QUESTIONS
- The diagram in Figure 1 shows some natural and industrial processes. Study it and answer the questions that follow.
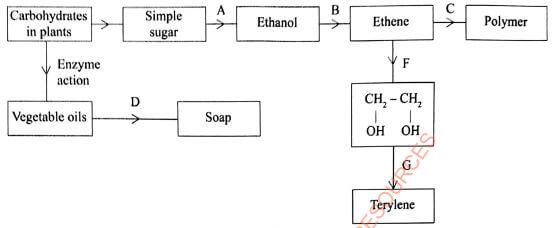
Figure 1- Identify the processes labelled:(2 marks)
- ............................................
- ...........................................
- ...........................................
- ...........................................
- State the reagents and conditions required for processes B and D.
- Process B:
Reagent ............................................. (1 mark)
Conditions ..............................................(1 mark) - Process D:
Reagent.............................................. (1 mark)
Conditions .............................................(1 mark) - Describe how process D is carried out.(2 marks)
- State two additives used to improve the quality of soap.(1 mark)
- Process B:
- State the reagents required in steps F and G.
- F..................................................... (1 mark)
- G.................................................... (1 mark)
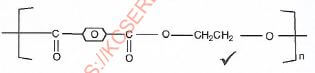
- Draw the structure of terylene.(1 mark)
-
- Name the polymer formed in step C.(1 mark)
- State one disadvantage of the polymer formed in (d) (i).(1 mark)
- Identify the processes labelled:(2 marks)
- Figure 2 is a section of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of elements
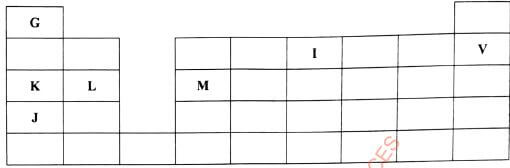
Figure 2-
- Select elements which belong to the same chemical family.(1 mark)
- Write the formulae of ions for elements in the same period.(1 mark)
- The first ionisation energies of two elements K and M at random are 577 kJ/mol and 494 kJ/mol
- Write equations for the 1st ionisation energies for elements K and M and indicate their energies.(1 mark)
- Explain the answer in (b) (i).(1 mark)
- Write the formula of the compound formed when L and I react.(1 mark)
- Give one use of element V.(1 mark)
-
- State another group that G can be placed in Figure 2. Explain.(2 marks)
- How do the reactivity of elements J and K compare? Explain.(2 marks)
-
- Elements L and M form chlorides. Complete the following table by writing the formulae of each chloride and state the nature of the solutions. (2 marks)
elements formula of chloride nature of chloride solution L M - The chloride of element M vapourises easily while its oxide has a high melting point. Explain.(2 marks)
- Elements L and M form chlorides. Complete the following table by writing the formulae of each chloride and state the nature of the solutions. (2 marks)
-
-
- Complete Table 1 by indicating the observations, type of permanent or temporary change and name of new compound formed.
Table 1
(6 marks)experiment observations Type of Change Name of product (i) Heat candle wax strongly in a test tube. (ii) Anhydrous copper(II) sulphate is left exposed overnight (iii) Iron wool is soaked in tap water for two days - Use the set-up in Figure 3 to answer the questions that follow. The flask was covered with a cloth that had been soaked in ice-cold water.
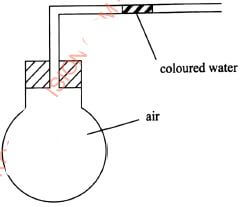
Figure 3- State the observation made on the coloured water. Explain.(2 marks)
- Name the gas law illustrated in Figure 3.(1 mark)
- Use the standard electrode potentials in Table 2 to answer the questions that follow.
Table 2
half-cell Eθ/volts Z+/Z +0.80 V2+/V -0.40 W+/W2 0.00 Y2+/Y -2.87 U+/U +1.90 - Write the half-cell representation for the element whose electrode potential is for hydrogen.(1 mark)
- Arrange the elements in order of reducing power, starting with the weakest reducing agent.(1 mark)
-
- Select two half cells which combine to give a cell with the least e.m.f.(1 mark)
- Calculate the e.m.f of the half cells identified in (iii) I.(1 mark)
- Complete Table 1 by indicating the observations, type of permanent or temporary change and name of new compound formed.
- An experiment was carried out to prepare crystals of magnesium sulphate. Excess magnesium powder was added to 100 cm3 of dilute sulphuric(VI) acid in a beaker and warmed until no further reaction took place. The mixture was filtered and the filtrate evaporated to saturation, then left to & cool for crystals to form.
-
- Write an equation for the reaction.(1 mark)
- Explain why excess magnesium powder was used.(1 mark)
- State how completion of the reaction was determined.(1 mark)
- What is meant by a saturated solution?(1 mark)
- Explain why the filtrate was not evaporated to dryness.(2 mark)
- When bleaching powder, CaOCI2 is treated with dilute nitric(V) acid, chlorine gas is released. This reaction can be used to determine the chlorine content of various samples of bleaching powders and liquids.
- Write an equation for the reaction of nitric(V) acid with bleaching powder.(1 mark)
- Calculate the volume of chlorine produced when 10g of CaOCL2 is treated with excess nitric(V) acid. (Ca = 40.0; 0 = 16.0; CI = 35.5; 1 mole of gas occupies 22.4 dm3 at s.t.p)(3 marks)
- Apart from use of chlorine gas in bleaches and water treatment, state two other uses of chlorine gas.(1 mark)
-
-
- The diagram in Figure 4 was used to prepare hydrogen chloride gas which was passed over heated iron powder.
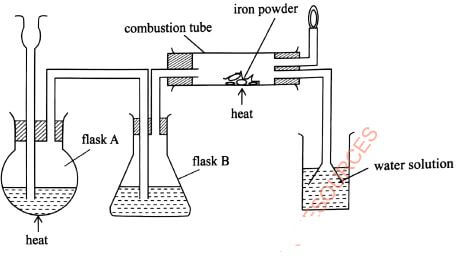
Figure 4- Give a pair of reagents that will produce hydrogen chloride gas in flask A.(2 marks)
- Name the substance in flask B.(1 mark)
- State the observation made in the combustion tube.(1 mark)
- Write an equation for the reaction in the combustion tube(1 mark)
- Describe a chemical test for hydrogen chloride gas.(1 mark)
-
- Identify the gas that burns at the jet.(1 mark)
- Explain why the gas in (b) (i) is burned.(1 mark)
- Give reasons why excess hydrogen chloride gas is dissolved using the funnel arrangement.(2 marks)
- State what will be observed when the reaction in the combustion tube is complete.(1 mark)
- Another experiment was carried out where hydrogen chloride gas was bubbled through methylbenzene and water in separate beakers. The resulting solutions were tested with blue litmus papers and marble chips.
- Write the observations made in the following table.
(2 marks)Solution of hydrogen chloride gas in: blue litmus paper marble chips water methylbenzene - Explain the observations in (e) (i).(2 marks)
- Write the observations made in the following table.
- The diagram in Figure 4 was used to prepare hydrogen chloride gas which was passed over heated iron powder.
-
- In Kenya, sodium carbonate is extracted from trona at Lake Magadi.
- Give the formula of trona.(1 mark)
- Name the process of extracting sodium carbonate from trona.(1 mark)
- The flow chart in Figure 5 summarises the steps involved in the production of sodium carbonate. Use it to answer the questions that follow.
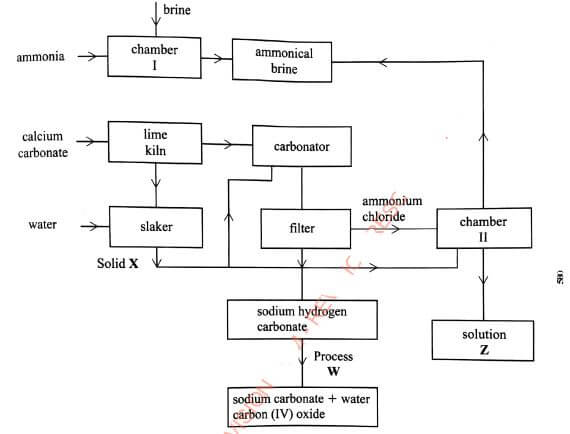
Figure 5- Name the process illustrated in Figure 5.(1 mark)
- Identify the starting raw materials required in the production of sodium carbonate.(2 marks)
- Write equations for the two reactions that occur in the carbonator.(2 marks)
- Name two substances that are recycled.(1 mark)
- Identify:
Solid X;(1 mark)
Process W.(1 mark) - Write an equation for the reaction that produces solution Z(1 mark)
- Apart from softening hard water, state two other uses of sodium carbonate.(2 marks)
- In Kenya, sodium carbonate is extracted from trona at Lake Magadi.
MARKING SCHEME
-
- A-Fermentation
B - Dehydration
C-Addition polymerization / polymerization
D-Saponification -
-
- Process B
- Reagent - Concentrated sulphuric(VI) acid
- Conditions - Temperature of 160°C - 180°C.
OR - Al2O3
- Temperature 300 °C
OR - H3PO4
- warm
-
- Process D
- Reagent - Potassium hydroxidevi/Sodium hydroxide
- Condition - Boil 1/Boiling
- The vegetable oil is mixed with sodium hydroxide and boiled
Solid sodium chloride is added to the resulting mixture, to precipitate out the soap from glycerol. - Perfume and builders / tetraoxophosphates / dyer
-
-
- Step F-acidified potassium manganate (VII)
- Monomer G-Benzene-1.4- dioic acid /

-
-
- Polyethene / polythene
-
- It is non-biodegradable hence pollutes the environment;
- Produces poisonous gases when bumt.
(Any one correct)
- A-Fermentation
-
-
- K and J
- K, L2+ , M3+
3 ions for 1 mark;
2 ions for mark 1/2
1 ion for 0 mark
-
- K(g) → K+(g) + e ΔHIE = 494kJ/mol
M(g) → M+(g) + e ΔHIE = 577kJ/mol - Across the period, size of atoms decreases therefore more energy required to remove electrons from an atom in its gaseous state hence, 1st ionization energy for M will be greater than that of K
OR
K has lower nuclear charge / attraction than M/Khas less protons than M. - L3I2
- Being an inert gas, V is used in fluorescent tubes and bulbs 1/arch welding
- K(g) → K+(g) + e ΔHIE = 494kJ/mol
-
- Group 7. Because G can either lose an electron to form G+ or gain an electron to form G. Jis more reactive than because of increase in the size of atoms. As we go down the group, the atoms increase in size so does reactivity. Outer electrons do not experience much nuclear attraction for bigger atoms.
OR
Reactivity increases down the group, effective nuclear attraction is greater in Kthan J atomic radius of J is greater than that of K.
- Group 7. Because G can either lose an electron to form G+ or gain an electron to form G. Jis more reactive than because of increase in the size of atoms. As we go down the group, the atoms increase in size so does reactivity. Outer electrons do not experience much nuclear attraction for bigger atoms.
-
-
Element Formula of chloride Nature of chloride solution L LCL2 Neutral M MCL3 / M2CL6 Acidic - Chloride of M vaporizes easily because of weak van der Waals forces between its dimer/. Its oxide has a high melting point because of strong ionic bond is difficult to break.
-
-
-
-
experiment observations Type of Change Name of product (i) Heat candle wax strongly in a test tube. It melts and solidifies on cooling Temporary Candle wax (ii) Anhydrous copper(II) sulphate is left exposed overnight Turns from white to blue Temporary Hydrated copper(II) sulphate (iii) Iron wool is soaked in tap water for two days Turns from grey to brown Permanent. Hydrated Iron(III)oxide / rusty -
- Coloured water moves towards the flask. Cold cloth contributes to decrease in temperature causing decrease in volume; this creates a vacuum making the ink to move towards the flask.
- Charle's law
-
- W+(aq), W2(g) /Pt
- U, Z, W2, V, Y
-
- V2+/V and W+/W2
- e.m.f = 0.00-(-0.40)
= ± 0.40 V
-
-
-
- Mg(s) + H2SO4(aq) → MgSO4(aq) + H2(g)
- To ensure all the acid was used up.
- When effervescence stops and presence of unreacted magnesium
- Saturated solution is one that cannot dissolve any more solute at a particular temperature.
- Because magnesium sulphate is a hydrated salt and evaporation to dryness causes it to lose it water of crystallization / crystals would not be formed because water of crystallization is lost through heating
- CaOCl2(s) +2HNO3(aq) → Ca(NO3)2(aq) + Cl2(g) +H2O(l)
- Volume of chlorine produced
Mass of CaOCI2 = 40 + 16+71
= 56 + 71
= 127
Moles of CaOCl2 = 10 = 0.0787 moles
127
Moles of CaOCl2 = moles of Cl2
1 mole CI2, = 22.4 dm3
0.0787 = ?
0.0787 x 22.4 = 1.763 dm3 -
- Manufacture of hydrochloric acid;
- Manufacture of PVC polymers:
- Manufacture of potassium chlorate(V):
- Manufacture of carbon tetrachloride;
- Manufacture of chloroform.
(Any 2 correct @ 1/2 mark)
-
-
-
- Concentrated sulphuric VI) acid and sodium chloride
- Concentrated sulphuric(VI) acid.
- Grey Iron powder turns green / solid glows red.
- Fe(s) + 2HCl(g) → FeCl2(s) + H2(g)
- Open a bottle of concentrated ammonia and place it near HCI(g) dense white fumes of NH4Cl are observed.
-
- Hydrogen gas
- To prevent an explosion since a mixture of hydrogen and oxygen explodes
- to prevent suck back.
to increase surface area for dissolution of hydrogen chloride in water. - The flame will go off
-
-
Solution of hydrogen chloride gas in: blue litmus paper marble chips water Turns red Effervescence / gas bubbles methylbenzene Remain blues No effervescence - In water the hydrogen chloride ionizes to form hydrogen ions and chloride ions. hydrogen ions turn blue litmus red and also react with marble chips to liberate carbon(IV) oxide hence effervescence.rs In methylbenzene which is non polar, HCl is not able to ionize since it is polar. Therefore, negative results with blue litmus paper and marble chips.
-
-
-
-
- Na2CO3.NaHCO3.H2O
- Fractional crystallization.
-
- Solvay process
- Brine, ammonia, calcium carbonate and water
- NH3(g) +CO2(g) + H2O(l) → NH4HCO3(aq)
NH4HCO3(aq) + NaCl(aq) → NH4Cl(aq) + NaHCO3(s) - Ammonia and Carbon(IV) oxide, water
(Any 2 correct @ 1/2 mark) -
- Calcium hydroxide Vi (Ca(OH)2)
- Thermal decomposition
- NH4Cl(aq) + Ca(OH)2(s) → 2NH3(g) +CaCl2(aq) + H2O(l)
- Uses of sodium carbonate
- Glass making
- Paper industry
- Sodium silicate in making detergents
(Any 2 correct @ 1 mark)
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2018 Chemistry Paper 2 with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students

