QUESTIONS
- The diagram shows part of the Periodic Table. The letters are not the actual symbols of the elements.
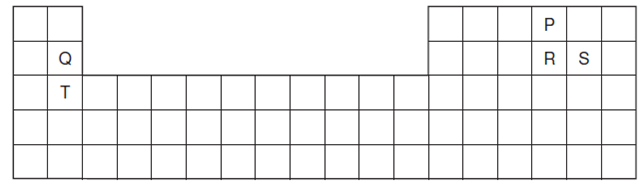
- Compare the reactivity between elements Q and T. (2 marks)
- Explain the electrical conductivity of the chloride of element T. (2 marks)
- Compare the melting and boiling points of elements R and S. (2 marks)
- Write an equation for the second ionization energy of element Q. (1mark)
- How does the atomic and ionic radius of each of the following elements compare?
- Element T. (1½ marks)
- Element P (1½ marks)
- Compare atomic radius of elements R and Q. (1½ marks)
- Explain the difference in the melting points of the oxides of element Q and the oxide of element R. (1½ marks)
-
- The diagram below shows the structure of an allotrope of sulphur
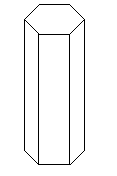
- What are allotropes? (1mark)
- Identify the allotrope shown in the diagram above. (1mark)
- State two properties of the allotrope above. (2 marks)
- Study the flow chart below and answer the questions that follow.
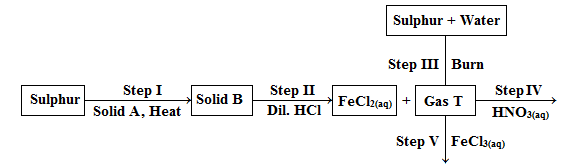
- Write the equation for the reactions in:
- step I. (1mark)
- Step II. (1mark)
- State two observations made in step II. (2 marks)
- Explain the observations made in:
- Step IV. (1½ marks)
- Step V. (1½ marks)
- State one use of gas T. (1mark)
- Write the equation for the reactions in:
- The diagram below shows the structure of an allotrope of sulphur
- Next to each letter, A to F, in the table below is the molecular formula of an organic compound.
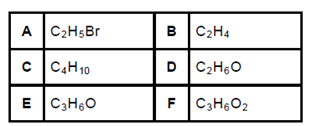
- Choose a molecular formula above that represents an organic compound below. Write down only the letter (A to F) next to the question numbers
- A haloalkane (½ mark)
- An alcohol (½ mark)
- An unsaturated hydrocarbon (½ mark)
- A product of thermal cracking of compound C. (½ mark)
- If compound F is a carboxylic acid, write down the following:
- The structural formula of a functional isomer (an isomer with a different functional group) of F. (1mark)
- The IUPAC name of a functional isomer of F. (1mark)
- Compound B is a monomer used to make a polymer. Write down the:
- Definition of a polymer. (1mark)
- IUPAC name of the polymer. (1mark)
- Balanced equation for the polymerisation reaction (1mark)
- Compound A is used as a reactant in the production of compound D.
- Name the type of reaction that takes place. (1mark)
- State two changes that can be made to the reaction conditions in (d) (i) to obtain compound B, instead of D, as product. (2 marks)
- Choose a molecular formula above that represents an organic compound below. Write down only the letter (A to F) next to the question numbers
- The flow chart below summarizes the extraction of Zinc, study it and answer the questions that follow.
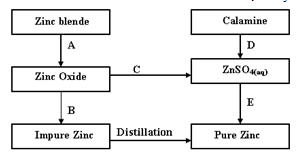
- Name the process represented by A and B
- Identify the reagents required for process B, C and D (3 marks)
- Write a chemical equation of the reaction that occurs in process B (1mark)
- With an aid of a diagram, explain how you would obtain a pure sample of Zinc by process E. (2 marks)
- State two uses of Zinc metal (2 marks)
-
- The table below gives some values of standard enthalpies of formation (ΔHfƟ).
The enthalpy change for the reaction C2H6(g) + 7F2(g) → 2CF4(g) + 6HF(g) is –2889 kJ mol–1.Substance F2(g) CF4(g) HF(g) ΔHfƟ (kJmole-1) 0 -680 -269
Use this value and the standard enthalpies of formation in Table 2 to calculate the standard enthalpy of formation of C2H6(g). (3 marks) - In an experiment to determine the enthalpy of solution of concentrated sulphuric (VI) acid (specific gravity = 1.84gcm-3) the following procedure was used:
- A clean 250.0 cm3 glass or plastic beaker is wrapped with a newspaper leaf.
- About 50.0 cm3 of tap water is measured into the beaker and the steady temperature noted.
- The beaker is held in a tilted position and 2.0 cm3 of and sulphuric acid added into the water
- Why was the beaker wrapped with newspaper leaf? (1mark)
- Why was the acid added into water and not vice versa? (1mark)
- Explain the reason for tilting the beaker during addition of the acid into water. (2 marks)
- Calculate the molar enthalpy of solution of concentrated sulphuric (VI) acid given that ∆T for the reaction = 1ºC. (Density of water = 1gcm-3; specific heat capacity of water = 4.2kJkg-1K-1). (4 marks)
- The table below gives some values of standard enthalpies of formation (ΔHfƟ).
-
- Consider the electrochemical cell represented by the cell notation below, where X is an unknown metal:
Pt(s) | Fe2+(aq), Fe3+(aq) || X+(aq) | X(s)
(The cell potential of this cell was found to be 0.03 V.- Write down the type of electrochemical cell illustrated above. (1mark)
- What does the single line (|) in the above cell notation represent? (1mark)
- Write down the half-reaction that takes place at the anode in the above cell. (1mark)
- Given that:
Identify X with the aid of a calculation. (2 marks)Half reaction EƟ (volts) Fe3+(aq) + e → Fe2+(aq) +0.77 Ag+(aq) + e → Ag(s) +0.80 Na+(aq) + e → Na(s) -2.87 K+(aq) + e → K(s) -2.92
- The diagram below shows the apparatus that can be used to electrolyse dilute Sulphuric acid. Study it to answer the questions that follow.
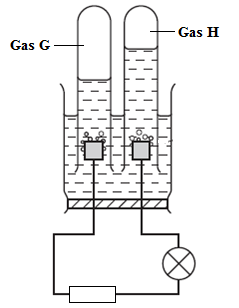
- Identify the gases H and G
- H (1mark)
- G (1mark)
- What happens to the concentration of the Sulphuric acid during the process with time? Explain (2 marks)
- During the electrolysis a current of 0.72A was passed through the electrolyte for 15 minutes. Calculate the volume of gas H produced. (1 Faraday = 96,50ºC, molar gas volume = 24 dm3 at r.t.p). (3 marks)
- Identify the gases H and G
- Consider the electrochemical cell represented by the cell notation below, where X is an unknown metal:
-
-
- What is a salt? (1mark)
- Write the formula of any two double salts. (1mark)
- A student has found that her sample of potassium nitrate is contaminated with small amounts of a green solid. She picks out a small piece of the green solid and finds that it is insoluble in water.
- Describe how you would make a pure sample of potassium nitrate from the impure mixture. (3 marks)
- The student believes that the green solid is copper (II) carbonate. Describe a series of 3 tests that the student could use to confirm this. (6 marks)
Test Procedure Observations Conclusions 1 2 3
- In an experiment 50g of a saturated solution of a salt X was heated to dryness in an evaporating dish. The mass of crystals when weighed gave a reading of 2.7g. Determine the solubility of the salt. (2 marks)
-

MARKING SCHEME
-
- T is more reactive than Q; T has more energy levels than Q hence larger atomic radius; the valence electrons in T are less strongly attracted to the nucleus hence easier to lose;
Owtte - Cannot conduct in solid state, but can conduct in solution and molten states; T has giant ionic structure; in solid state the ions are held in the crystal lattice; in solution and molten states, the ions are free and mobile for electrical conductivity;
- R has higher melting and boiling points than S; both have molecular structures; but R ahs larger molecules than S; hence more van der waals forces which need more heat to break (compared to S);
- Q+(g) → Q2+(g) + e;
-
- Atomic radius of T is larger than its ionic radius; T forms ions by losing electrons; leading to lose of an entire energy level; hence ions have fewer energy levels than atoms;
- Atomic radius of P is smaller than the ionic radius; P ionizes by gaining electrons hence increased electron-electron repulsion;
- Element Q has a larger atomic radius than R; for the same number of energy levels R has more protons than Q hence a stronger effective nuclear pull of valence electrons towards the nucleus;
- Oxide of Q has a higher MP and BP than the oxide of R; Q oxide has a giant ionic structure with strong ionic bonds which require more heat to break; R has a simple molecular structure; with weak van deer waals forces which require less heat to break;
- T is more reactive than Q; T has more energy levels than Q hence larger atomic radius; the valence electrons in T are less strongly attracted to the nucleus hence easier to lose;
-
-
- Different crystalline forms of then same element in the same physical state;
- Monoclinic sulphur;
-
- Prismatic / hexagonal in shape;
- Stable above 96ºC;
- Needle-like in shape;
- Pale yellow in colour;
- Has a melting point of 119ºC;
- Has a density of 1.98g/cm3;
-
-
- S(s) + Fe(s) → FeS(s);
Heat must be on the arrow - FeS(s) + 2HCl(aq) → FeCl2(aq) + H2S(g);
- S(s) + Fe(s) → FeS(s);
- Formation of a green solution;
Colourless gas with a rotten egg smell; -
- Brown fumes; yellow deposits; H2S is oxidized to sulphur; HNO3 is reduced to NO2 and H2O;
- Yellow solution changes tom a green solution, yellow solid // suspension; H2S reduces Fe3+ to Fe2+; while H2S is reduced to S;
- Production of sulphur and sulhuric (VI) acid;
Create inorganic sulphides for the manufacture of pharmaceuticals, pesticides, leather, dyes etc
-
-
-
-
- A;
- D;
- B;
- B;
-
- Structural functional isomer
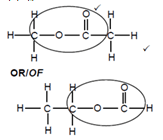
Mark whole structure - Any one of:
Methyl ethanoate;
OR
Ethylmethanoate;
- Structural functional isomer
-
- A large molecule composed of smaller monomer units covalently bonded to each other in a repeating pattern;
- Polyethene;
Accept Polyethylene/polythene - Equation
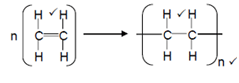
-
- Hydrolysis/Substitution;
- Use concentrated strong base / NaOH / KOH / LiOH OR ethanolic / alcoholic strong base / NaOH / KOH / LiOH. / Use ethanol instead of water. / No water.
Heat strongly;
Accept: Increase temperature;
-
-
- A – Roasting;
B – Reduction; - B – C/ Coke;
C – H2SO4(aq) / Dilute sulphuric VI acid;
D – Dilute H2SO4; - ZnO(s) + C(s) → Zn(s) + CO(g)
- Diagrams
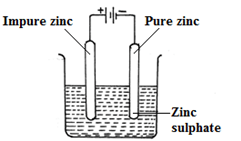
- Dip Zinc rods in a solution of ZnSO4(aq);
- Complete circuit where anode dissolves (impure Zinc) and pure Zinc is discharged at the cathode;
-
- Cases in dry cells;
- Galvanizing iron sheets;
- Making alloys (Brass i.e. copper and Zinc)
Any two)
- A – Roasting;
-
- Calculation
ΔH = Σ ΔfH products – Σ ΔfH reactants or a correct cycle
Hence = (2 × –680) + (6 × –269) – (x) = –2889
x = 2889 – 1360 – 1614 = –85 (kJ mol–1) -
- To reduce heat loss to the surrounding;
Reject to stop // prevent heat loss; - Addition of acid into water will lead to an exothermic reaction; the heat energy evolved would cause vapourization of the acid which may cause acid burns; adding acid to water ensures the denser acid sinks to the bottom of the container so that no surface vapourization (which may be dangerous) occurs;
Owtte; - Avoid spurting of the acid;
Ensure the acid sinks faster to the bottom of the container; - ∆H = MC∆T;
Total volume = 50 cm3 + 2cm-3 = 52cm3;
Note: addition of liquid into a liquid causes a change in volume;
Mass = 52 cm3 x 1gcm-3 = 52g = 0.051kg;
Heat change = 0.052 x 4.2 x 1;
=- 0.2184 kJ;
Mass of sulphuric acid = 2 x 1.84 = 3.68g;
Thus 3.68g = -0.2184 kJ;
Then 98g = 98 x 0.2184 = -5.8085 kJ per mole;
3.68
Penalize ½ mark for wrong or missing sign // SI unit;
- To reduce heat loss to the surrounding;
- Calculation
-
-
- Galvanic (cell)/Voltaic (cell);
- Indicates phase boundary / Interphase / phase separator;
- Fe2+(aq) → Fe3+(aq) + e-;
- Calculation:
EƟ = EƟreduction – EƟoxidation
0.03V = EƟreduction – 0.77
EƟreduction = 0.03 + 0.77
= + 0.80V
Thus X is silver;
Accept any other correct formula.
-
- Solution or a melt that allow current to pass through and is decomposed. √1
-
- G: Oxygen gas;
- H: Hydrogen gas;
- Concentration increases because OH- ions and H+ ions are discharged; hence continuous dissciationofwaterr from the electrolyte;
- Calculation
2H+(aq) + 2e → H2(g)
Q = 0.72 x 15 x 6ºC.
= 738C;
2 x 96,50ºC → 24dm3
738C → 24 x 738 dm3
193,000
= 0.0918dm3 (91.8cm3)
-
-
-
- A substance formed when a cation from an alkali reacts with an anion from an acid;
- (KAl(SO4)2.12H2O;
[Fe(NH4)2(SO4).6H2O;
Na2CO3.NaHCO3.2H2O;
-
-
- Add water to the mixture and stir;
- Filter to obtain potassium nitrate solution as the filtrate;
- Heat the filtrate until saturated;
- Allow saturated solution to cool and crystallize;
- Pour off the mother liquor;
- Dry the crystals between filter papers;
- Series of tests
Test Procedure Observations Conclusions 1 To the green solid add drops of dilute nitric (V) acid / hydrochloric acid / sulphuric (VI) acid; Effervescence of a colourless odourles gass that extinguishes a glowing splint; Production of carbon (IV) oxide confirms presence of carbonate 2 To a sample of the solution from test 1 add aqueous sodium hydroxide dropwise till in excess Blue nprecipitate that is insoluble in excess;
Formation of copper (II) hydroxide which is blue in colour; 3 To a sample / portion of solution from test 1 add drops of aqueous ammonia dropwise till in excess Blue precipitate soluble in excess to form a deep blue solution; Formation of blue copper (II) hydroxide with little ammonia which dissolves in excess ammonia to form a deep blue solution of tetra amine copper (II) ions;
-
- Calculation:
Mass of solvent: 50 – 2.7 = 47.3g
Thus if 47.3g → 2.7g of salt
Then 100g → 100 x 2.7 = 5.7082g/100g of solvet;
47.3
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - Asumbi Girls Mock Examinations 2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students
