- What name is given to the process by which alcohol is formed from a carbohydrate? (1 mark)
- Explain why the solubility of ethane in water is lower than that of ethanol. (2 marks)
- Complete the nuclear equation below.
(1 mark)
- The half life of
is 8 days.
Determine the mass of 1 remaining of 50 grams decayed for 40 days. (1 mark) - Give one harmful effect of radioisotopes. (1 mark)
- A mixture contains ammonium chloride, copper (ii) oxide and sodium chloride. Describe how each of the substances can be obtained from the mixture. (3 marks)
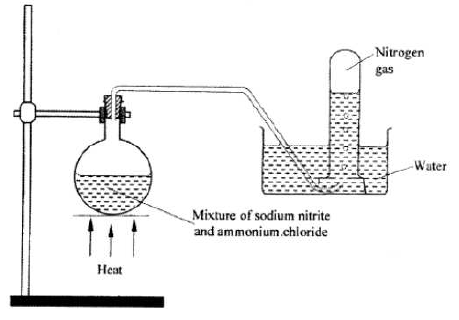
- The setup below shows how nitrogen gas is prepared in the laboratory.
- Describe how nitrogen gas is formed in the flask. (2 marks)
- Nitrogen is inert .State one use of the gas based on this property. (1 mark)
- Describe how nitrogen gas is formed in the flask. (2 marks)
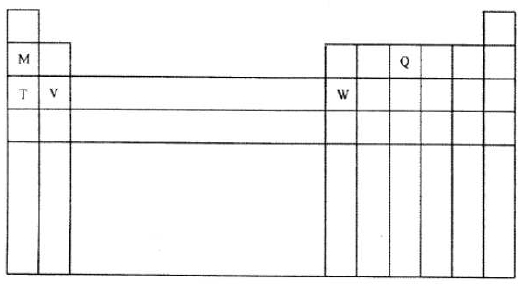
- The diagram below represents part of the periodic table . Use it to answer the questions that follow.
- Write the electronic arrangement for the stable ion formed by W.( 1 mark)
- Write an equation for the reaction between Y and Q. (1 mark)
- How do the ionization energies of the elements M and T compare?
Explain. (1 mark)
- Write the electronic arrangement for the stable ion formed by W.( 1 mark)
- A certain mass of gas occupies 0.15dm3 at 293K and 98.648.5Pa Calculate its volume at 101325Pa and 273K.(2 marks)
- When lead(II) nitrate is heated ,one of the products is a brown gas.
- Write the equation of the reaction that occurs.(1 mark)
- If 0.290dm3 of the brown gas was produced ,calculate the mass of the Lead(II) nitrate that was heated .(RFM of lead (II)nitrate =331;Molar gas volume =24dm3). (2 marks)
- What is meant by a strong acid? (1 mark)
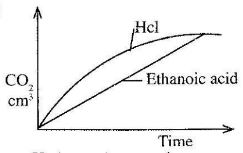
- In an experiment, 40cm3 of hydrochloric acid was reacted with excess sodium carbonate and the volume of carbon(IV)oxide produced recorded with time. In another experiment ,the same volume and concentration of ethanoic acid was also reacted with excess sodium carbonate and the volume of carbon (IV) oxide produced recorded with time.
On the grid below sketch and label the curves of the volumes if carbon (IV) oxide were plotted against time. (2 marks)
- What is meant by a strong acid? (1 mark)
- State two reasons why hydrogen is not commonly used as fuel. (2 marks)
- During a class experiment ,chlorine gas was bubbled into a solution of potassium iodide.
- State the observations made.(1 mark)
- Using an ionic equation ,explain why the reaction is redox. (2 Marks)
- Exhaust fumes of some cars contain carbon (II)oxide and other gases.
- Explain how carbon(II)oxide is formed in the internal combustion engines. (1 mark)
- Name two gases other than carbon (II) oxide that are contained in exhaust fumes and are pollutants. (2 marks)
- Explain how carbon(II)oxide is formed in the internal combustion engines. (1 mark)
- Sodium hydroxide can be prepared by the following methods I and II.
- Sodium metal -----(cold water)----> sodium hydroxide + Hydrogen
- Concentrated Sodium Chloride -----(process A)-----> sodium hydroxide +chlorine +Hydrogen
a. Name one precaution that needs to be taken inn method I. (1 mark)
b. Give the name of process A. (1 mark)
c. Give one use of sodium hydroxide. (1 mark)
- Sodium metal -----(cold water)----> sodium hydroxide + Hydrogen
- Distinguish between the terms deliquescent and efflorescent as used in chemistry. (2 marks)
- Two organic compounds P and Q decolourise acidified potassium manganate (VII) solution; but only P reacts with sodium metal to give a colorless gas. Which homologous series does compound P belong ? Give a reason. (2 marks)
- Soap dissolves in water according to the equation below;
NaSt(aq) ---------> Na(aq)+ + St- where St- is the stearate ion.- Write the formula of the scum formed when soap is used in hard water. (1 mark)
- Write the ionic equation for the reaction that occurs when sodium carbonate is used to remove hardness in water. (1 mark)
- Write the formula of the scum formed when soap is used in hard water. (1 mark)
- Ethanoic acid and ethanol react as shown in the equation below;
CH3COOH(l) + C2H5OH(l) ------><------- CH3COOC2H5(l) + H2O(l)
Other than warming, how would the state of equilibrium be established within a short time? (1 mark) - The setup below was used to prepare a gas and study some of its properties. Study it and answer the questions that follow;
- State and explain the observations made in the;
- Tube labeled A; (1 mark)
- Beaker labeled B;( 1 mark)
- State one precaution that should be taken when carrying out this experiment. (1 mark)
- State and explain the observations made in the;
- Under certain conditions , chlorine gas reacts with sodium hydroxide to form sodium hypochlorite.
- Name the conditions under which sodium hydroxide reacts with chlorine to form sodium hypochlorite. (1 mark)
- State two uses of sodium hypochlorite. (2 marks)
- 50 kg of ammonium sulphate (NH4)2SO4 and 30 kg of urea CO(NH2)2 fertilizers were applied in two equal sizes of plots A and B to enrich nitrogen content .Show by working, which plot was more enriched with nitrogen .(N=14,S=32,O=16,C=12,H=1). (3 marks)
- Describe how PH of anti acid (Actal) powder can be determined in the laboratory. (2 marks)
- Graphite is one of the allotropes of carbon.
- Name one other element which exhibits allotropy. (1 mark)
- Name one other element which exhibits allotropy. (1 mark)
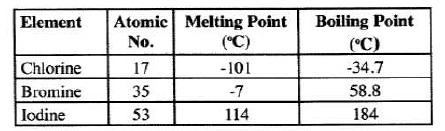
- The table below gives some properties of three elements in group (VII) of the periodic table. Study it and answer the questions that follow.
- Which element is in liquid form at room temperature? Give a reason . (1 mark)
- Explain why the boiling point of iodine is much higher than that of chlorine . (2 marks)
- Which element is in liquid form at room temperature? Give a reason . (1 mark)
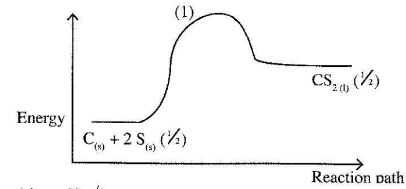
- The thermalchemical reaction between carbon and sulphur is as shown by the equation below:
C(s) + 2S(s) -----------> CS2(l) ΔH = +117.0 kJmol-1
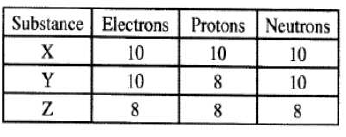
On the grid below, sketch and label the energy level diagram for the reaction. (2 marks) - The table below gives the number of electrons, protons and neutrons in substances X,Y and Z. Study It and answer the questions that follow.
- Which letter represents an ion? (1 mark)
- Which of the substances are isotopes? Give a reason. (2 marks)
- State the Gay Lussac’s law. (1 mark)
- 10cm3 of gaseous hydrocarbon, C2HX required of oxygen for complete combustion. If steam and 20cm3 of cabon (IV) oxide were produced , what is the value of X? (2 marks)
- The data given below was recorded when metal M was completely burnt in air. M is not the actual symbol of the metal,( R.A.M;=56,O=16)
Mass of empty crucible and lid =10.240g
Mass of crucible, lid and metal M=10.352g
Mass of crucible ,lid and metal oxide=10.400g- Determine the mass of:
- Metal M; (1 ½ marks)
- Oxygen. (1 ½ marks)
- Determine the empirical formula of the metal oxide. (2 marks)
- Determine the mass of:
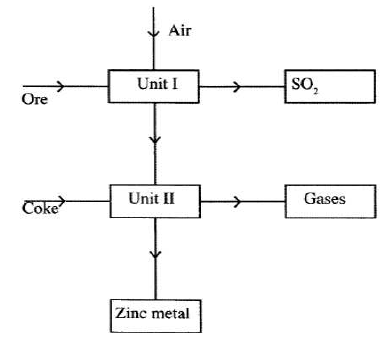
- The flowchart below shows some processes involved in the industrial extraction of Zinc metal.
- Name one ore from which zinc is extracted. (1 mark)
- Write the equation of the reaction taking place in unit II.(1 mark)
- Name two uses of Zinc metal.(1 mark)
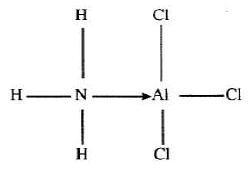
- The diagram below shows the bonding between aluminum chloride and ammonia.
- Name the types of bonds that exist in the molecule. (1 mark)
- How many electrons are used for bonding in the molecule? (1 mark)
- Name the types of bonds that exist in the molecule. (1 mark)
- Explain why the following substances conduct an electric current .
- Magnesium metal. (1 mark)
- Molten magnesium Chloride. (1 mark)
- A sample of river water is suspected to contain zinc and sulphate ions. Describe how the presence of zinc ions and sulphate ions can be established. (3 marks)
MARKING SCHEME
-
- Fermentation. (1 mark)
- Ethane remains in molecular form while ethanol forms hydrogen bonds with water. (2 marks)
-
- (1 mark)
- 50g → 25g → 12.5g → 6.25 → 3.125 → 1.5625g.
(1 mark) - Instant death, or gene mutation, induce cancer. V (1 mark)
-
- Heat the mixture to sublime the ammonium chloride. V(1 mark)
- Add water to dissolve the sodium chloride, copper (ii) oxide does not dissolve (1 mark)
- Filter and evaporate the filtrate to obtain sodium chloride. (1 mark)
-
- Oxygen is used up.
- Making of bulbs
-
- 2.8 (1 mark)
- 3V+Q2 → V3Q2 (1 mark)
OR
3Mg +N2 → Mg3N2 - T has a lower ionisation energy than M.
T has an extra energy level and hence electrons is less attracted by the positive nucleus. (1 mark
- P1V1 = P2V2
T1 T2
V2 = P1V1 x T2 = 98,658.5 x 150 x 273
T1 P2 293 x 101,325
V2 = 0.136 dm3 -
- 2Pb(NO3)2(s) → 2PbO(s) + 4NO2(g) +O2(g)
- Moles of brown gas (NO2) = 0.29/24 = 0.012 (2 marks)
Moles of lead (II) nitrate = 1/2 x 0.29/24 = 0.0067/
Mass of lead (II) nitrate = 0.006 x 331= 1.9998 g
-
- Strong acid ionises fully.(1 mark)
-
(2 marks)
-
- Hydrogen is expensive.
Hydrogen is explosive.
(2 marks)
- Hydrogen is expensive.
-
- Green colour of chlorine disappears.
Brown gas is produced or black solid is deposited. (1 mark) - Cl2(g) + I-(aq) → 2Cl(aq) + I(s)(1 mark)
Explanation: lodine oxidation state changes from - 1 to 0 hence oxidation while chlorine oxidation state changes from 0 to -1 hence reduction. (1 mark)
- Green colour of chlorine disappears.
-
- Carbon (II) oxide is formed in the internal combustion engines when fuel burns under limited oxygen, (1 mark)
- Pollutant gas - Carbon (IV) oxide, Nitrogen (IV) oxide and Sulphur (IV) oxide. (Any two)(2 marks) .
-
- Small piece of sodium metal (pea size) with alot of water.
Perform the experiment wearing goggles. (1 mark) - Electrolysis. (1 mark)
- Manufacture of soap. (1 mark)
- Small piece of sodium metal (pea size) with alot of water.
- Deliquescent substance absorbs water from the atmosphere to form a solution, while a fluorescent substance loses water of crystallisation to the atmosphere. (2 marks)
- P is in alkanol R-OH. The alkanol reacts with sodium metal to produce the colourless gas. (2 marks)
-
- Ca (st)2 or Mg (st)2
Ca2+(aq) + CO2-3(aq) → CaCO3(s) (1 mark)
OR
Mg2+ CO2-3 → MgCO3(s)
- Ca (st)2 or Mg (st)2
- By adding Conc. H.SO, as a catalyst.
-
-
- Black solid is deposited. (1 mark)
- The indicator turns red. (1 mark)
- The experiment should be done in fume chamber or in open air. (1 mark)
-
-
- Cold and dilute sodium hydroxide. (1 mark)
- Used in sterilising of water.
Used as a bleaching agent. (2 marks)
- Plot A
Percentage of Nitrogen in (NH4)2 SO4
= 21/132 x 100 = 21.2%
Amount Nitrogen in 50 kg (NH4)2SO4 = 21.2/100 x 50
= 10.6 kg
Plot B
Percentage of Nitrogen in area = 28 x 100 = 46.7%
The amount of Nitrogen in 30 kg = 46.7/100 x 30
= 14.01 kg
∴ Plot B is more enriched with nitrgen since if has higher amount of nitrogen than plot A.(3 marks) -
- Add water to dissolve the anti-acid powder.
- Add universal indicator and match the colour of solution with pH chart and read the value. (2 marks)
-
- Sulphur or phosphorus.(1 mark)
- Carbon atoms in graphite are arranged in layers of hexagons which are held by weak van der waal forces. The layers slide over each other when some force is applied on them; hence suitable in making pencil leads. (3 marks)
-
- Bromine
At room temperature (25°C), Bromine is liquid since its MP and bP is between -7 and 59 - Atomic mass of iodine is higher than that of chlorine.
Van der Waal's forces are stronger in iodine than chlorine hence iodine's bP is higher than that of chlorine. (3 marks)
- Bromine
-
-
- Y and Z
- They have the same number of protons (8) but different atomic masses. (3 marks)
-
- When gases combine together at constant temperature and pressure they do so in volumes which bear a simple ratio to each other, and to the volumes of the products if gaseous.
- C2H3 + 302 → 2CO2 + 2H2O(g)
Vol. 10 : 30 20 20
Mole ratio 1: 3 2 2
∴ X=4 (3 marks)
-
-
- Mass of oxygen = 10.400 - 10.352 = 0.048 g
- Mass of M powder = 10.352 - 10.24 -0.118 g
M O
Mole ratio 0.112 0.48
56 16
0.0020 0.0030 (1)
Simplest ratio 2 3
Empirical formula M2O3(l) (3 marks)
-
-
- Zinc blende or calamite
- ZnO(s) + C(s) → Zn(s) + CO(g)
- Use of Zinc metal:
- dry cells;
- galvanising iron sheet.
- as electrodes. (3 marks)
-
-
- Single covalent bonding 12
- Dative (coordinate) bonding
- 7 bonds x 2 = 14 electrons (2 marks)
-
-
- Mg metals have mobile delocalised electrons which carry the current
- Molten magnesium chloride has Mg2+ and C1 ions which are free to move (2 marks)
-
- Add aqueous ammonia to fill v in excess. A formation of white precipitate which dissolves in excess shows presence of zinc ions.
Add aqeous actified Barium Nitrate (3 marks) - Formation of a white precipitate shows
- Presence of sulphate ions
- Add aqueous ammonia to fill v in excess. A formation of white precipitate which dissolves in excess shows presence of zinc ions.
- Alkaline earth metals.(1 mark)
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2011 Chemistry Paper 1 Questions with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students