Questions
- State two factors that should be controlled in manufacturing a cylindrical container of uniform thickness, which should normally be in a standing position.
- A butcher has a beam balance and masses 0.5 kg and 2 kg. How would he measure 1.5 kg of meat on the balance at once?
- The number of molecules in 18cm3 of a liquid is 6 x 1023. Assuming that the diameter of the molecules is equivalent to the side of a cube having the same length as the molecule. Determine the diameter of the molecule.
- Determine the density in kg/m3 of a solid whose mass is 40g and whose dimensions in cm are 30 x 4 x 3
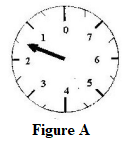
- Record as accurately as possible the masses indicated by the pointer in figure A.
- The figure below shows the reading on a burette after 55 drops of a liquid have been used.
If the initial reading was at 0cm mark, determine the volume of one drop. (2 marks) - The figure below shows the change in volume of water in a measuring cylinder when an irregular solid is immersed in it.
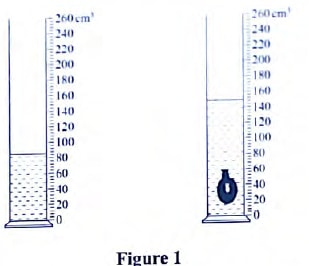
Given that the mass of the solid is 567g, determine the density of the solid in gcm-3. (Give your answer correct to 2 decimal places. - A thin wire was wound 30 times closely over a boiling tube. The total length of the windings was found to be 9.3 mm. Calculate the radius of the wire.
-
- Given that a kilogram of copper contains about 1025 atoms and that density of copper is about 9000kg/m3. Estimate the diameter of the copper atom?
- State the assumption made in (9a) above.
- A drop of oil of volume 1.0 x 10-3 spreads out on clean water surface to a film of area 10cm2. Calculate the thickness of the film.
- A small drop of oil has a volume of 5 x 10-8 m3. When it is put on the surface of some clean water, it forms a circular film of 0.1 m2 in area;
- What is the size of a molecule of oil?
- State two assumptions you make in your calculations?
- The density of concentrated Sulphuric acid is 1.8gcm-3. Calculate the volume of 3.6kg of the acid.
- 1600 cm3 of fresh water of density 1 g/cm3 are mixed with 1400cm3 of seawater of density 1.25g/cm3. Determine the density of the mixture.
- The figure shows a measuring cylinder which contains water initially at level A. A solid mass 11g is immersed in the water, the level rises to B.
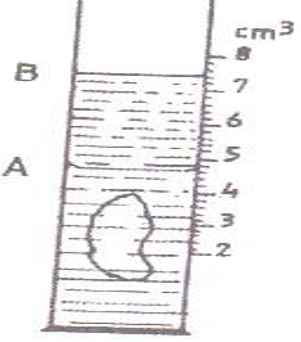
Determine the density of the solid. (Give your answer to 1 decimal point)
Answers
- height, base area
- Put 0.5kg mass together with meat and balance them against the 2 kg.
- Volume of one molecule = 18 = 3x 10-23 cm
6x1023
X3 = 3x 10-23 cm3
X = 3.11 x 10 -8 cm3 - d= m/v=40g/ 30 x 4 x 3cm3 = 0.1111 g/cm3
- 1.5 kg
- Vol. of 1 drop = (9/55) cm3 = 0.163cm3
- D= m/r =567/(150 - 80)
= 576 − 80
70g/cm3 - 4.1 × 10-8 M
-
- 4.06 × 10-10m
- That atoms are spherical and that mass is uniformly distributed on the atom and not in the nucleus.
- 0.001mm
-
- 5 x 10-7m
- - Atoms spherical
-Mass uniformly distributed
- 2000 cm3
- 1.12 g/cm3
- Density = mass/volume
Volume of solid = Level A - level B
= 7.4 − 4.6
= 2.8 cm3
Density = 11
2.8 cm3
= 3.9 g/cm3
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Measurement I Questions and Answers - Physics Form 1 Topical Revision.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students


