INSTRUCTIONS TO CANDIDATES.
- Answer all the questions in the spaces provided.
- All workings must be clearly shown where necessary.
- KNEC mathematical tables and non-programmable electronic calculators may be used.
- Candidates must answer the questions in English.
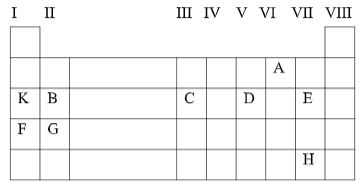
- The grid below shows part of the periodic table. Study it and answer the questions that follow. The letters do not represent the true symbols of the elements.
- Which element forms ions with charge of -2? Explain your answer. (2mks)
- Which element is the most reactive metal? (1mk)
- Name the chemical family to which element B and G belong. (1mk)
- What is the nature of the oxide formed by C? (1mk)
- How does the reactivity of H compare with that of E? Explain. (2mks)
- Write a balanced equation between B and Chlorine. (1mk)
- Explain how the atomic radii of F and G compare. (2mks)
- If the oxides of B and D are separately dissolved in water, what effects will their aqueous solutions have on litmus paper? (2mks)
-
-
- State what is meant by “dynamic equilibrium”. (1mk)
- Dichromate(VI) ions are orange in colour while chromate(VI) ions are yellow. Consider the following equilibrium.
Cr2 O72- (aq) + 2OH-(aq)2CrO42-(aq) + 2H2O(1)
State and explain the observations that will be made if sulphuric(VI) acid is added to the mixture. (2mks)
- One of the reactions in the manufacture of Nitric(V) acid involves catalytic oxidation of ammonia as shown in the equation.
4NH3(g) + 502(g)4NO(g) + 6H2O(g); ∆H = -90kjmol-1
The reaction is carried out at a pressure of 10 atmospheres and a temperature of 900°C.- Other than Nitric(V) acid, name another product that is formed. (1mk)
- State and explain the effect on the position of equilibrium if the reaction is carried out;
- At 10 atmospheres pressure and 450°C. (2mks)
- At 900°C and 20 atmospheres pressure. (2mks)
- In the absence of a catalyst. (1mk)
- State and explain the effect on the rate of the reaction if the reaction is carried out at 10 atmospheres and 450°C. (2mks)
- A factory uses 100kg of ammonia each day to produce 160kg of nitrogen (II) oxide.
Calculate the percentage yield of nitrogen(II) oxide. (3mks)
-
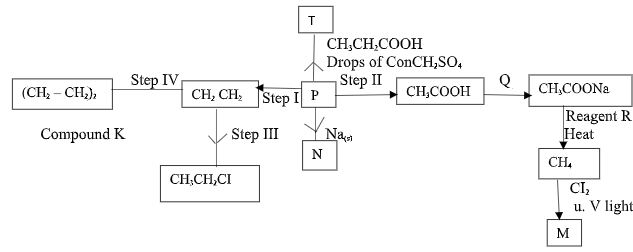
- The scheme below shows reactions of some organic compounds. Study it and answer the following questions.
- Give the name and draw the structural formula of compound P. (2mks)
- Give the reagents and conditions necessary for carrying out:-
- Step (I) (1mk)
- Step (II) (2mks)
- Give the name of the type of reaction that takes place in:-
- Step (1) ---- ( ½ )
- Step (II) ----- ( ½ )
- Step (IV) ----- ( ½ )
- Name reagent Q. -------( ½ )
- Write an equation forming compound T (1Mk)
- Draw the structural formula of M and give its name (1Mk)
-
- Name compound K (1Mk)
- If the relative molecular mass of K is 84,000, determine the value of n (C=12, H=1) (2Mks)
- The following data was obtained during an experiment to determine the molar heat of combustion of methanol.
Volume of water used = 100cm3
Initial temperature of water = 22°C
Final temperature of water = 38°C
Mass of methanol + Lamp before burning = 88.10g
Mass of methanol + Lamp after burning = 87.78g- Calculate:-
- The highest temperature change. (1mk)
- The mass of methanol burnt (1mk)
- The number of moles of methanol used in the experiment ( C=12, O = 16, H = 1). (1mk)
- The heat change in the experiment. (2mks)
(Density of water 1g/cm3, specific heat capacity of water = 4.2 Jg-1K-1) - The molar heat of combustion of methanol. (2mks)
- Write the thermochemical equation for the combustion of methanol. (1mk)
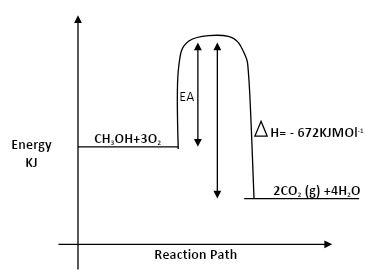
- In the space provided, sketch a simple energy level diagram for the above change, showing activation energy. (2mks)
- Calculate:-
-
- Study the standard electrode potentials for the half cells given below and answer the questions that follow. (The letters do not represent the actual symbols of the elements).
Half cells Eᶿ Volts N+(aq) e ⟶ N(s) -2.92 J+(aq) + e ⟶ J(s) +0.52 K+(aq) + e ⟶ ½ K2(g) 0.00 ½ G2(g) + e ⟶ G-(aq) +1.36 M2+(aq) + 2e ⟶ M(s) -0.44 - Identify the strongest reducing agent. Give a reason for your answer. (1mk)
- Which two half cells would produce the highest potential difference when combined. (1mk)
- Explain whether the reaction represented below can take place. (2mks)
2N+(aq) + M(s) ⟶ 2N(s) + M2+(aq)
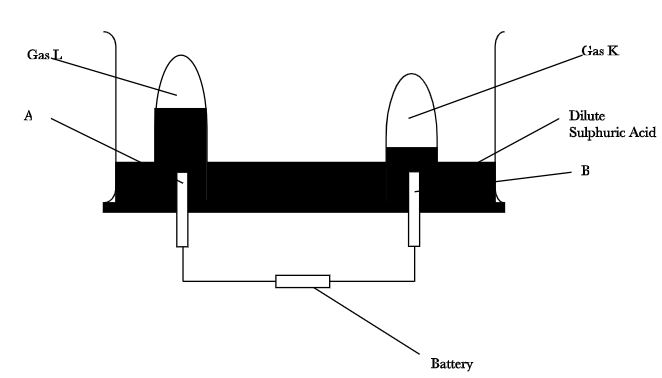
- 100cm3 of 2M Sulphuric(VI) acid was electrolyzed using the set-up represented by the diagram below.
- Name electrode A and B (1mk)
- Write an equation for the reaction that produces gas L. (1mk)
- Describe how gass K can be identified. (1mk)
- Explain the difference in.
- The volume of the gases produced at the electrodes. (1mk)
- Brightness of the bulb if 100cm3 of 2M ethanoic acid was used in place of sulphuric (VI) acid. (1mk)
- Study the standard electrode potentials for the half cells given below and answer the questions that follow. (The letters do not represent the actual symbols of the elements).
-
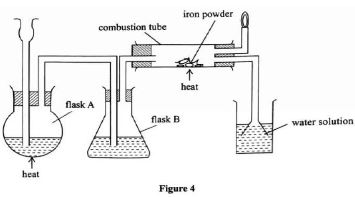
- The diagram below was used to prepare hydrogen chloride gas which was passed over heated iron powder.
- Give a pair of reagents that will produce hydrogen chloride gas in flask A. (1mk)
- Name the substance in flask B. (1mk)
- State the observation made in the combustion tube. (1mk)
- Write an equation for the reaction in the combustion tube. (1mk)
-
- Identify the gas that burns at the jet. (1mk)
- Explain why the gas in b (i) is burned. (1mk)
- Give reasons why excess hydrogen chloride gas is dissolved using the funnel arrangement. (1mk)
- State what will be observed when the reaction in the combustion tube is complete. (1mk)
- The diagram below was used to prepare hydrogen chloride gas which was passed over heated iron powder.
-
- Define radioactivity. (1mk)
- Give two differences between chemical and nuclear reactions. (2mks)
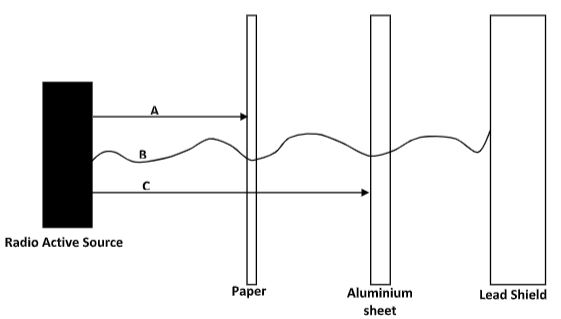
Chemical reactions Nuclear reactions - Study the diagram below and answer the questions that follow.
- What property of radiations is being investigated by the illustration above. (1mk)
- Give the name of the radiation B and give a reason. (1mk)
- Below is the radioactive decay starting with 214 83 Bi, study it and answer the questions that follow.
- dentify the radiations emitted at: - (1mk)
Step 1
Step V - Write a nuclear equation for step II. (1mk)
- dentify the radiations emitted at: - (1mk)
- State one application of radioactivity in: -
- Medicine (1mk)
- Agriculture (1mk)
- State two dangers associated with radioactivity. (1mk)
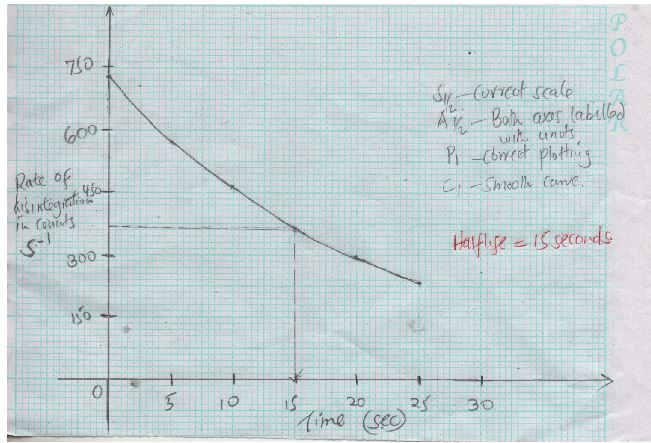
- The decay rates of a sample of radioisotope of Bismuth at different time. Intervals as indicated in the following table.
Time (Hours) 0 5 10 15 20 25 Rate of disintegration in counts 5-1 730 570 455 365 292 232 - Draw a graph of disintegration rate against time. (3mks)
- Determine the half life of Bismuth. (1mk)
- Draw a graph of disintegration rate against time. (3mks)
MARKING SCHEME
-
- Element A√1 has six electrons in the outermost energy level and can therefore accept two more√1 electrons to achieve an octet.
- F
- Alkaline Earth metals
- Amphoteric
- E is more reactive than H√1 E has a larger electron affinity than H due to its small atomic size√.1
- B(s) + Cl2(q) → BCI2(s) √1
- G has a small atomic radius than F√.1. The effective nuclear charge increase due to increase in number of protons. √1
- Oxide of B in water forms an alkaline √ solution. (Turns red litmus blue). Oxide of D in water forms an acidic √ solution. (Turns blue litmus red)
-
-
- State of reaction where the rate of forward and backward reaction are the same but in opposite direction.
- Intensity of orange colour increases √1. Addition of H+ removes OH- hence backward reaction is favoured.√1. OR Equilbrium shifts to the left.
- Nitrous acid / HNO2.
-
- Equilbrium shifts to the right √1. Forward reaction is favoured since it is exothermic. √1.
- Equilbrium shifts to the left √1. Backward reaction is favoured.√1. OR favours the direction with fewer molecules due to the decrease in number of molecules.
- No effect
- Rate of reaction decreases. / Reduces. √½ . Decrease in temperature leads to a decrease in Kinetic √ energy of the molecules hence less effective collisions.
Molar mass of NH3 = 17√ ½
Molar mass of NO = 30 √ ½
Moles of NH3 = 1000,000 = 5882.35
17
Moles of NO = Moleration 1:1 = 5882.35
Mass of NO = 5882.35 X 30√ ½ = 176470.58
% yield of NO = 160 000 X 100 √ ½
176470
= 90.667%
Alternative
160 X 17 X 100
30 X 100
= 90.667%
-
-
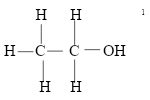
- Name: Ethanol √1
Structural formula
-
- Step II
Reagent: Acidified KMnO4 / K2Cr2O7 √ ½
Condition: Warming √ ½ - Step I:
Reagent: Concentrated H2SO4 √ ½
Conditions: Temp 160°C - 180°C. √ ½
- Step II
-
- Step I: - Dehydration √ ½
- Step II - Oxidation √ ½
- Step (IV) – Addition Polymerisation. ½
- Q - NaOH / Sodiumhydroxide / Sodalime. √ ½
- CH3CH2OH + CH3CH2COOH CH3CH2COOCH2CH3+H2O
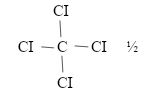
- Name: Tetrachloromethane √ ½
-
- K - Polyethene.
- n (24+4) = 28n √1 = 3000√1
28n = 84000
- Name: Ethanol √1
-
- 38°C – 22°C = 16°C = 16K
- Mass of methanol burnt = 0.32g or 0.00032Kg.
88.10 – 87.78 = 0.32g. - Moles of methanol = 0.32 = 0.01 moles.1
32 - ∆H = MC∆H
= 100 X 4.2KJ/Kg 1K x 16 K.
1000
= - 6.72KJ. - 0.01 moles ⟹ 6.72KJ
1 mole ⟶ 6.72 X 1
0.01
= - 672KJ/mol. - 2CH3OH(I) + 3O2(g) 2CO2(g) + 4H2O(l) ∆H = -672KJ/mol.
-
-
- Concentrated sulphuric(VI) acid and sodium chloride.
- Concentrated sulphuric(VI) acid
- Grey iron powder turns green / solid glows red.
- Fe(s) + 2HCl(g) FeCl2(s) + H2(g)
- Open a bottle of concentrated ammonia and place it near HCl dense white fumes of ammonium chloride are observed.
-
- Hydrogen gas
- To prevent an explosion since a mixture of hydrogen and oxygen explodes.
-
- To prevent suck back.
- To increase surface area for dissolution of hydrogen chloride in water.
- The flame will go off.
-
-
-
- N √1 - It has the highest negative √1 electrode potential.
- G and N
- No reaction √1. E value is negative √1 Or M is below N in the reactivity series hence cannot displace N+ ions from the solution.
-
-
- A - Anode
- B - Cathode
- 4OH-(aq) → O2(g) + 4e + 2H2O(l)
- Insert a burning splint in gas K. It is extinguished with a pop sound.
-
- The volume of hydrogen gas produced is higher than the volume of oxygen produced
OR volume of hydrogen is twice the volume of oxygen. - The bulb is brighter √ ½ with sulphuric (VI) acid, Sulphuric (VI) acid is a strong √ ½ acid hence its fully √ ½ ionized while ethanoic acid is a weak acid √ thus partially √ ionized.
- The volume of hydrogen gas produced is higher than the volume of oxygen produced
-
-
-
- Radioactivity is the process where an unstable nuclide breaks up to yield another nuclide of different composition with emission of particles and energy.
-
Chemical reactions Nuclear reactions - Takes place on the outer energy levels containing the valence electrons. - Takes place within the nucleus and involves neutrons and protons. - The heat energy released is much less. - Releases large amounts of heat energy. - Are affected by environmental factors such as temperature and pressure. Not affected by environmental factors such as temperature. -
- Penetrating power of radiation.
- B - Gamma rays
- They have the highest penetrating power.
- They are stopped by a thick lead block
-
-
- Step I - Beta radiation
- Step V - Alpha radiation
- 214 84 PO → 210 82 Pb + 4 2 He
-
-
-
- Used to destroy cancerous tissue
- Sterilisation of surgical instruments
- Used to monitor growth in bones and healing of fractures. (Any one)
- Agriculture.
- In monitoring photosynthesis and related process.
- Absorption of phosphate fertilisers.
-
- Dangers of radioactivity.
- Long exposure to low dosages of radiations can cause genetic mutation in living tissues.
- Disposal of nuclear wastes leads to environmental pollution.
- Radioisotopes can be used as weapons of mass destruction.
Download Chemistry Paper 2 Questions and Answers - Lugari Constituency Joint Pre Mock Exams 2023.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students