.
- Common salt/sodium chloride is a neutral salt.
- When an acid and an alkali/base are mixed, the final product has pH 7 and is neutral.

Properties of acids
Physical properties of acids
- Acids have a characteristic sour taste
- Most acids are colourless liquids
- Mineral acids are odorless. Organic acids have characteristic smell
- All acids have pH less than 7
- All acids turn blue litmus paper red, methyl orange red and phenolphthalein colourless.
- All acids dissolve in water to form an acidic solution. Most do not dissolve in organic solvents like propanone, kerosene, tetrachloromethane, petrol.
Chemical properties of acids
Reaction with metals
All acids react with reactive metals to form a salt and produce /evolve hydrogen gas.
Metal + Acid → Salt + Hydrogen gas
Experiment: reaction of metals with mineral acids.
- Place 5cm3 of dilute hydrochloric acid in a small test tube.
- Add 1cm length of polished magnesium ribbon.
- Stopper the test tube using a thump.
- Light a wooden splint.
- Place the burning splint on top of the stoppered test tube.
- Release the thump stopper.
- Record the observations made.
- Repeat the procedure in above using Zinc granules, iron filings, copper turnings, aluminum foil in place of Magnesium ribbon
- Repeat the procedure in using dilute sulphuric (VI) acid in place of dilute hydrochloric acid.
Sample observations
- effervescence/bubbles produced/fizzing in all cases except when using copper
- Colourless gas produced in all cases except when using copper
- Gas produced extinguishes a burning wooden splint with an explosion/pop sound.
Explanation
- Some metals react with dilute acids, while others do not.
- Metals which react with acids produce bubbles of hydrogen gas.
- Hydrogen gas is a colourless gas that extinguishes a burning splint with a pop sound. This shows acids contain hydrogen gas.
- This hydrogen is displaced/removed from the acids by some metals like Magnesium, Zinc, aluminium, iron and sodium.
- Some other metals like copper, silver, gold; platinum and mercury are not reactive enough to displace/remove the hydrogen from dilute acids.
Chemical equations
- Magnesium + Hydrochloric acid → Magnesium chloride + Hydrogen
Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g) - Zinc + Hydrochloric acid → Zinc chloride + Hydrogen
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g) - Iron + Hydrochloric acid → Iron (II) chloride + Hydrogen
Fe(s) + 2HCl(aq) → FeCl2(aq) + H2(g) - Aluminium + Hydrochloric acid → Aluminium chloride + Hydrogen
2Al(s) + 3HCl(aq) → AlCl3(aq) + 3H2(g) - Magnesium + Sulphuric (VI) acid → Magnesium sulphate (VI) + Hydrogen
Mg(s) + H2SO4(aq) → MgSO4(aq) + H2(g) - Zinc + Sulphuric (VI) acid → Zinc sulphate (VI) + Hydrogen
Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g) - Iron + Sulphuric (VI) acid → Iron (II) sulphate (VI) + Hydrogen
Fe(s) + H2SO4(aq) → FeSO4(aq) + H2(g) - Aluminium + Sulphuric (VI) acid → Aluminium sulphate (VI) + Hydrogen
2Al(s) + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2(g)
Reaction of metal carbonates and hydrogen carbonates with mineral acids.
All acids react with carbonates and hydrogen carbonates to form salt, water and produce/evolve carbon (IV) oxide gas.
Metal carbonate + Acid → Salt + Water+ Carbon(IV)oxide gas
Metal hydrogen carbonate + Acid → Salt + Water + Carbon (IV) oxide gas
Experiment: reaction of metal carbonates and hydrogen carbonates with mineral acids.
- Place 5cm3 of dilute hydrochloric acid in a small test tube.
- Add half spatula full of sodium carbonate.
- Stopper the test tube using a cork with delivery tube directed into lime water.
- Record the observations made.
- Test the gas also with burning splint.
- Repeat the procedure in above using Zinc carbonate, Calcium carbonate, copper carbonate, sodium hydrogen carbonate, Potassium hydrogen carbonate in place of Sodium carbonate.
- Repeat the procedure in using dilute sulphuric (VI) acid in place of dilute hydrochloric acid.
Set up of apparatus
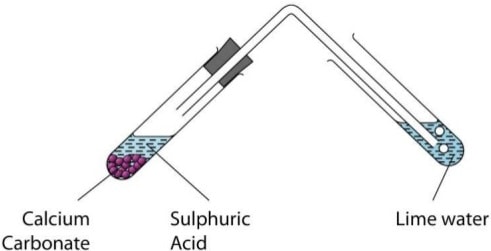
Sample observations
- effervescence/bubbles produced/fizzing in all cases.
- Colourless gas produced in all cases.
- Gas produced forms a white precipitate with lime water.
Explanation
All metal carbonate/hydrogen carbonate reacts with dilute acids to produce bubbles of carbon (IV) oxide gas. Carbon (IV) oxide gas is a colourless gas that extinguishes a burning splint. When carbon (IV) oxide gas is bubbled in lime water, a white precipitate is formed.
Chemical equations
- Sodium carbonate +Hydrochloric acid → Sodium chloride + Carbon (IV) Oxide+ Water
Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(g) + CO2(g) - Calcium carbonate +Hydrochloric acid → Calcium chloride + Carbon (IV) Oxide+ Water
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(g) + CO2(g) - Magnesium carbonate +Hydrochloric acid → Magnesium chloride + Carbon (IV) Oxide+ Water
MgCO3(s) + 2HCl(aq) → MgCl2(aq) + H2O(g) + CO2(g) - Copper carbonate +Hydrochloric acid → Copper (II) chloride + Carbon (IV) Oxide+ Water
CuCO3(s) + 2HCl(aq) → CuCl2(aq) + H2O(g) + CO2(g) - Copper carbonate +Sulphuric (VI) acid → Copper (II) sulphate (VI) + Carbon (IV) Oxide+ Water
CuCO3(s) + H2SO4(aq) → CuSO4(aq) + H2O(g) + CO2(g) - Zinc carbonate + Sulphuric (VI) acid → Zinc sulphate (VI) + Carbon (IV) Oxide+ Water
ZnCO3(s) + H2SO4(aq) → ZnSO4(aq) + H2O(g) + CO2(g) - Sodium hydrogen carbonate +Sulphuric (VI) acid → Sodium sulphate (VI) + Carbon (IV) Oxide+ Water
NaHCO3(s) + H2SO4(aq) → Na2SO4(aq) + H2O(g) + CO2(g) - Potassium hydrogen carbonate +Sulphuric (VI) acid → Potassium sulphate (VI) + Carbon (IV) Oxide+ Water
KHCO3(s) + H2SO4(aq) → K2SO4(aq) + H2O(g) + CO2(g) - Potassium hydrogen carbonate +Hydrochloric acid → Potassium chloride + Carbon (IV) Oxide+ Water
KHCO3(s) + HCl(aq) → KCl(aq) + H2O(g) + CO2(g) - Sodium hydrogen carbonate +Hydrochloric acid → Sodium chloride + Carbon (IV) Oxide+ Water
NaHCO3 (s) + HCl(aq) → NaCl(aq) + H2O(g) + CO2(g)
Neutralization by bases/alkalis
All acids react with bases to form a salt and water only. The reaction of an acid with metal oxides/hydroxides (bases) to salt and water only is called neutralization reaction.
Since no effervescence/bubbling/fizzing take place during neutralization:
- The reaction with alkalis requires a suitable indicator. The colour of the indicator changes when all the acid has reacted with the soluble solution of the alkali (metal oxides/ hydroxides).
- Excess of the base is added to ensure all the acid reacts. The excess acid is then filtered off.
Experiment 1: reaction of alkali with mineral acids.
- Place about 5cm3 of dilute hydrochloric acid in a boiling tube.
- Add one drop of phenolphthalein indicator.
- Using a dropper/teat pipette, add dilute sodium hydroxide dropwise until there is a colour change.
- Repeat the procedure with dilute sulphuric (VI) acid instead of hydrochloric acid.
- Repeat the procedure with potassium hydroxide instead of sodium hydroxide.
Sample observation:
Colour of phenolphthalein change from colourless to pink in all cases.
Explanation
- Bases/alkalis neutralize acids.
- Acids and bases/alkalis are colourless.
- A suitable indicator like phenolphthalein change colour to pink, when all the acid has been neutralized by the bases/alkalis.
- Phenolphthalein change colour from pink, to colourless when all the bases/alkalis has been neutralized by the acid.
Chemical equation
- Sodium oxide + Hydrochloric acid → Sodium chloride + Water
Na2O(s) + HCl(aq) → NaCl(aq) + H2O(l) - Potassium oxide + Hydrochloric acid → Potassium chloride + Water
K2O(s) + HCl(aq)→ KCl(aq) + H2O(l) - Sodium hydroxide + Hydrochloric acid → Sodium chloride + Water
NaOH(s) + HCl(aq) → NaCl(aq) + H2O(l) - Ammonia solution + Hydrochloric acid → Ammonium chloride + Water
NH4OH(s) + HCl(aq) → NH4Cl(aq) + H2O(l) - Potassium hydroxide + Hydrochloric acid → Potassium chloride + Water
KOH(s) + HCl(aq) → KCl(aq) + H2O(l) - Sodium hydroxide + sulphuric (VI)acid →Sodium sulphate(VI) + Water
2NaOH(s) + H2SO4(aq) → Na2SO4(aq) + 2H2O(l) - Potassium hydroxide + sulphuric (VI) acid → Potassium sulphate (VI) + Water
2KOH(s) + H2SO4(aq) → K2SO4(aq) + 2H2O(l) - Ammonia solution + sulphuric (VI) acid → Ammonium sulphate (VI) + Water
2NH4OH(s) + H2SO4(aq) → (NH4)2SO4 (aq) + 2H2O(l) - Magnesium hydroxide + sulphuric (VI) acid → Magnesium sulphate (VI) + Water
Mg(OH)2(s) + H2SO4(aq) → MgSO4(aq) + 2H2O(l) - Magnesium hydroxide + Hydrochloric acid → Magnesium chloride + Water
Mg(OH)2(s) + HCl(aq) → MgCl2(aq) + 2H2O(l)
Download ACIDS, BASES AND INDICATORS - Chemistry Notes Form 1.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

