- The set up below was used to prepare dry hydrogen chloride gas, and investigate its effect on heated iron fillings.
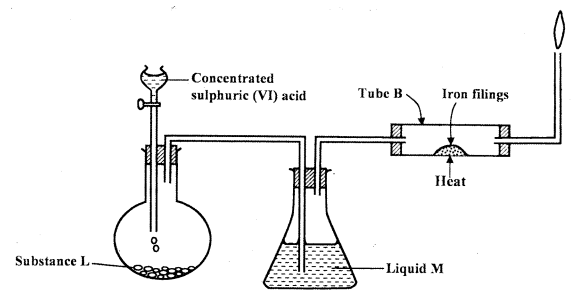
- Name substance L. (1 mark)
- Name liquid M. (1 mark)
- What will be observed in tube B? (1 mark)
- Write an equation for the reaction that occurs in tube B. (1 mark)
- Why is the gas from tube B burnt? (1 mark)
- Explain the following observations:
- A white precipitate formed when hydrogen chloride gas is passed through aqueous silver nitrate. (1 mark)
- Hydrogen chloride gas fumes in ammonia gas. (1 mark)
- State two uses of hydrogen chloride gas. (1 mark)
- Explain the following observations:
- The diagram below is a representation of an industrial process for the manufacture of a bleaching powder.
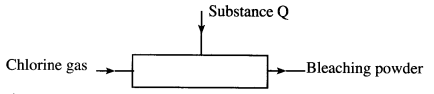
- Name substance Q. (1 mark)
- When the bleaching powder is added to water during washing, a lot of soap is used. Explain. (1 mark)
- The set up below was used to prepare dry hydrogen chloride gas, and investigate its effect on heated iron fillings.
-
- The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbols of the elements.
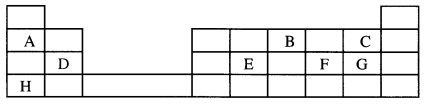
- Select the most reactive metal. Explain. (2 marks)
- Select the element that can form an ion with a charge of 3-. (1 mark)
- Select an alkaline earth metal. (1 mark)
- Which group I element has the highest first ionization energy? Explain. (2 marks)
- Element A combines with chlorine to form a chloride of A. State the most likely pH value of a solution of the chloride of A. Explain. (2 marks)
- Explain why molten calcium chloride and magnesium chloride conduct electricity while carbon tetrachloride and silicon tetrachloride do not. (2 marks)
- Under the same conditions, gaseous neon was found to diffuse faster than gaseous fluorine. Explain the observation. (F= 19.0: Ne= 20.0) (2 marks)
- The grid below represents part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbols of the elements.
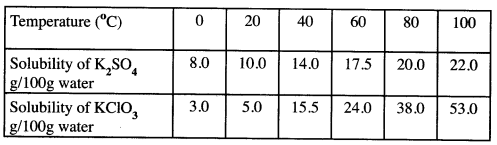
- Draw the structures of the following:
- Butan-1-ol (1 mark)
- Hexanoic acid. (1 mark)
- Study the flow chart below and answer the questions that follow.
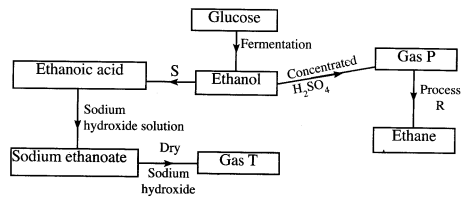
- State the conditions necessary for fermentation of glucose to take place. (1 mark)
- State one reagent that can be used to carry out process S. (1 mark)
- Identify gases: (2 marks)
P…………
T………… - How is sodium hydroxide kept dry during the reaction? (1 mark)
- Give one commercial use of process R. (1 mark)
- When one mole of ethanol is completely burnt in air, 1370 KJ of heat energy is released. Given that 1 litre of ethanol is 780 g, calculate the amount of heat energy released when 1 litre of ethanol is completely burnt. (3 marks)
(C = 12.0; H = 1.0; O = 16.0) - State two uses of ethanol other than an alcoholic drink. (2 marks)
- Draw the structures of the following:
- Other than temperature, state two factors that determine the rate of chemical reaction. (1 mark)
- A solution of hydrogen peroxide was allowed to decompose and the oxygen given off collected. After 5 minutes, substance G was added to the solution of hydrogen peroxide. The total volume of oxygen evolved was plotted against time as shown in the graph below
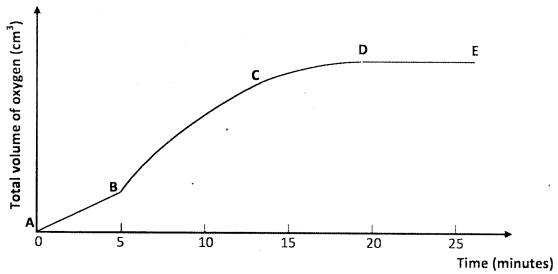
- Describe the procedure of determining the rate of reaction at minute 12. (3 marks)
- How does the production of oxygen in region AB compare to that in region BC? (2 marks)
- Write an equation to show the decomposition of hydrogen chloride. (1 mark)
- Sulphur (IV) oxide reacts with oxygen to form Sulphur (VI) oxide as shown in the equation below:
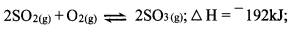
- Explain the effect of the yield SO3 of lowering the temperature for this reaction. (2 marks)
- Name one catalyst used for the reaction. (1 mark)
- Other than temperature, state two factors that determine the rate of chemical reaction. (1 mark)
- The scheme below shows some of the reactions of solution D. Study it and answer the questions that follow.
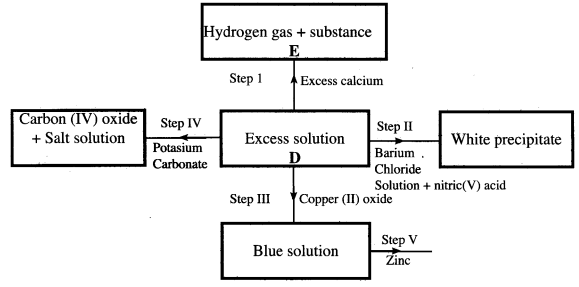
- Give a possible cation present in solution D. (1 mark)
- Write an ionic equation for the reaction in step II. (1 mark)
- What observations would be made in step V? Give a reason. (2 marks)
- Explain why the total volume of hydrogen gas produced in step I was found to be very low although calcium and solution D were in excess. (2 marks)
- State one use of substance E. (1 mark)
- Starting with sodium chloride, describe how a pure sample of lead (II) chloride can be prepared in the laboratory. (3 marks)
- State a property of anhydrous calcium chloride which makes it suitable for use as a drying agent for chlorine gas. (1 mark)
- Name another substance that can be used to dry chlorine gas. (1 mark)
- The scheme below shows some of the reactions of solution D. Study it and answer the questions that follow.
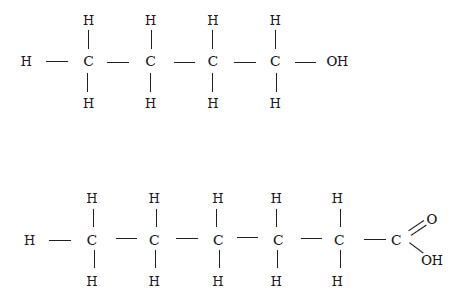
- The diagram below represents a set up of an electrolytic cell that can be used in the production of aluminium.
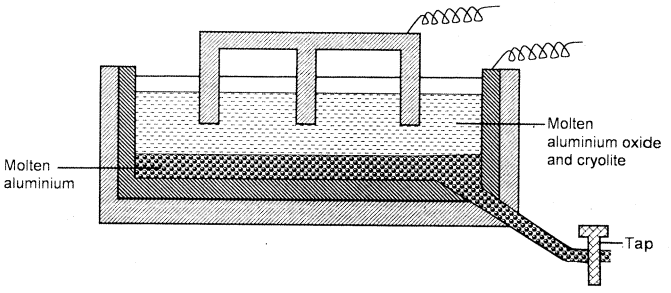
- On the diagram, label the anode. (1 mark)
- Write an equation for the reaction at the anode. (1 mark)
- Give a reason why the electrolytic process is not carried out below 9500C. (1 mark)
- Give a reason why the production of aluminium is not carried out using reduction process? (1 mark)
- Give two reasons why the aluminium ions are discharged. (2 marks)
- State two properties of duralumin that make it suitable for aircraft industry. (2 marks)
- Name two environmental effects caused by extraction of aluminium. (2 marks)
- On the diagram, label the anode. (1 mark)
-
- Dissolving of potassium nitrate in water is an endothermic process. Explain the effect of increase in temperature on the solubility of potassium nitrate. (2 marks)
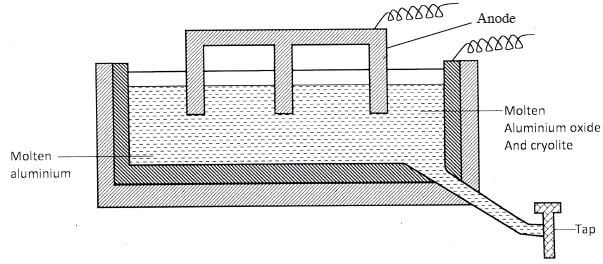
- The table below shows the solubilities of potassium sulphate and potassium chlorate (V) at different temperatures.
- Draw the solubility curves for both salts on the same axis. (Temperature on the X- axis) (3 marks)

- A solution of potassium sulphate contains 20g of the salt dissolved in 100g of water at 1000C. This solution is allowed to cool to 250C.
- At what time will the crystals first appear? (1 mark)
- What mass of crystals will be present at 250C? (1 mark)
- Which of the two salts is more soluble at 300C? (1 mark)
- Determine the concentration of potassium nitrate in moles per litre when the solubility of the two salts is the same. (K = 39.0; O = 16.0; S =32.0)
- 100g of water at 1000C contains 19g of potassium chlorate (V). Describe how a solid sample of potassium sulphate at 600C can be obtained. (2 marks)
- Draw the solubility curves for both salts on the same axis. (Temperature on the X- axis) (3 marks)
- Dissolving of potassium nitrate in water is an endothermic process. Explain the effect of increase in temperature on the solubility of potassium nitrate. (2 marks)

MARKING SCHEME
-
-
- Sodium chloride / potassium chloride /rock salt. (1 mark)
- Concentrated sulphuric (VI) acid (1 mark)
- Grey solid turns green (1 mark)
- Fe(s) + 2 HCl(g) → FeCl2(s) + H2(g). (1 mark)
- To avoid explosion. (1 mark)
-
-
- The gas reacts with silver nitrate to form insoluble silver chloride. (1 mark)
- Both gases form ammonium chloride which is white. (1 mark)
-
- To make hydrochloric acid.
- Manufacture of ammonium chloride.
- Manufacture of PVC.
- Making chloroethene /vynil chloride (1 mark)
-
-
- Q is Ca(OH)2 (aq) /calcium chloride (1 mark)
- Presence of Ca2+ which make water hard / forms scum. (1 mark)
-
-
-
- K - Has largest atomic radius / it most readily loses its outermost electron.
- B /N (1 mark)
- D / Mg (1 mark)
- A (1) It has the smallest/smaller atomic radius/ its outermost electron is more/most strongly held by nucleus.(1) (2 marks)
- PH is seven (7). The chlorides of group 1 elements are neutral salts. (2 marks)
-
- Both CaCl2 and MgCl2 have mobile ions in molten state (1) while both CCl4 and SiCl4 are molecular compounds with no mobile ions (1).
- Neon has molar mass of 20 while Flourine has a molar mass of 38 (1). Therefore Neon diffuses faster. (1) Since it has a lower molecular mass the faster the rate of diffusion.
-
-
-
 (1 mark)
(1 mark) -
- Yeast/enzymes/zymase/temperature of 35 - 40ºC. (2 marks)
- Acidified KMnO4 or K2Cr2O7 (1 mark)
- P = Ethene / C2H4 (1 mark)
T = Methane / CH4 (1 mark) - Addition of CaO or NaOH (1 mark)
- Converting oils into fats. (1 mark)
Manufacture of mangarine /hardening oils.
- CH3 CH2 OH →1370 kJ(1)
R.M.M. of CH3CH2OH 46(1/2)
1 litre (780g) = 1370 × 780 (1)
46
= 23,230.43 kj (1/2) - Fuel
Solvent
Anti-septic
Manufacture of synthetic fibres
Manufacture of gasohol
Manufacture of ethanoic acid/vinegar
Used in themometers
Manufacture of other organic compounds.
Any 2 (2 marks)
-
-
-
- Pressure
- Concentration
- Catalyst
- Particle size/surface area
- Light intensity (1 mark)
-
- Draw a tangent to the graph at 12 min. (1)
Determine change in volume(1/2) /Calculate gradient.
Determine change in time.(1/2)
Divide change in volume by change in time (1) - AB Low production of gas(1) while BC the rate is very high because catalyst B was added. (1)
- 2H2O2(aq) O2(g)+ 2H2O(l)(1)
- Draw a tangent to the graph at 12 min. (1)
-
- Lowering the temperature shifts the equilibrium to the right /favours the forward reaction. (1) Hence more SO3 will be produced. (1)
- Platinum or Vanadium pentoxide/vanadium (v) oxide / V2O5/ platinised asbestos any (1 mark)
-
-
-
- Cation present in solution D is H+ (1 mark)
- Ba2+(aq) + SO42-(aq) → BaSo4(s)
- Zinc disappears /zinc dissolves.
Blue colour disappears and brown solid is deposited.(1)
Zinc being more reactive than copper displaces /Cu2+ ions from solution, copper which is brown is formed.(1)
Apparatus feels warm/reaction is exothermic. - The reaction forms CaSO4 which is insoluble(1) the insoluble CaSO4 coats the surface of calcium preventing further reaction. (1)
- Making plaster of paris / making plaster. (1)
- Dissolve sodium chloride in distilled water. (1/2) Add aqueous lead nitrate. (1)
Filter the mixture, (1/2) wash residue with distilled water. (1/2)
Dry residue in oven at controlled temperatures/ between filter papers.( 21 ) -
- It absorbs moisture/water. / deliquescent / hygroscopic (1)
- Conc. H2SO4(1) / H2SO4(l) / concentrated sulphuric (VI) acid/
-
-
- On the diagram (1)
- 2O2-(l) → O2(g) + 4e (1)
- Below 950 ºC, the electrolyte is not in molten state.(1)
- Aluminium is more reactive than carbon (coke)(1) therefore the reduction process is not possible / carbon / carbon (II) oxide / coke cannot reduce Al2O3.
-
- Aluminium is less reactive than Sodium (1) therefore: It is preferentially discharged.
- Al3+ ions are in higher concentration than Na+.(1)
-
- Global warming due to production of CO2 / F2 pollution. (1)
- Creation of gullies during excavation. (1)
-
- Light (1)
- Strong (1)
-
- Solvent molecules move further apart hence more solid particles dissolve / creating more space for solid particles(1)
therefore:The solubility increases.(1) -
- (3 marks)
-
- Value read from the graph (1)
- 10.5 g (value read) (1)
- Salt identified from the graph (1)
- Solubility of K2SO4 = 12.8 (1/2) g/100 g
Mass of K2SO4 in 100 cm3 =12.8 × 1000 (1/2)
100
= 128 g(1/2)
Molar mass of K2SO4 = 174g (1/2)
Conc of K2SO4 = 128 (1/2) = 0.7356M (1/2)
174 - Filter crystals of K2SO4(1/2)
Dry between filter papers. (1/2)
- Solvent molecules move further apart hence more solid particles dissolve / creating more space for solid particles(1)
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2014 Chemistry Paper 2 Questions with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

