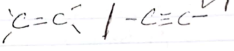THE KENYA NATIONAL EXAMINATIONS COUNCIL
Kenya Certificate of Secondary Education
233/3 CHEMISTRY Paper 3 (PRACTICAL)
Instructions to candidates
- Write your name and index number in the spaces provided above
- Sign and write the date of examination in the spaces provided above
- Answer all the questions in the spaces provided in the question paper
- You are not allowed to start working with the apparatus for the first 15 minutes of the 2¼ hours allowed for this paper. This time is to enable you to read the question paper and make sure you have all the chemicals and apparatus that you may need.
- All working must be clearly shown where necessary
- Non-programmable silent electronic calculators and KNEC mathematical tables may be used.
- This paper consists of 8 printed pages.
- Candidates should check the question paper to ascertain that all the pages are printed as indicated and that no questions are missing.
- Candidates should answer the questions in English.
- You are provided with:
- 5.3g solid A, sodium carbonate;
- Solution B, hydrochloric acid.
You are required to determine the: - Molar heat of the solution of solid A;
- Concentration of the hydrochloric acid, solution B.
- PROCEDURE I
- Using a burette, place 30.0 cm3 of distilled water in a 100ml plastic beaker. Stir the water with a thermometer and measure its temperature after every half-minute interval. Record the readings in Table 1.
- At exactly 2 minutes, add all of solid A to the water at once. Stir well and continue measuring the temperature of the mixture after every half-minute interval. Record the readings in Table 1. Retain the mixture in the beaker for use in Procedure II.
-
(3 marks)Time (minutes) 0 ½ 1 1½ 2 2½ 3 3½ 4 4½ 5 Temperature(ºC) x - On the grid provided, plot a graph of temperature (vertical axis) against time.(3 marks)
- Determine from the graph, the temperature change, AT ( mark)
- Calculate the:
- number of moles of solid A used. (RFM = 106)
- molar enthalpy of solution, ΔHsoln and show the sign of ΔHsoln
(Assume that for the solution, density = 1.0gcm3and specific heat capacity = 4.2 Jg-1K-1) (2 marks)
-
- PROCEDURE II
- Fill a burette with solution B.
- Transfer all of the mixture in the 100 ml plastic beaker from procedure I into a 250 ml volumetric flask. Add distilled water to make up to the mark and shake. Label the mixture as solution A.
- Using a pipette and pipette tiller, place 25.0cm3 of solution A into a 250 ml conical flask. Add two or three drops of phenolphthalein indicator and titrate with solution B. Do not pour out the contents of the conical flask.
Record the readings in Table 2
Add two or three drops of methyl orange indicator to the contents of the conical flask. Titrate the mixture with solution B and record the readings of this second titration in Table 3.
- Repeat Procedure II. step (iii) and complete Tables 2 and 3.
-
- Table 2. using phenolphthalein indicator
(3 marks)I II Final burette reading Initial burette reading Volume of Solution B used, cm3
Average volume, V1 of solution B used = .. (½ mark) - Table 3, using methyl orange indicator.
(3 marks)I II Final burette reading Initial burette reading Volume of Solution B used, cm3
Average volume, V2 of solution B used = .. (½ mark)
- Table 2. using phenolphthalein indicator
- Calculate the:
- concentration, in moles per litre, of sodium carbonate in solution A. RFM 106 (1 mark )
- number of moles of sodium carbonate in 25.0 cm3 of solution A (1 mark)
- number of moles of hydrochloric acid in the total volume, V1 + V2 of solution B. (1 mark)
- concentration, in moles per litre, of hydrochloric acid in solution B. (1 mark)
- You are provided with solid C. Carry out the following tests. Write the observations and inferences in the spaces provided.
Place all of solid C in a boiling tube. Add about 20 cm3 of distilled water and shake until all of the solid dissolves. Label the solution as solution C. Use about 2 cm3 of solution C in a test tube for each of the following tests.- Add aqueous sodium hydroxide dropwise until in excess.
(1 mark) (2 marks)Observations Inferences - Add three drops of aqueous sodium sulphate.
(1 mark) (1 mark)Observations Inferences - Add three drops of aqueous barium nitrate.
(1 mark) (2 marks)Observations Inferences - Add three drops of aqueous lead(1) nitrate. Heat the mixture
(1 mark) ( 1 mark)Observations Inferences
- Add aqueous sodium hydroxide dropwise until in excess.
- You are provided with an organic compound, solid D. Carry out the following tests. Record the observations and inferences in the spaces provided.
- Describe the appearance of solid D. (1 mark)
- Divide solid D into four portions.
- Place the first portion of solid D on a watch glass and burn it with a Bunsen burner flame.
(1 mark) (1 mark)Observations Inferences - Place the second portion of solid D in a test tube. Add about 3cm3 of aqueous sodium hydroxide and shake
(1 mark) ( 1 mark)Observations Inferences - Place the third portion of Solid D in a test tube. Add about 3cm3 of distilled water. Heat the mixture and add three drops of acidified potassium manganate(VII)
( 1 mark) ( 1 mark)Observations Inferences - Place the fourth portion of solid D in a test tube. Add about 3 cm3 of distilled water. Heat the mixture and add all the solid sodium hydrogen carbonate provided.
(1 mark) (1 mark)Observations Inferences
- Place the first portion of solid D on a watch glass and burn it with a Bunsen burner flame.
MARKING SCHEME
- PROCEDURE
-
- Table 1
Award a total of 3 mks distributed as follows:
A Complete table (1 mark.)
Award (1 mark) for complete table with to temperature reading
Penalties / conditions- Penalize ½ mk ONCE for any space not filled subject to at least 5 readings given, otherwise penalize Fully, ie, award 0 mk
- Penalize ½ mk ONCE for unrealistic temperature readings of less than 10ºC and /or greater than 40°C for t=0 to t = 1½ minutes and for temperature reading(s) of greater than 50°C for readings t= 21⁄2 to t= 5 minutes
- Penalize ½ mk if ALL temperature readings are consistent
- Penalize ½ mk ONCE if temperature reading at t=2½ minutes is below or equal to the initial temperature reading at t = 1½ minutes
- If two or more rows of temperature readings are given, Penalize ½ mk on Complete table. However, for use of decimal, accuracy and trend to be credited, the two or more sets of rows MUST meet the two criteria provided for each case.
NOTE! Any reading after t=2½ minutes falls below room temperature(t=1½ mins)
Penalize ½ mk
- Use of decimals (½ mk)
Subject to at least 2 temperature readings.- All temperature readings MUST be recorded consistently either as a whole number, to 1dp or 2 dps, otherwise, penalize fully.
- If readings are recorded to 2 dps, then it should be .00, .025, .50,0.75, otherwise, penalize fully
- If readings are recorded to 1 dp then it should be .0 or .5, otherwise, penalize fully
- ACCURACY (½ mk)
Compare the Candidate's value at t= 1½ mins. With the s.v- If Within +2.0ºC of s.v award ½ otherwise award O mk for accuracy
NOTE . If the Candidate's Value earns the mark, tick (v) the reading on the table. - If no school value is given by the teacher or where the S.V given is unrealistic, Sample and average the candidates' values at t= 1½ mins
Per Session that are close.
However, if Candidates' Values are too varied, then use KNEC value of 22.5°C as the S.v (at t=1½ Mins)
- If Within +2.0ºC of s.v award ½ otherwise award O mk for accuracy
- Trend (1 mk)
Conditions- Award the first ½mk if temperature reachings from t=½ min to t= 1½ mins are constant
- Award the second ½mk for either
- a Continuous rise to a Maximum followed by a constant at maximum and then a continuous drop.
- a Continuous rise up to a maximum followed by a continous drop
- a Constant at the maximum followed by a continuous drop or
- a sudden rise followed by a Continuous drop
- Table 1
- GRAPH(3 mks)
Award a total of 3 mks distributed as follows.- Labelling of axes ½
Award ½mk only if BOTH ares are correctly labelled.
- Penalties
- Penalize. Fully for inverted axes.
- units may or may not be used but if given MUST be Correct, otherwise Penalize Fully for wrong units.
NOTE : Both axes. Must be marked accordingly before posting the mark for labelling axes.
- Penalties
- Scale --- ------ ½ mk.
- Area Covered by the actual plots must be at least half the grid provided ie, 7 big squares vertically and 9 big squares horizontally.
- Scale intervals must be consistent ON EACH 9 the axes:
- Scale chosen MUST be able to accommodate all the readings whether plotted or not.
- If the scale is such that it leaves no room for extrapolation the scale is credited 0 mk.
NOTE- Award for correct scale even if the axes are inverted.
- Penalize Fully if any of the above conditions is not met.
- Plotting (1mk)
Conditions- If 10 or 9 are correctly plotted, award 1mk.
- If only 8 to 5 are correctly plotted, award ½mk.
- If less than 5 are correctly Plotted award 0mk.
NOTE:- Accept correct plots even if axes are inverted and award accordingly
- If any scale intervals are inconsistent, mark the plots, if any - within the first correct interval and treat all the other plots as wrong and award accordingly.
- Mark all the plots on the graph with either a tick (√) or a Cross (X).
- Shape / lines ------- 1mk)
- Accept 2 Straight lines Correctly extrapolated up to t = 2 minutes with the initial line being horizontal and the other line a dropping one for 1mk.
- Accept 2 lines not extrapolated and not joined with the initial line being horizontal and the other a dropping one for ½ mk.
- Each of the two lines MUST pass through at least 2 correctly plotted points, otherwise award 0mk.
- Labelling of axes ½
- Temperature change; ΔT (1 mk)
Conditions- Accept the correct value of ΔT from the correct graph with or without showing on the graph even if the axes are inverted for 1mk.
- if shown on the graph Correctly but Δt is either missing or wrong award ½mk for correct showing on the graph.
- If not shown on two correct graph, award ½mk for correct working and another ½mk for correct ΔT
NOTE:- Reject reading and showing from a wrong graph ie, a graph that has score 0 mk for shape/lines.
- The units may or may not be shown but it shown Must be correct, otherwise Penalize ½ for wrong unit.
-
- No. moles of solid A used = 5.3 = 0.05 marks
106
Conditions / Penalties
- If the expression is NOT shown but answer is correct, award only ½
- The units may or may not be shown, but if shown MUST be correct, otherwise penalize ½mk for wrong units used.
- The 5.3 and lo6 MUST be transferred intact, otherwise penalize FULLY
- ΔH = 30x4.2 x Ans (c) above = correct answer
Molar enthalpy of solution =Correct answer = Final correct answer
ans d(i)above (J mol-1)
OR
ΔH = 30x 4.2 x Ans (c) above = correct answer.
1000
Molar enthalpy of solution = Correct answer = Final correct answer( KJmol-1)
Ans. d(i) above
OR
Molar enthalpy of solution = 30x4 24 ans (c) above = Final correct answer(Jmol-1)
Ans. d(i) above
OR
Molar enthalpy of solution = 30x4 24 ans (c) above = Final correct answer(KJmol-1)
1000 x Ans. d(i) above
Conditions / Penalties- Accept correct transfer of ans (C) and d(i) even if rejected in (C) and d(i) above.
- Penalize ½mk for wrong transfer of either ans. (C) or ans.d(i) or BOTH otherwise Penalize FULLY for strange figures used.
- Penalize ½ mk on final correct answer if the negative sign and for the unit is or are wrong. or missing
- Penalize ½mk for wrong answer if arithmetic error is outside ±2 units in the 3rd digit.
- Penalize FULLY for unrealistic find answer if outside the range of -12.6 kJmol-1 to -28.1.kjmol-1
PROCEDURE II
- No. moles of solid A used = 5.3 = 0.05 marks
-
- Table 2 (4mks)
Award 4 distributed as follows!- Complete table ( 1 mk)
Conditions- Complete table with BOTH titrations done, award( 1 mk)
- Incomplete table with ONLY ONE titration done, award ½mk
Penalize- Wrong arithmetic
- Inverted table
- unrealistic titre(s) (below 1.0cm3 or in hundreds.)
- burette reading (s) beyond 50.0cm3, unless explained
NOTE- Penalize ½mk for each of the above mistakes to a maximum of ½mk Cile (Penalize ½ mk once)
- If No TITRATION is done, award 0mk for complete table as well as for EACH of the other Marking points.
- Use of decimals (tied to 1st and 2nd rows only) ---½mk
Conditions- Accept either 1 or 2 dps used consistently otherwise Penalize FULLY
- If 2 dps are used the second dip should be a "0" or "5" otherwise Penalize Fully.
- Accept inconsistency in the use of Zeros as INITIAL burette reading(s) i.e 0,0.0,0.00
- Accuracy 1mk
compare the candidates correct titre values with the s.v
Conditions- If at least one titre is within ±0.10cm3 of the S.V award 1mk
- If no titre is within ±0.10cm3 to±0.10cm3 of the S.V award but at least one is within ±0.20cm3 of the s.v award ½mk
- If no titre i within ±0.20cm3 of the s.v award 0mk
NOTE!- If there was wrong arithmetic / subtraction in the table, compare the S.v with the worked out CORRECT titre(s) and award accordingly.
- If no S.V is given by the teacher or s.v cannot be worked out from the teacher's titres according to tul principles of averaging, then ,
- write down all the candidate's' average titres per session and sample those that are close and average them to get the s.v.
- If the candidates' average titres are too varied then use the KNEC Value 16.5cm3 as the s.v.
- Tick (√) the candidate's chosen titre on the table, if it earns a mark before posting the mark.
- Principles of averaging ½mk.
Conditions- If both titrations are done are consistent and are averaged correctly, award ½mk
- If both titrations are done, are inconsistent and yet averaged, award 0 MK for both
Principles of averaging and final accuracy. - If only one titration is done, award 0mk for both PA and FA .
NOTE- Answer should be expressed to at least 2d.ps unless it works out exactly to 1dp or whole number otherwise Penalize Fully.
- If no working is shown but answer given is correct, credit FULLY
- The working under Principles of averaging MUST be marked before posting the mark.
Penalties- Penalize FULLY for wrong arithmetic it error is outside ± 2 units in the 2nd dp
- Penalize Fully if no working is shown and answer given is wrong!
- Final accuracy -(1 mk)
(Tied to correct average titre)
Compare the candidate's correct average titre with the s.v
Conditions- If average titre is within ±0.1 cm3 F S.V award look
- If the average titre is not within ±0.1cm3 of s.v but within ±0.20cm3 of s.v award ½
- If the average titre is beyond ±0.2cm3 of the s.v, award 0 mk.
- Complete table ( 1 mk)
- Table 3
Mark as per table 2. However if the KNEC Value is to be used as the s.v then it is 16.45 cm3,
N/B Table 2 & 3 check on school value if realistic / Unrealistic
If school value of table 3 almost double of table 2 , it is unrealistic hence sample out Candidates' Values.
Table 2 and 3 values should be more or less the same.- Calculations
Molarity of soln A = Ans. d(i) above x 1000
250 =correct ans. ½
OR
Conc of Solution A = 1000 x 5.3 = 21.2gdm3
250
Molarity of solution A = 21.2 = 0.2 M
106
NOTE
If there is arithmetic error in the intermediate answer of 21.2 g/dm3 then award ½mk for the correct expression and penalize FULLY for the final answer .
OR
Molarity of solution A = 1000 x 5.3 = 0.2M
25 x 106 - Moles of Na2CO3 in 25 cm3 of solution A
= Ans. f(i) above x 25 = correct ans
1000
OR
5.3 x 25 = 0.05moles
250x106
OR
Ans d(i) above x 25 = correct ans
250 - Mols of HCl solution B in V1+V2
= Ans. f(ii) above x 2 = Correct ans - Molarity of HCl = Ans. F(iii) X 1000= correct ans
V1+V2
OR
MHCl= Ans. f(i) x 25 x 2 = Correct ans
V1+V2
Conditions / Penalties- Answer d(i) and f(i) to f(iii) MUST be transferred INTACT otherwise Penalize back for wrong transfer in each case. However, Penalize FULLY for Strange figure in each case.
- Answer f(i) and f(iv) should be expressed to at least 3dps, unless they work out exactly to 2d.p or 1.d.p. Otherwise, penalize ½mk for round off in each case.
- In answer f(i) to f(iv) units may or may not be given but if given must be Correct otherwise penalize ½ in each case for wrong units used.
- Ans. f(iv) should be in the range of 0.1M to 0.5M otherwise Penalize FULLY for unrealistic answer.
- Calculations
- Table 2 (4mks)
-
-
Observation Inferences White ppt formed which is insoluble in excess alkali
Note:White ppt in excess ( full credit)
- Accept white solid/ suspension
- Reject: - white solution/substance/particlesCa2+, Mg2+ or Ba2+
Note:
- Credit 1 mark for any 2 ions givens
-If one ion given only credit ½mk
- Penalize ½mk for each contradictory ion to max of 1 mk
-Ignore mention od Al3+ , Zn2+ and Pb2+ as absent
Observation Inference White ppt formed
Note:
- Accept white solid/ suspension in place of white ppt
-Ignore mention of insoluble in excess but penalize fully if soluble in excess.
Reject:-White solution
-ppt on its own or ppt with contradictory colourCa2+ Ba2+ present
Note:-Credit 1mk for either of the 2 ions given on its own
-Correct inference tied to Ca2+ Ba2+ having been correctly infered in 2(a) above
-Penalize fully for any contradictory ion given
In the absence of Ca2+ Ba2+ credit ½ for Mg2+ absent.
-Subject to having been correctly infered in 2(a) above as present.-
Observation Inference No white ppt formed
Note:-Accept: - no ppt
-colourless solution
-No observable change for 1/2mk but accept correct
inference and credit accordinglySO42-, SO32-, CO32- absent.
- All 3 ions given - 2mks
-only 2 given - 1mk
- only 1 given - ½mkNote:
- Where there's a contradictory ion, mark out of 1½ and penalize deduct ion to max of 1½ -
Observation Inference A white ppt is formed which dissolves on heating
Note:Reject:
- White solution
white substance
General note:
-Accept ions written in words each for only ½mkCl-/Br- present
note:
-Credit 1mk for either of the two ions
-Correct inference tied to white ppt dissolve on heating
-Penalize fully for any contradictory ion given
-
- White crystalline solid/ white crystals
Note- Accept white solid/ white powder for only ½mk
- Reject solid on its own
- Colourless crystals ( reject)
- Reject white ppt or white solution
-
-
Observation Inference Solid melts and burns with a yellow/ luminous and
sooty/ smoky flame
Note
presentNote:
-Accept either of the following statements given in words in place of structures for full credit.
- unsaturated organic compound
-organic compound with high C:H ratio
-Aromatic organic compound
-Long chain organic compound
-Carbon to carbon double( triple) bond presentReject:
-C=C / C=C
Ignore alkenes/alkybes present
Ignore long chain hydrocarbon
Observation inferences The solid dissolves to form a colourless solution
Note:
-Reject- colurless liquid used instead of colourless solution
Credit fully- colourless solution without use of the word dissolving-Acidic compound.
Note-
-Accept carboxylic/ alkanoic acid present for full credit
-Correct inference is tied to solid dissolving
-Ignore H+, H3O+ present
Observation Inference Acidified KMnO4 solution not dicolorised
OR
Purple colour of acidified solution does not change to colourless
OR
Purple colour of acidified KMnO4 solution retained/ remains/persistsReject- Solution remains purple
- Solution not decolourised
- Purple colour persists on its own
- Colour of KMnO4 remains/ persists/ retained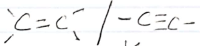
and ROH absent
Notes:
-in the absence of the above structures, accept the correct inference given in words for full credit written as:
-unsaturated organic compound absent
- alkanol/ alcohol absent-
Observation Inference Effrevescence/ bubbles of a colourless gas/ fizzing
Reject- Hissing, fizzling, zizzling, colourless gasR-COOH present
Note:
Accept either of the following given in place of above structure:
- Carboxylic/ alkanoic acid present
- Solution is acidic
-H3O+ / H+ present
General note: -- Credit fully for inference in b(i) and b(ii) even if observation has only scored ½
- Penalize fully for any contradictory functional group in the inferences in each case.
-
- White crystalline solid/ white crystals
Download Chemistry Paper 3 Questions and Answers - KCSE 2020 past papers.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students