INSTRUCTIONS TO CANDIDATES
- Answer all the questions in the spaces provided above.
- KNEC Mathematical tables and silent electronic calculators may be used.
- All working must be clearly shown where necessary.
- Candidates should answer the questions in English.
-
- Define the following terms:
- Strong bases (1mk)
- Amphoterism (1mk)
- Solubility (1mk)
- Using the equation below, identify the species that acts as the base in the forward reaction. Give a reason. (2mks)
NH3(g) + H2O(l)NH4+(aq) + OH-(aq)
- A solution of ammonia gas in water causes a greater deflection of the ammeter whilesolution of ammonia gas in methylbenzene does not cause deflection. Explain this observation. (1mk)
- Write a well-balanced chemical equation for the reaction between sodium hydroxide solution and zinc oxide. (1mk)
- Explain how hard water is softened by ion exchange method. (2mks)
- The table below gives the solubilities of sodium chloride and sodium sulphate at 0°C and 40°C.
Substance Solubility in g/100g of water 0°C 40°C Sodium chloride 55 75 Sodium Sulphate 10 12
When an aqueous mixture containing 60g of sodium chloride and 7g of sodium sulphate in 100g of water at 80°C was cooled to 0°C, some crystals were observed.- Identify the crystals and determine the mass of the crystals formed. (2mks)
- Name the method used to obtain the crystals. (1mk)
- Define the following terms:
-
- Define
- Molar heat of combustion. (1mk)
- Heating value of a fuel. (1mk)
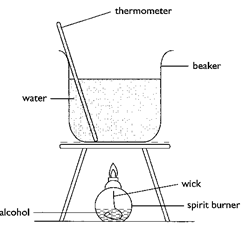
- In an experiment to determine the heat of combustion of ethanol. CH3CH2OH, a student set up apparatus as shown in the diagram below. Study the set up and the data and answer the questions that follow.
Volume of water = 100cm3
Final temperature of water = 36.0°c
Initial temperature of water = 22.0°c
Final mass of lamp an ethanol = 84.75g
Initial mass of lamp and ethanol = 85.10g
Density of water = 1g/cm3
(Specific heat capacity of water = 4.2kJKg-1K-1)- Calculate:
- Number of moles of ethanol used in this experiment. (C=12, O=16, H=1)(1 mk)
- The amount of heat given out in this experiment. (2mks)
- The heat of combustion per mole of ethanol. (1 mk)
- Write a thermochemical equation for the combustion of ethanol. (1 mk)
- Explain how the molar heat of combustion for ethanol obtained above differs with the theoretical value. (2mks)
- State one precaution that should be adhered to when carrying out this experiment. (1mk)
- In this experiment an assumption that links ethanol and water is made. State the assumption. (1 mk)
- Draw an energy level diagram for the combustion of ethanol. (2mks)
- Calculate:
- Define
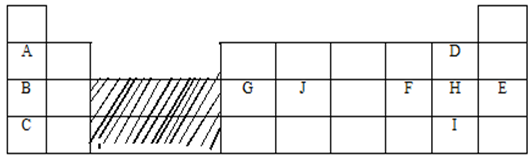
- The figure below represents a section of the periodic table. Study it and answer the questions that follow. Note that the letters do not represent the actual symbols of the elements.
- Consider elements H and I.
- Explain why theatomic radius of element H is smaller than its ionic (1mk)
- When element H was dissolved in water and blue litmus papers dipped, the colour of the litmus paper changed from blue to red to white. Explain. (2mks)
- Explain what is likely to be observed when element H is bubbled through a solution containing the ions of element I. (2mks)
- Compare the atomic radius of elements G and J. Explain the difference. (2mks)
- Use dot and cross diagram to show bonding in a compound of B and H. (1mk)
- G chloride has an unexpected bond type and structure.State the type of bond and the structure.
Bond type (lmk)
Structure (lmk) - A piece of blue litmus paper is placed in a solution of B chloride and a solution of G chloride. Explain what would be observed in each case.
- B chloride solution (lmk)
- G chloride solution (1mk)
- Consider elements H and I.
- In an experiment to study the rate of reaction, 2.5g of copper (II) sulphate crystals were added to a given mass of zinc granules and 100cm3 dilute hydrochloric acid at 27°C. The volume of hydrogen released was measured at 10 second intervals. The results obtained are tabulated below.
Time (seconds) 0 10 20 30 40 50 60 70 80 90 Volume (cm3) 0 60 85 105 114 116 118 122 122 122 - Why were the following not used in the reaction?
- Nitric (V) acid (1mk)
- Iron powder (1mk)
- On the grid below plot a graph of volume of gas against time and label it X (3mks
- Use the graph to calculate the rate of reaction at t=25seconds (2mks)
- Explain why the volume of gas produced does not exceed 122cm3 (1mk)
- Sketch graph Y on the same grid to show the results if the experiment is repeated at 20°C. (1mk)
- How does the catalyst used (copper (II) sulphate) speed up the reaction? (1mk)
-
- State Le’Chatelier’s principle. (1mk)
- What is the effect on the position of equilibrium when dilute hydrochloric acid is added to the closed system of the reaction below (1mk)
NH3 (g) + H2O (l)NH4+(aq) + OH-(aq)
- Why were the following not used in the reaction?
-
- Give the IUPAC names of the following organic compounds. (2 mks)
- CH3 – CH2 – CH – CH3
|
CH – CH3
|
CH3 - CH3 – C ≡ C – CH3
- CH3 – CH2 – CH – CH3
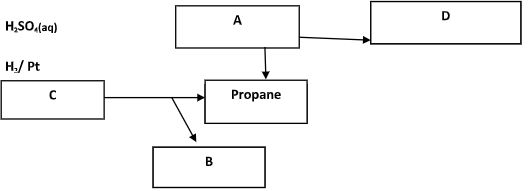
- Study the flow chart below and answer the questions that follow:
- Identify substances (2mks)
- A
- B
- C
- D
- State how substance A and propanecould be distinguished chemically. (1 mk)
- Give the components of soda lime in step I (2mks)
- What is the industrial application of the process that converts substance A to propane? (1mk)
- In the laboratory propene can be prepared using propanol, broken porcelain and sodium hydroxide.
State the use of broken porcelain and sodium hydroxide solution- Broken porcelain (1mk)
- Sodium hydroxide solution (1mk)
- Identify substances (2mks)
- Give the IUPAC names of the following organic compounds. (2 mks)
-
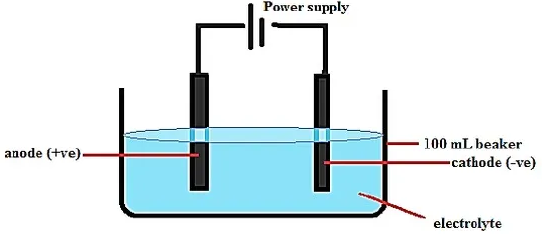
- Draw a fully labeled diagram of the apparatus you would use to electrolyse an electrolyte in the aqueous state. (3mks)
- Explain why crystals of sodium chloride are non-conductorsof electricity butwhen melted they conducts electric current more readily. (2mk
- Answer the following questions in relation to the electrolysis of molten lead (II) iodide.
- State what happens to molten lead (II) iodide when an electric current is passed through it. (1mk)
- At what electrode is a metal formed? Write an equation to show how the metal is formed. (2mks)
- Why is it necessary to carry out this experiment in a fume chamber? (1mk)
- What is a binary electrolyte? (1mk
- Give the application of electrolysis in
- Chemical manufacturing industry (1mk)
- Metal extraction industries (1mk)
- Jewellery e.g. necklaces manufacturing industries (1mk)
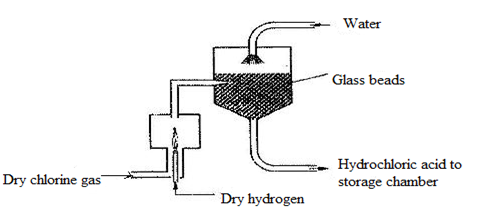
- The diagram below represents the industrial manufacture of hydrochloric acid. Study it and answer the questions that follow.
- Name the main source of hydrogen in this process. (1mk)
- The reaction between chlorine and hydrogen can be very explosive. How can this be avoided? (1mk)
- What is the role of glass beads in the absorption chamber? (1mk
- Explain why the storage chamber for hydrochloric acid is made up of steel lined with rubber. (1mk)
- The acid obtained is 35% pure. Calculate its molarity, given that at 25°C, the density of the acid is 1.08g/cm3 (H=1, Cl=35.5) (3mks)
- Explain why hydrochloric acid is not used to acidify potassium manganate (VII) solution.(1mk)

MARKING SHEME
-
- Define the following terms:
- Strong bases (1mk)
- Bases that dissociate completely in water to yield more hydroxide ions
- Amphoteric oxides (1mk)
- Oxides that react both as acids and bases
- Solubility (1mk)
- The maximum mass of a solute required to saturate 100 g of the solvent at a particular temperature
- Strong bases (1mk)
- Using the equation below, identify the species that acts as the base in the forward reaction. Give a reason. (2mks)
NH3(g) + H2O(l)NH4+(aq) + OH-(aq)
NH3; accepts a proton from water to form ammonium ion. - A solution of ammonia gas in water causes a greater deflection of the ammeter than a solution of ammonia gas in methylbenzene. Explain this observation. (1mk)
- In water ammonia dissolves and ionizes to produce hydroxide ions. In methylbenzene, ammonia dissolves but does not ionize to produce hydroxide ions.
- Write a well-balanced chemical equation for the reaction between sodium hydroxide solution and zinc oxide. (1mk)
Zn(s)+ NaOH(aq) → Na2ZnO2(aq)+ H2O(l) - Explain how hard water is softened by ion exchange method. (2mks)
- The Ca2+ and Mg2+ ions in hard water are exchanged with Na+ in the column and precipitated out
- The table below gives the solubilities of sodium chloride and sodium sulphate at 0°C and 40°C.
When an aqueous mixture containing 60g of sodium chloride and 7g of sodium sulphate in 100g of water at 80°C was cooled to 0°C, some crystals were observed.- Identify the crystals and determine the mass of the crystals formed. (2mks)
- Sodium chloride
Mass 60-55=5 g
- Sodium chloride
- Name the method used to obtain the crystals. (1mk)
- Fractional crystallization
- Identify the crystals and determine the mass of the crystals formed. (2mks)
- Define the following terms:
-
- Define
- Molar heat of combustion. (1mk)
- The enthalpy change/heat change when one mole of a substance is completely burned in oxygen
- Heating value of a fuel. (1mk)
- The amount of heat energy given out when a unit mass or a unit volume of a fuel is completely burned in oxygen.
- Molar heat of combustion. (1mk)
- In an experiment to determine the heat of combustion of ethanol. CH3 CH2OH, a student set up apparatus as shown in the diagram below. Study the set up and the data and answer the questions that follow.
Volume of water = 100cm3
Final temperature of water = 36.0°c
Initial temperature of water = 22.00=°c
Final mass of lamp an ethanol = 84.75g
Initial mass of lamp and ethanol = 85.10g
Density of water = 1g/cm3
(Specific heat capacity of water = 4.2kJKg-1K-1)- Calculate:
- Number of moles of ethanol used in this experiment. (C=12, O=16, H=1) (1 mk)
Mass of methanol = ( 85.10- 84.78) = 0.32 g
Moles = 0.32=0.01 moles
32 - The amount of heat given out in this experiment. (2mks)
100×4.2 ×14 = 5880 J OR 5.88 k J - The heat of combustion per mole of ethanol. (1 mk)
0.01= 5.88
1= ?
1×5.88 = -588 KJ/mol
0.01
- Number of moles of ethanol used in this experiment. (C=12, O=16, H=1) (1 mk)
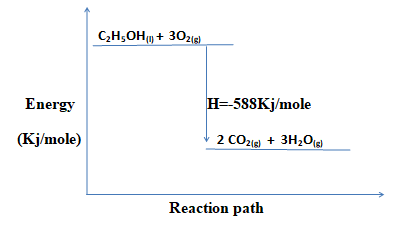
- Write a thermochemical equation for the combustion of ethanol. (1 mk)
C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(g) H=-588Kj/mol - Explain how the molar heat of combustion for ethanol obtained above differs with the theoretical value. (2mks)
- The molar heat of combustion is lower than the theoretical value due to heat lost to the surrounding or heat absorption by the apparatus
- State one precaution that should be adhered to when carrying out this experiment. (1mk)
- The thermometer should not come into contact with the bottom of the hot beaker
- In this experiment an assumption that links ethanol and water is made. State the assumption. (1 mk)
- Heat released by the ethanol=heat gained by the water
- Draw an energy level diagram for the combustion of ethanol. (2mks)
- Calculate:
- Define
- The figure below represents a section of the periodic table. Study it and answer the questions that follow. Note that the letters do not represent the actual symbols of the elements.
- Consider elements H and I.
- Explain why the atomic radius of element H is smaller than its ionic radius (1mk)
- Due to electron-electron repulsion between the existing electrons and the incoming electron
- When element H was dissolved in water and blue litmus papers dipped, the colour of the litmus paper changed from blue to red to white. Explain. (2mks)
- Dissolving element H in water form chlorine water which is acidic; the chloric (I) acid in chlorine water bleaches the litmus paper//Accept: A solution with bleaching properties
- Explain what is likely to be the observation when element H is bubbled through a solution of element I. (2mks)
- The solution turns orange because element H displaces the ions of element I
- Explain why the atomic radius of element H is smaller than its ionic radius (1mk)
- Compare the atomic radius of of Element G and J. Explain the difference. (2mks)
- J has a smaller atomic radius than G; because it has more protons and hence a stronger nuclear force of attraction that pulls the energy levels closer to the nucleus.
- Use dot and cross diagram to show bonding in a compound of B and H. (1mk)
- It is the ionic bond of NaCl
- G chloride has an unexpected bond type and structure. State the type of bond and the structure.
- Bond type - Covalent/dative (lmk)
- Structure - Molecular (lmk)
- A piece of blue litmus paper is placed in a solution of B chloride and a solution of G chloride. Explain what would be observed in each case.
- B chloride solution (lmk)
- Remains blue; a solution of B chloride in water is neurtal
- G chloride solution (1mk)
- Turns red; G chloride is hydrolysed in water to form an acidic solution
- B chloride solution (lmk)
- Consider elements H and I.
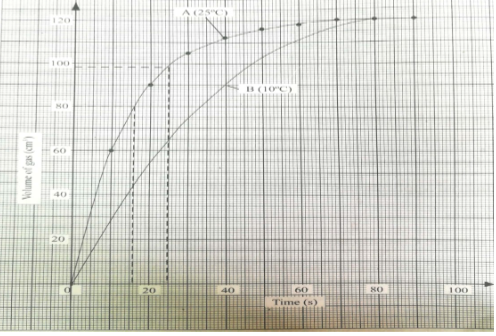
- In an experiment to study the rate of reaction, 2.5g of copper (II) sulphate crystals were added to a given mass of zinc granules and 100 cm3 dilute hydrochloric acid at 25°C. The volume of hydrogen released was measured at 10 second intervals. The results obtained are tabulated below.
- Why were the following not used in the reaction?
- Nitric (V) acid (1mk)
- Oxidises hydrogen produced to water
- Iron powder (1mk)
- Produces poisonous hydrogen sulphide gas
- Nitric (V) acid (1mk)
- On the grid below plot a graph of volume of gas against time and label it X (3mks)
- Use the graph to calculate the rate of reaction at t=25seconds (2mks)
- A tangent must be done on the curve & its gradient determined
112-80 =0.275cm3/sec
130-14
- A tangent must be done on the curve & its gradient determined
- Explain why the volume of gas produced does not exceed 122cm3 (1mk)
- As the reaction proceeds, hydrochloric acid and is being used up. When the volume is 122, all the acid has been used up.
- Sketch graph Y on the same grid to show the results if the experiment is repeated at 10°C. (1mk)
- How does the catalyst used (copper (II) sulphate) speed up the reaction? (1mk)
- By lowering activation energy; or by forming a short-lived intermediate product that decomposes to release the final products; or by providing a large surface area for effective collision of reacting particles. (accept 1 only).
- Use the graph to calculate the rate of reaction at t=25seconds (2mks)
-
- State Le’Chatelier’s principle. (1mk)
- When a change in condition is applied to a system in equilibrium, the system moves so as to oppose the change.
- What is the effect on the position of equilibrium when dilute hydrochloric acid is added to the closed system of the reaction below (1mk)
NH3(g) + H2O(l)NH4+(aq) + OH-(aq)
- The position of equilibrium shifts to the right.
- State Le’Chatelier’s principle. (1mk)
- Why were the following not used in the reaction?
-
- Give the IUPAC names of the following organic compounds. (2 mks)
- CH3 – CH2 – CH – CH3
|
CH – CH3
|
CH3
2,3-dimethylpentane - CH3 – C ≡ C – CH3
But-2-yne
- CH3 – CH2 – CH – CH3
- Study the flow chart below and answer the questions that follow:
- Identify substances (2 mks)
- A Propene
- B Sodium carbonate
- C Sodium butanoate
- D Propyl hydrogen sulphate
- State how substance A and propanecould be distinguished chemically. (1 mk)
- A decolourises bromine water; propane does not. (accept use of dichromate or manganate)
- Give the components of soda lime in step I (2mks)
- Sodium hydroxide and calcium oxide
- What is the industrial application of the process that converts substance A to propane? (1mk)
- Manufacture of margarine/Hardening oils to fats
- Identify substances (2 mks)
- In the laboratory preparation of propene using propanol, broken porcelain and sodium hydroxide solution are used. State the use of broken porcelain and sodium hydroxide solution
- Broken porcelain (1mk)
- Prevents bumping that can lead to flask cracking
- Sodium hydroxide solution (1mk)
- To remove carbon (VI) oxide and sulphur (VI) oxide formed when conc. sulphuric acid and ethanol decompose respectively due to heat.
- Broken porcelain (1mk)
- Give the IUPAC names of the following organic compounds. (2 mks)
-
- Draw a fully labeled diagram of the apparatus you would use to electrolyse an electrolyte in the aqueous state. (3mks)
- Explain why crystals of sodium chloride are non-conductors electricity but when melted they conducts electric current more readily. (2mk)
- Solid- Has fixed ions,
- Melt—Has mobile ions
- Answer the following questions in relation to the electrolysis of molten lead (II) iodide.
- State what happens to molten lead (II) iodide when an electric current is passed through it.
- It decomposes; Pb2+ migrate to the cathode while I- migrate to the anode (1mk)
- At what electrode is a metal formed? Write an equation to show how the metal is formed. (2mks)
- Cathode
Pb2+(l) + 2e → Pb(s)
- Cathode
- Why is it necessary to carry out this experiment in a fume chamber? (1mk)
- Iodine is poisonous
- What is a binary electrolyte? (1mk)
- Contains only one type of cation and one type of anion
- State what happens to molten lead (II) iodide when an electric current is passed through it.
- Give the application of electrolysis in
- Chemical manufacturing industry (1mk)
- Manufacture of hydrogen, chlorine and sodium hydroxide
- Metal extraction industries (1mk)
- Extract very reactive metals like Na & Al; Purify impure metals
- Jewellery e.g. necklaces manufacturing industries (1mk)
- To electroplate to add beauty/ improve appearance
- Chemical manufacturing industry (1mk)
- Draw a fully labeled diagram of the apparatus you would use to electrolyse an electrolyte in the aqueous state. (3mks)
- The diagram below represents the industrial manufacture of hydrochloric acid. Study it and answer the questions that follow.
- Name the main source of hydrogen in this process. (1mk)
- Cracking of long chain alkanes or from natural gas
- The reaction between chlorine and hydrogen can be very explosive. How can this be avoided?
- Pass hydrogen through a jet and burn in excess chlorine (1mk)
- What is the role of glass beads in the absorption chamber? (1mk)
- Increase the surface area for the absorption of hydrogen chloride gas in water
- Explain why the storage chamber for hydrochloric acid is made up of steel lined with rubber.
- Acid reacts with steel but not with rubber (1mk)
- The acid obtained is 35% pure. Calculate its molarity, given that at 25°C, the density of the acid is 1.08g/cm3 (H=1, Cl=35.5) (3mks)
Concentration in g/litre
1 cm3 contains 1.08g
1000=1000 × 1.08=1080g/litre
1
Molarity= 1080 = 29.59M
36.5
Molarity of pure acid = 35/100 × 29.59=10.36M - Explain why hydrochloric acid is not used to acidify potassium manganate (VII) solution.(1mk)
- It will be oxidized to chlorine by potassium manganate (VII).
- Name the main source of hydrogen in this process. (1mk)
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - Momaliche Post Mock 2020 Exam.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students