SECTION A: BIOLOGY (34 marks)
Answer all the questions in the spaces provided.
-
- Name two abiotic factors in a soil ecosystem.(2 mark)
- Give an example of parasitism in wood-land ecosystem. (2 marks)
-
- State two reasons for sexual reproduction in animals.(2 marks)
- Distinguish between fertilization and ovulation. (1 mark)
- Name the hormone that is responsible for the growth of beards.(1 mark)
-
- State one role played by both bacteria and fungi in nitrogen cycle.(1 mark)
- Give one adaptation of hydrophyte roots.(1 mark)
- Name a method that can be used to control air pollutants from a factory.(1 mark)
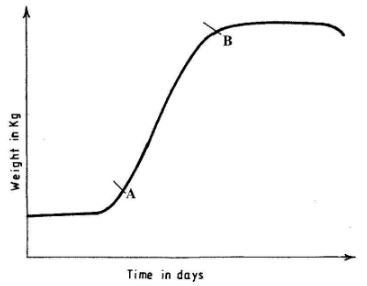
- The graph below illustrates growth in an animal.(1 mark)
- Explain what happens between points A and B.(2 marks)
- What is meant by the term seed dormancy?(1 mark)
- Explain what happens when the shoot apex of a plant is removed.(2 marks)
-
- Explain how the sex of a child is determined by a man.(2 marks)
- A heterozygous red-eyed fruit fly was crossed with a recessive white-eyed one. Using 'R' to represent the dominant gene, work out the cross to show the offspring,(3 marks)
- State the phenotypic ratio of the offspring.(1 mark)
- State two ways in which sexually transmitted infections can be avoided.(2 mark)
- Explain the importance of strictly following the prescription given when taking medicine.(2 mark)
-
- Name two supportive tissues in dicotyledonous plants.(1 mark)
- Give an example of a hinge joint in humans.(1 mark)
-
- State two differences between the endocrine and the nervous systems.(2 marks)
- State one function for each of the following structures in the mammalian ear.
- pinna(1 mark)
- ossicles(1 mark)
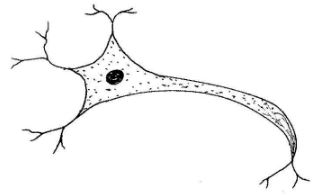
- The diagram below represents a type of neurone found in animals.
- Name the neurone.(1 mark)
- Give the reason for your answer in (a) above.(1 mark)
SECTION B: CHEMISTRY (33 marks)
Answer all the questions in this section in the spaces provided.
- Under the same conditions of temperature, volume and pressure, which one of the following gases, steam, carbon(IV) oxide, nitrogen, oxygen and ammonia would diffuse the slowest? Explain. (R.A.M: H=1, C = 12, N = 14 and 0 = 16). (2 marks)
- Hydrogen combines with oxygen to form water. How many moles of hydrogen atoms does 3.6g of water contain?(2 marks) (H=I, O = 16)
- One of the methods used for large scale production of ethanol is by fermentation of cane sugar.
- What is meant by fermentation?(2 marks)
- The ethanol obtained is about 4 -8% concentrated. How can this concentration be increased?(1 mark)
- Other than being used as an alcoholic beverage, state one use of ethanol. (1 mark)
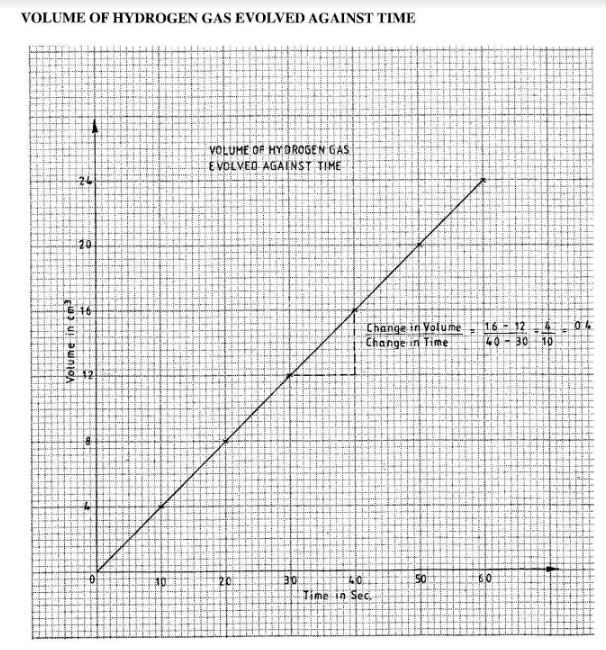
- Dilute hydrochloric acid was reacted with magnesium ribbon and the volume of hydrogen gas evolved was measured at 10 seconds interval up to 60 seconds. The volumes were recorded as in the table below.
Time in seconds 0 10 20 30 40 50 60 Volume of hydrogen in cm3 0 4 8 12 16 20 24 - Using the grid provided draw a graph of volume of hydrogen (vertical axis) against time (horizontal axis). (3 marks)
- Using the graph, determine the average rate of production of hydrogen. (1 mark)
- In the manufacture of ammonia using the haber process, nitrogen and hydrogen gases react as shown in the equation below.
- State the source of hydrogen gas.(1 mark)
- Explain the effect of increasing pressure on the yield of ammonia.(2 marks)
- Give one use of nitrogen (IV) oxide.(1 mark)
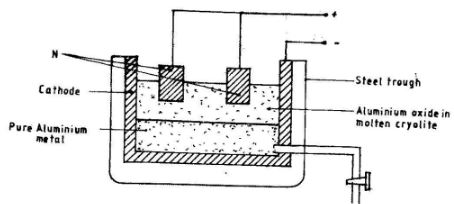
- The diagram below represents electrolytic production of aluminium metal. Study it and answer the questions that follows.
- Why is aluminium extracted by electrolysis and not reduction?(1 mark)
- Why is N replaced regularly?(1 mark)
- State one use of the molten cryolite in the above process.(1 mark)
- State two properties that makes aluminium metal to be widely used in electric cables.(2 marks)
-
- What is meant by the term molar solution?(1 mark)
- Calculate the molarity of a solution containing 6.24g of hydrated copper (II) sulphate crystals in 250cm3 of solution. (R.FM CuSO45H20 = 249.5).(2 marks)
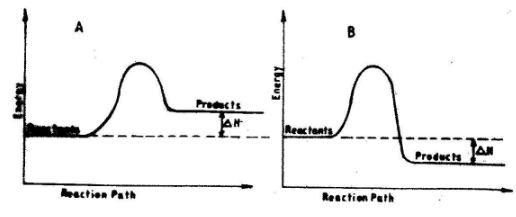
- The energy level diagrams below shows the enthalpy changes for reactions A and B.
- State with a reason the type of reactions represented by A and B.(2 marks)
- State two properties of carbon (IV) oxide that make it qualify to be used as a fire extinguisher.(2 marks)
- Give one advantage of using biogas as a fuel instead of firewood.(1 mark)
- State Graham's law of diffusion.(1 mark)
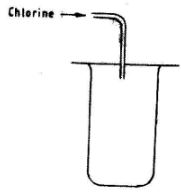
- Chlorine gas is collected using the method shown in the figure below.
- Name the method of collection.(1 mark)
- Which property of chlorine enables it to be collected using the method shown above?(1 mark)
- Give two uses of chlorine gas.(1 mark)
SECTION C - PHYSICS (33 marks)
Answer ALL the questions in this section in the spaces provided.
-
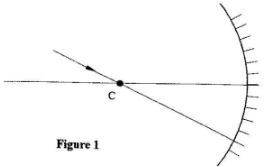
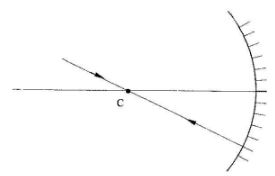
- Figure 1 shows a ray of light incident on a converging mirror through the centre of a curvature C.
Complete the diagram to show the reflected ray.(1 mark)
- Figure 1 shows a ray of light incident on a converging mirror through the centre of a curvature C.
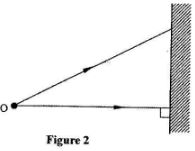
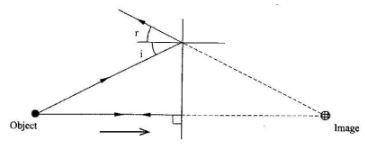
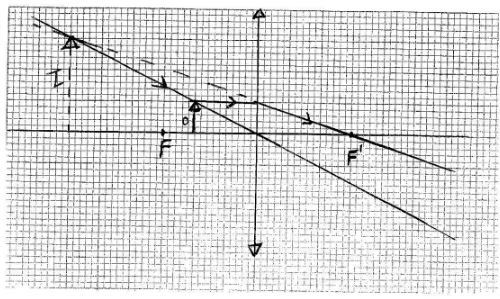
- Figure 2 shows two rays from a point object incident on a plane mirror
Complete the diagram to show the position of the image.(2 marks) - A plastic ruler becomes negatively charged when rubbed with a piece of cloth. Explain how the ruler acquires the charge.(2 marks)
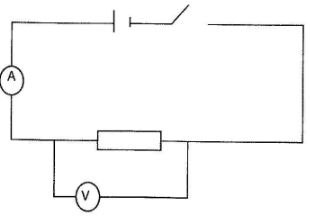
- Given a voltmeter, an ammeter, a resistor, a switch and a cell, draw a circuit diagram that may be used to measure the voltage across the resistor and the current through it. (2 marks)
- Draw a labelled diagram showing how two bar magnets should be stored using keepers(1 mark)
- Figure 3 shows a graph of displacement against distance for a wave in a medium.
Determine:- the amplitude of the wave,(1 mark)
- the wavelength of the wave.(1 mark)
- A girl standing in front of a wall claps her hands. She hears the echo 0.4 seconds later, Determine the distance between the girl and the wall. (take speed of sound in air to be 340ms) (3 marks)
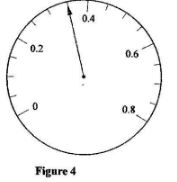
- Figure 4 shows the face of an ammeter indicating a current.
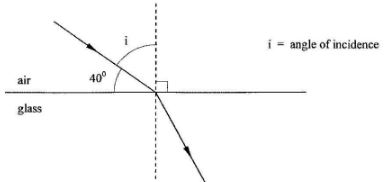
State the value of the current indicated.(1 mark) - Figure 5 shows a ray of light travelling from air to glass.
- Indicate on the diagram the angle of incidence of the ray,(1 mark)
- Given that the refractive index for a ray travelling from air to glass is 1.5, determine the angle of refraction.(2 marks)
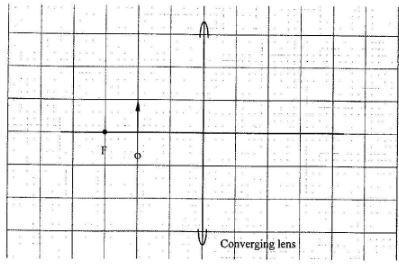
- Figure 6 shows an object O placed in front of a converging lens whose principal focus is F.
Draw a ray diagram to locate the image formed. (3 marks) - An electric cooker has a resistance of 202. Determine the power it dissipates when connected to a 240V mains supply(3 marks)
-
- State the purpose of creating a vacuum in a cathode ray tube.(1 mark)
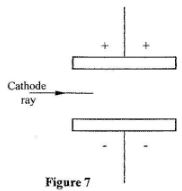
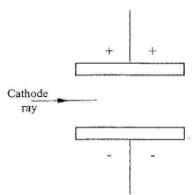
- Figure 7 shows a horizontal cathode ray entering an electric field between two charged plates.
Complete the diagram to show the path of the ray in the electric field.(1 mark)
- State how the produced X-rays change, when the anode potential of the X-ray tube is increased (1 mark)
- Name two medical uses of radioactive radiations. (2 marks)
-
- State one difference between a semiconductor and a conductor. (1 mark)
- Draw a circuit diagram to show a diode connected in the reverse bias mode(1 mark)
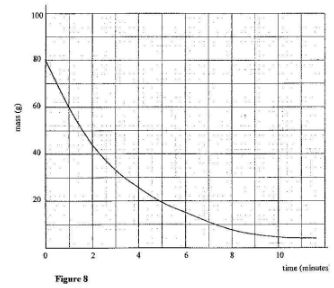
- Figure 8 shows a graph of mass against time for a radioactive sample.
Use the graph to determine the half life of the sample.(2 marks)
MARKING SCHEME
-
- Air; moisture;salinity; PH; temperature; any two.(2x 1 )
- Ticks on buffaloes/tse-tse flies on water bucks/ fieas on monkeys; accept any other correct relationship.(1 x 2)(2 marks)
-
- Pass hereditary characteristics to future generations;
Perpetuate the species/survival;
Continuation of life
Natural selection/enhances variations; any two.
(2 x 1) (2 marks) - Fertilization - fusion of sperm and egg to form zygote while ovulation is the release of the ovum from the ovary into the fallopian tube; (mark as a whole)(1 mark)
- Testosterone;(1 mark)
- Pass hereditary characteristics to future generations;
-
- Decomposition/decay;(1 mark)
- Long/fibrous roots; for anchorage in /absorption of nutrients from water.(1 mark)
- By converting pollutants to harmless substances; accept recycling.(1 mark)
-
- Fast/rapid/exponential growth; many cells are dividing/optimum environmental conditions;(2 marks)
- A period during which a viable seed undergoes no growth;(1 mark)
- Lateral buds sprout; due to reduced supply of auxins;(2 marks)
- A period during which a seed does not germinate even if in favourable conditions.
-
- The man produces two types of sperms one containing X chromosomes and the other Y chromosomes; while the woman produces ova with only X chromosomes, If the X sperm fertilizes the ovum the result is a girl and if the Y sperm fertilizes the ovum the result is a boy; (maximum two marks).(2 marks)
- Parental Rr rr
Genoty
Meiosis
Gametes
Fertilization
F1 Genotype
F1 Phenotype Red-eyed White-eyed
F1 Phenotypic ratio 1 1
(3 marks)
Red eyed : White eyed - Phenotypic ratio 1 1 (1 mark)
- Avoid sharing contaminated equipments and clothing; Abstinence; protected sex; Being faithful to uninfected partner. (any two)(2 marks)
- Completely cure the disease;
Prevent resistance to the medicine;
Prevent overdoes/organ damage/death;
Avoid weakening of immune system;
(any two)
(2 marks) -
- Parenchyma/ sclerenchyma/
Xylem/collenchyma;/
(any correct two) (1 mark) - Knee joint/elbow joint;
- Parenchyma/ sclerenchyma/
-
- Nervous system Endocrine system
message as electrical impulses message in form of chemicals
Transmitted within nerves transmitted in blood
Rapid slow
Effects specific Effects generalised/diffused,
(any two correctly contrasted) (2 marks) -
- pinna: collect sound waves/direct sound waves into the external auditory canal; (1 mark)
- ossicles: Amplify sound vibrations/transmit sound vibrations to the inner ears; (1 mark)
- Nervous system Endocrine system
-
- Relay/connect/intermediate neurone;(1 mark)
- Presence of many dendrites from the cell body in all directions/has no myelin sheath;(1 mark)
-
RFM
H2O 2(1) + 16 = 18
CO2 12+2(16) = 44
N2 (14)2 -- 28
O2 (16)2 = 32
NH3 14+3(1) = 17
CO2 will difuse with slowest rate (1)
Since it has the largest molecular mass (44g) (1/2) (2 marks) - H2O 2(1) + 16 = 18
H atoms has 2 moles in 18g (1/2)
x moles in 3.6g
∴ 2 x 3.6 = 18x
x = 2 x 3.6 = 0.4 moles of H atoms.
18
(2 marks) -
- Fermentation is a process in which cane sugar substances is converted into ethanol and carbon (IV) oxide (1) in absence of oxygen. (1)
- Distillation. (1)(4 marks)
- Fuel, solvent, pharmaceutical, Chromatography, cosmetics. (1)
Preparation of Esters, Ethene, Ethanoic
As an antiseptic
(Any one)
-
- Plotting (1)
scale (1)
curve (1)
(If graph is inverted maximum 2) - 0.4cm3 per second (1)
(4 marks)
- Plotting (1)
-
- Cracking Hydrocarbons/electrolysis of acidulated water/electrolysis of Brine.(any one)
- Increasing pressure increases yield of ammonia. (1)
4 volumes of reactants against 2 volumes of products; hence increase in pressure raises the volumes of products. (1) - Manufacture of nitric(V) acid explosives, nylon & plastics. (any one) (1).(4 marks)
-
- Aluminium is a reactive metal. (1)
- N is made from carbon electrodes which react with oxygen evolved, forming CO2(1)
Hence requires to be replaced regularly. - Lower the melting point of bauxite. (1)
- It has a low density (1) and a good conductor of electricity (1)(5 marks)
-
- Is a solution that contains one mole of a substance per litre. (1)
- 6.24g of CuSO4.5H2O contains 6.24/249.5 = 0.025 moles
250cm3 of solution contains 0.025mole
1000 cm3
contains 1000/250 x 0.025
= 4 x 0.025
= 0.1 moles
Molarity of the solution is 0.1M (3 marks)
-
- A endothermic reaction. 1/2 heat is absorbed. 1/2
B exothermic reaction heat is evolved. - It does not support burning (1)
It is denser than air (1) - biogas is clean, (no smoke);firewood produce more smoke. (any 1)
conservation of forest.
heat value of biogas is high.
No residue in biogas after burning while in firewood ash remains. (5 marks)
- A endothermic reaction. 1/2 heat is absorbed. 1/2
- Under the same conditions of temperature and pressure, the rate of diffusion of a gas is inversely proportional to the square root of its density. (1)(1 mark)
-
- Upward displacement of air or downward delivery.(1)
- Chlorine is denser than air,
- Water treatment/treatment of sewerage.
Manufacture of PVC.
CFCs/CCI4/CHCI3, any two V 1 mark
As a bleaching agent.
(3 marks)
-
-
- On rubbing electrons leave the cloth and accumulate on the plastic ruler.(1 mark)
The ruler becomes negatively charged while the cloth is left with a net positive charge.(1 mark) -
-
- a = 10cm (1 mark)
- λ = 20cm(1 mark)
- Distance = speed x time
= 340 x 0.4/2 (1 mark)
= 68m - I = 0.35A
-
-
- n = sin i
sin r
1.5= sin (90 - 40°)
sin r
sin r= sin. 50 = 0.756 = 0.5106
1.5 1.5
r~30.71° (1 mark)
-
-
- P= VI
= V2
R
=240 x 240 =
20
= 2880 watts -
- to minimize collisions between cathode rays and air molecules;
to minimise reduction of KE of the cathode rays;
to reduce ionization of air molecules. -
- to minimize collisions between cathode rays and air molecules;
- higher frequency X -rays are produced more penetrating X-rays/hard x-rays/Higher energy/ X-rays/High quality x-rays.
- used in treatment of cancer;
used to sterilize medical equipment;
used in detecting abnormal tissue in people.
(any two 1 mark each) -
- conduction in semiconductors is by electrons and holes while in conductors it is by electrons.
conductivity of a semiconductor increases with increase in temperature while that of a conductor decreases with increase in temperature. (any 1 correct) (1 mark) -
- conduction in semiconductors is by electrons and holes while in conductors it is by electrons.
- From the graph,
Mass at t = 0 is 80g;
time when mass is 40g; is 2.25 minutes.
∴ half life period is 2.25 minutes;
or 1 mark for reading off values of mass reducing by half .
1 mark for the time taken for mass to reduce to half the original.
Download KCSE 2011 General Science Paper 2 Questions with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students