-
- Draw the structural formula for all the isomers of C2H3Cl3.( 2 marks)
- Describe two chemical tests that can be used to distinguish between ethane and ethane. (4 marks)
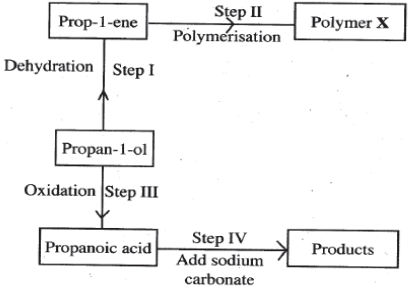
- The following scheme represents various reactions starting with propan-1-ol . Use it to answer the questions that follow.
- Name one substance that can be used in step 1. (1 mark)
- Give the general formula of X. (1 mark)
- Write the equation for the reaction in step IV. (1 mark)
- Calculate the mass of propan-1-ol which when burnt completely in air at room temperature and pressure would produce 18 dm3 of gas. (C=12.0; O=16.0; H=1.0; Molar gas Volume =24 dm3 ).
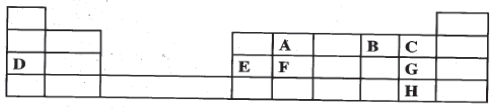
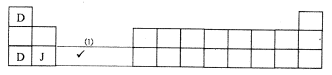
- The grid below is part of the periodic table. Use it to answer the questions that follow. (The letters that follow are not the actual symbols of the elements).
- Which is the most reactive non-metallic element shown n the table? Explain. (2 marks)
-
- Write the formula of the compound formed when element A reacts with element B. (1 mark)
- Name the bond type in the compound formed in b(i) above. ( 1 mark)
-
- What is the name give to the group of elements where C,G and H belong?(1 mark)
- Write an equation for the reaction that occurs when C in gaseous form is passed through a solution containing ions of element H. (1 mark)
- The melting points of elements F and G are 14100C and -1010C respectively. In terms of structure and bonding, explain why there is a large difference in the melting points of F and G. ( 2 marks)
- D formed two oxides. Write the formula of each of the two oxides. (1 mark)
- J is an element that belongs to the 3rd period of the periodic table and is a member of the alkaline earth elements; Show the position of J in the grid. (1mark)
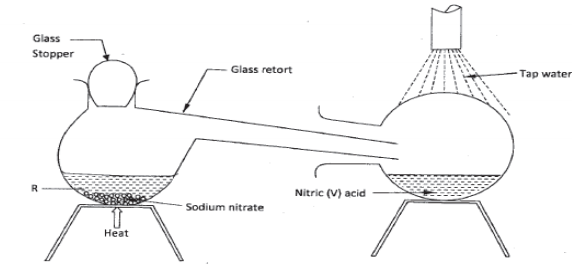
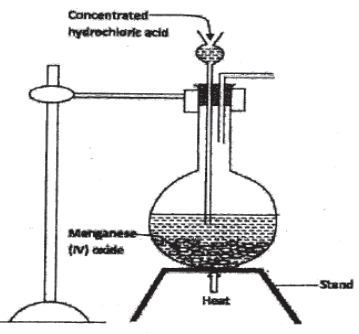
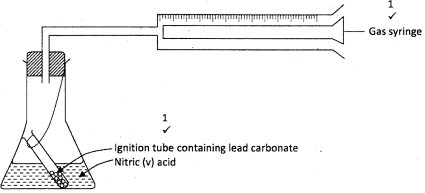
- In the laboratory, small quantities of nitric (V) acid can be generated using the following setup. Study it and answer the questions that follow.
-
- Give the name of substance R. (1 mark)
- Name the other substance that can be used in place of sodium nitrate.(1 mark)
- What is the purpose of using tap water in the set up above? ( 1 mark)
- Explain the following
- it is not advisable to use stopper made of rubber in the setup. (1 mark)
- the reaction between copper metal wit 50% nitric acid in an open test-tube produces brown fumes .(1 mark)
-
- Nitrogen is one of the reactants used in the production of ammonia, name two sources of the other reactant (2 marks)
- A factory uses nitric (v) acid and ammonia gas in the preparation of a fertilizer. If the daily production of the fertilizer is 4800 kg; calculate the mass of ammonia used in kg.(N=14.0; O = 16.0; H=1.0) (3 marks)
- State two other uses of nitric (V) acid other than the production of fertilizers. (2 marks)
-
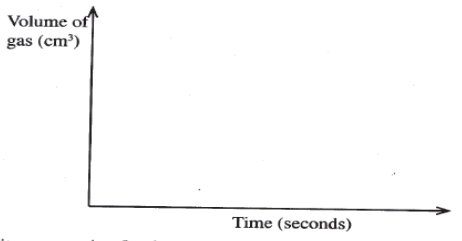
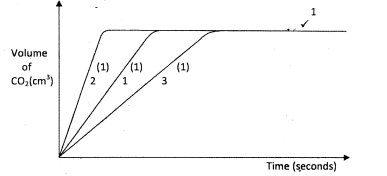
- The factors which affect the rate of reaction between lead carbonate and dilute nitric (V) acid were investigated by carrying out three experiments:
- Other than concentration, name the factor that was investigated in the experiments. (1 mark)
- For each experiment, the same volume of acid (excess) and mass of lead carbonate were used and the volume of gas liberated measured with time.
- Draw a set up that can be used to investigate the rate of reaction for one of the experiments. (3 marks)
- On the grid provided, sketch the curves obtained when the volume of gas produced was plotted against time for each of the three experiments and label each as 1,2 or3. (4 marks)
- Write an equation for the reaction that took place. ( 1 mark)
- If the experiments were carried out using dilute hydrochloric acid in place of nitric (V) acid, the reaction would slow down and eventually stop. Explain these observations. (2 marks)
- A solution of bromine gas in water is an example of a chemical reaction in a state of balance. The reaction involved is represented by the equation below.
State and explain the observation made when hydrochloric acid is added in the mixture at equilibrium.(2 marks)
-
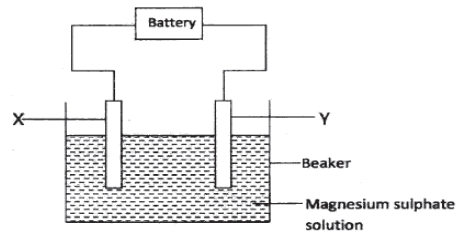
- The set up below was used to investigate the products formed at the electrodes during electrolysis of aqueous magnesium sulphate. Use it to answer the questions that follow.
- During the electrolysis, hydrogen gas was formed at electrode Y .Identify the anode . Give a reason for your answer. (2 marks)
- Write the equation for the reaction which takes place at electrode X.( 1 mark)
- Why is the concentration of magnesium sulphate expected to increase during electrolysis?
(2 marks) - What will be observed if red and blue litmus papers were dipped into the solution after electrolysis? (2 marks)
- During electrolysis of magnesium sulphate, a current of 0.3 A was passed for 30 minutes. Calculate the volume of gas produced at the anode. (Molar gas volume =24dm3; 1 Faraday =96,500 C)
(3 marks) - State two applications of electrolysis. (1 mark)
- The set up below was used to investigate the products formed at the electrodes during electrolysis of aqueous magnesium sulphate. Use it to answer the questions that follow.
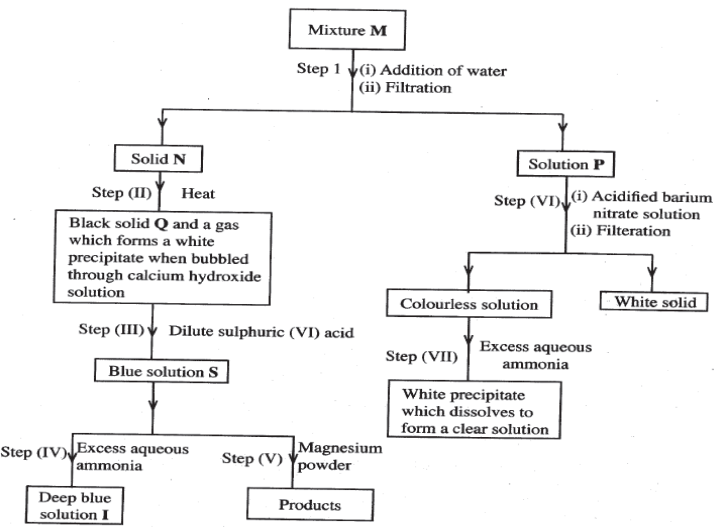
- The flowchart below shows a sequence of reactions involving a mixture of two salts , mixture M. Study it and answer the questions that follow.
- Write the formula of the following:
- Anion in solid Q. (1 mark)
- The two salts present in mixture M. ( 2 marks)
- Write an ionic equation for the reaction in step (VI). (1 mark)
- State and explain the observations made in step (V) ( 3 marks)
-
- Starting with lead (II) oxide, describe how a pure solid sample of lead sulphate can be prepared in the laboratory. ( 2 marks)
- How can one determine whether the lead sulphate prepared is pure? (2 marks)
- Write the formula of the following:
-
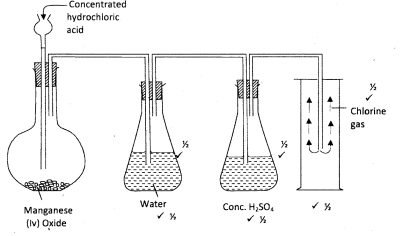
- The diagram below is part of a set up used to prepare and collect dry chlorine gas.
- Complete the diagram to show how a dry sample of chlorine gas can be collected (3 marks)
- Name another substance and condition that can be used instead of manganese (IV) oxide. (1 mark)
- Write an equation for each if the following
- Chlorine gas reacting with iron (1 mark)
- Chlorine gas reacting with hot concentrated sodium hydroxide solution. ( 1 mark)
- An oxide of chlorine of mass 1.83g was found to contain 1.12g of oxygen. Determine the empirical formula of the oxide (O=16.0; Cl =35.5) . (3 marks)
- Other than the manufacture of weed killers, name two uses of chlorine. (2 marks)
- The diagram below is part of a set up used to prepare and collect dry chlorine gas.

MARKING SCHEME
-
-
✓2
-
- Bubble each through acidified potassium dichoromate (VI) ✓1 with ethene the solution changes from orange to green ✓½ while in ethane the solution remains orange. ✓½
- Bubble each through acidified Potassium manganate(VII) ✓1 with ethene the solution changes from purple to ✓½ colourless while in ethane the solution remains purple. ✓½
- Add a few drops of bromine water ✓1 with ethene the solution changes from orange/brown ✓½ to colourless, while in ethane the solution remains orange/ brown.✓½
- Ethene burns with yellow or sooty flame.
Ethane burns with non-luminous or blue flame.
Choose any 2
-
- Concentrated sulphuric (VI) acid or Al2O3 or H3PO4✓1
-
✓1
- 2CH3CH2COOH + Na2CO3 → 2CH3CH2COONa(aq) + CO2(g) + H2O (1)
- 2CH3CH2CH2OH + 9O2 → 6CO2 +8H2O (1)
Moles of CO2 = 18/24 (½) - Moles of CH3CH2CH2OH = 18/24 × 1/3 (½)
R.M.M. of CH3CH2CH2OH = 60 (½)
Mass = 18/24 x 1/3 × 60 = 15g (½)
-
-
- C ✓1 has the smallest atomic radius and is the most electronegative element in the periodic table. ✓1 as one traverses the period number of protons increases hence the nuclear attraction increases.
-
- AB2/AB/CO2 or CO ✓1
- Covalent bond ✓1
-
- Halogens ✓1
- C2(g) + 2H−(aq) → 2CH+(aq) + H2(g) ✓1
- F has a giant atomic ✓½ structure with strong ✓½ covalent bond which is strong and difficult to break hence high melting point. While G although it exhibits covalent bond it has simple ✓½ molecular structure with weak van der Waal's forces between its molecules ✓½ hence the low melting point.
- D2O✓½ and D2O2✓½
-
-
-
- Concentrated ✓1 sulphuric (VI) acid.
- Potassium nitrate ✓1
- To condense the fumes or vapour of nitric (V) acid into liquid ✓1
-
- Nitric acid (V) will corrode the rubber ✓1
- The reaction produces nitrogen monoxide (colourless) ✓½ which is oxidised by oxygen from the air to form nitrogen(IV) oxide. ✓½
-
-
- Water
- Alkanes
- Biogas
- Water gas
any 2
- NH3 + HNO3 → NH4NO3 ✓1
Mass of NH4NO3= 80 ✓½
either
Moles of NH4NO3 = 4800 x 103 = 6 x 104
80
Moles of NH3 = 6 x 104 ✓½
Mass of NH3 = 6 x 17 x 104
1000
= 1020 kg -
- Explosives eg.T.N.T.
- Production of polymers (terylene)
- Textile dyes.
- Manufacture of drugs
any 2
-
-
-
- Surface area/particle size vi. (1)
-
-
-
- PbCO3(s) + 2HNO3(aq) → Pb(NO3)2 ✓1 + CO2(g) + H2O(l)
-
- With hydrochloric acid an insoluble lead chloride is formed, ✓½ which coats the lead carbonate ✓½ preventing the reaction between the acid and the carbonate from proceeding. ✓1
- The reaction would shift to the left changing the solution from colourless to yellow/ orange ✓1. Addition of HCl creates excess H+ which disturbs the equilibrium so it shifts to the left to get rid of the excess H+ ions. ✓1
-
-
- The anode is X. ✓1 Since hydrogen is liberated at the cathode which is Y. ✓1
- 40H−(aq) → 2H2O(l) + O2(g) + 4e ✓1
- The hydrogen ions and hydroxide ions which form water✓1 are discharged at the electrodes leaving MgSO4 concentrated. The amount of water electrolysed is more than the amount of water formed at the anode.✓1
- Blue litmus remains ✓½ blue while the red litmus remains red ✓½. Indicating that thesolution is neutral. ✓1
- Quantity of electricity = 0.3 x 30 x 60
= 540 ✓1
Oxygen requires 4 Faradays ✓½ of electricity
24 dm3 = 4 x 96500✓½
? 540
24 x 540 = 0.32 dm3
4 x 96500 - Electroplating
Purification of metals
-
-
-
- Cu2+✓1
- CuCO3✓1/ZnSO4✓1
- Ba2+(aq) + SO42−(aq) → BaSO4(s)✓1
- The solution changes from blue to colourless ✓1 and a brown solid is formed. ✓1 The magnesium which is above copper in the reactivity series displaces the copper ions ✓1 from the solution. Apparatus become warm. The reaction is exothermic.
-
- Add nitric (V) acid to ✓½ lead oxide, filter ✓½, add a soluble sulphate/ sulphuric acid to the filtrate ✓½. Filter ✓½, and wash residue with distilled water ✓½ to remove traces of the filterate, then dry residue between ✓½ filter papers /oven.
- Determine the melting✓1 point, if it is pure the melting point will be constant. ✓1
-
-
-
-
- Potassium Manganate (VII) ✓½ and remove heat ✓½.
PbO2 and heat OR CaOCL2 No heating. -
- 2Fe(s) + 3C12(g) →2FeCl3(s) (1)
- 3C12(g) + 6NaOH(aq) → NaCl(aq) + NaClO3(aq) + 3H2O(l) (1)
-
- Cl O
Mass 0.07 1.12
RAM 35.5 16
Moles 0.07 1.12
35.5 16
0.02 0.07
0.02 0.02
1 7/2
2 7
Empirical formula Cl2O7 ✓1 -
- Sterilising drinking water supplies ✓
- Manufacture of hydrochloric acid ✓
- Manufacture of plastics ✓
- Manufacture of chloroform ✓
- Manufacture of bleaching agents ✓
Any 2
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2012 Chemistry Paper 2 with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students