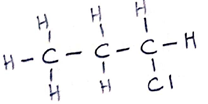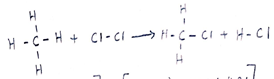INSTRUCTIONS TO CANDIDATES
- Answer all the questions.
- Mathematical tables and electronic calculators may be used
- All working must be clearly shown where necessary.
- State and explain the change in mass that occur when the following substances are separately heated in open crucibles.
- Copper metal (1½ marks)
- Copper (II) nitrate (1½ marks)
-
- State Graham’s law of diffusion (1 mark)
- A volume of 120cm3 of nitrogen gas diffused through a membrane in 40 seconds, how long will 240cm3 of carbon (IV) oxide defuse through the same membrane? (2 marks)
- A reaction of Propane with chlorine gas gave a compound of formula C3H7Cl.
- What condition is necessary for the above reaction to take place. (1 mark)
- Draw the structural formula of the compound C3H7Cl (2 marks)
- Name a gas which is used together with Oxygen in welding. (1 mark)
- Study the table below and answer the questions that follow.
(The letters are not the actual symbols of the elements)
Element B C D E F Atomic number 18 5 3 5 20 Mass number 40 10 7 11 40 - Which two letters represent the same elements? Give reason. (2 marks)
- Give the number of neutrons in an atom of element D. (Show your working) (1 mark)
- A hydrated salt of copper has the formula CuSO4.nH2O. About 25g of the salt was heated until all the water evaporated. If the mass of the anhydrous salt is 16.0g, find the value of n. (Cu = 64.0, S = 32.0, O = 16.0, H = 1)
(3 marks) - The table below shows the pH values of the solutions I, II, III and IV
Solution I II III IV pH 2 7 11 14 - Which solution is likely to be that of calcium hydroxide? (1 mark)
- Select the solution in which a sample of aluminum oxide is likely to dissolve. Give a reason for your answer.
(2 marks) - Select a pair of solutions that would likely give a pH of 7 when equal volumes are reacted with each other.
(1 mark)
- Sodium chloride has a higher melting point than hydrogen chloride, explain. (2 marks)
- Study the table below and answer the questions that follow
Place J, K, L and M in the appropriate categories from the following:Substance M.pt °C B.pt °C Electrical conductivity in solid state Electrical conductivity in molten state J 365 463 Nil Nil K 1323 2773 Good Good L 1046 1680 Nil Good M 2156 2776 Nil Nil - Metallic solid ______________________________ (1 mark)
- Covalent network solid ________________________ (1 mark)
- Ionic solid __________________________________ (1 mark)
- Covalent molecular solid_______________________ (1 mark)
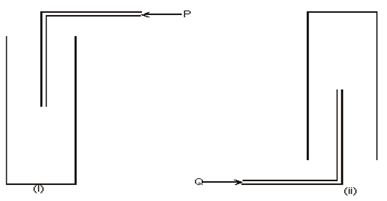
- The diagram below shows how two gases, P and Q were collected.
- Name the two methods used. (2 marks)
- State properties of P and Q that enable them to be collected through the methods shown. (2 marks)
- Study the information in the table below and answer the questions in the table below and answer the questions below the table
Calculate the enthalpy change of the following reactionBond Bond Energy (KJmol-1) C-H 414 Cl-Cl 244 C-Cl 326 H-Cl 431
U.V. light
CH4(g) + Cl2(g)CH3Cl(g) + HCl(g) (3 marks )
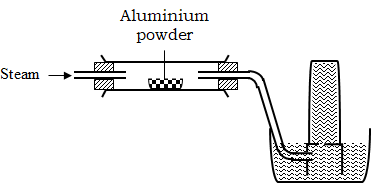
- Study the diagram below used to investigate the property of steam on aluminium
- Explain why no gas was collected in the set up above. (1 mark)
- Explain why the reaction between aluminium and steam stops after a short time. (2 marks)
- A pupil analyzed a commercial vinegar solution by titration and found that 24.5cm3 of 0.09 M sodium hydroxide solution was required for titration of 10cm3 of vinegar. Calculate the molarity of ethanoic acid CH3COOH in vinegar. (3 marks)
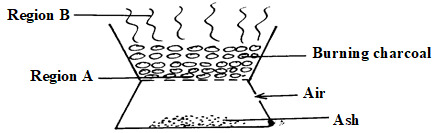
- The diagram below shows a ‘jiko’ when in use. Study it and answer the questions that follow
- Identify the gas formed at region B (1 mark)
- Using an equation, explain what happens at region A (2 marks)
- Sodium chloride is contaminated with copper (II) oxide. Explain how pure sodium chloride can be obtained from the mixture. (3 marks)
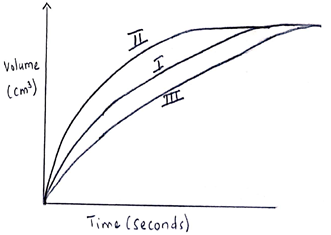
- The table below gives three experiments on the reaction of excess sulphuric (VI) acid and 0.5g of zinc done under different conditions. In each the volume of gas was recorded at different time intervals.
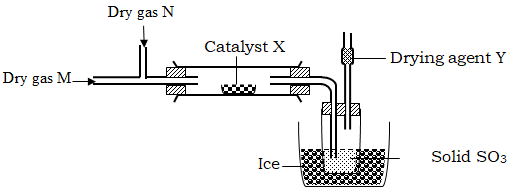
On the axis below, draw and label the three curves that could be obtained from such results. (3 marks)Experiment Form of zinc Sulphuric (VI) acid solution I Powder 0.8M II Powder 1.0M III Granules 0.8M - The set-up of apparatus below used to prepare sulphur (VI) oxide:
- Name Gas N………………………… and Gas M………………………….. (1 mark)
- Catalyst X …………………………………………………………………... (1 mark)
- Why is it necessary to use drying agent Y? (1 mark)
- State and explain the observation made when chlorine gas is bubbled into potassium iodide solution. (2 marks)
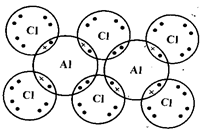
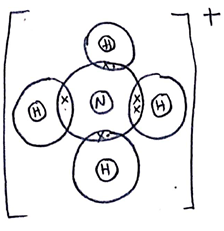
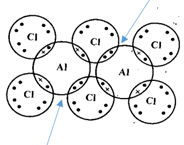
- The diagram below shows an arrangement of electrons in Aluminium chloride dimer.
- Write down the formula of the above molecule. (1 mark)
- On the diagram, indicate using an arrow the dative bond. (1 mark)
- When Magnesium metal is burnt in air, it reacts with both oxygen and nitrogen gases giving a white solid with black specs. Write two equations for the reactions that take place. (2 marks)
- Below is a representation of an electrochemical cell.
Pb(s)|Pb2+(aq)||Ag+(aq)|Ag(s)- What does || represent? (1 mark)
- Given the following:
Eᶿ (volts)
Pb2+(aq) + 2e- → Pb(s) -0.13
Ag+(aq) + e- → Ag(s) +0.80
Calculate the E.M.F of the electrochemical cell (2 marks)
- When hot concentrated nitric (V) acid is added to sulphur, a red – brown gas and a colourless liquid are formed.
- Write an equation for the reaction. (1 mark)
- Identify the oxidizing agent in the reaction above. (1 mark)
- State one environmental hazard of the nitrogen compounds. (1 mark)
- Draw the dot (.) and cross (x) structure of:
- Carbon (II) oxide - CO (2 marks)
- Ammonium ion - NH4+ (2 marks)
- Using sodium hydroxide solution, describe a chemical test that can be used to distinguish between copper (II) ions and iron (II) ions. (3 marks)
- State and explain what would be observed if concentrated sulphuric (VI) acid is added to:
- Sugar crystals (1 ½ marks)
- Copper (II) sulphate crystals (1 ½ marks)
- When 100cm3 of 0.5 M sulphuric acid solution, H2SO4, react with 100cm3 of 1 M sodium hydroxide solution, NaOH, the temperature rises by 6.85 Kelvins.
(Density = 1.0g/cm3, specific heat capacity = 4.2kJkg-1K-1)
Calculate the molar heat of neutralization described by the equation: (3 marks)
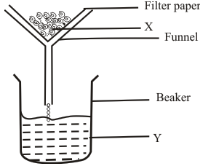
H2SO4(aq) + 2NaOH(aq) → Na2SO4(aq) + 2H2O(l) - Filtration is carried out in the apparatus shown
- Name X ……………………………………………………………………… (12 mark)
- State one property that makes it possible to separate mixtures using filtration. (12 mark)
- Calculate the oxidation numbers of sulphur in the following species: (3 marks)
- SO32-
- SO3
- S2O32-

MARKING SCHEME
-
- Increase in mass. Copper metal combines with oxygen in air to form copper (II) oxide
- Decrease in mass. Copper (II) nitrate decomposes to form copper (II) oxide, nitrogen (IV) oxide and oxygen gas. The gaseous products escape to the atmosphere.
-
- Graham’s law states that the rate of diffusion of a gas is inversely proportional to the square root of its density given that the temperature and pressure are kept constant.
- R.M.M N2 = 14 × 2 = 28
R.M.M CO2 = 12 + ( 16 × 2) = 44
Rate of diffusion of N2
120cm3 = 3cm3/s
40s
Rco2 = 3 × √28 = 2.393cm3/s
√44
1s → 2.393cm3
? → 240cm3
240 × 1 = 100.3s
2.393
-
- UV light or sunlight
-
- Acetylene or ethyne
-
- C and E. They have the same atomic number.
- 7 – 4 = 3 neutrons
- Mass of water 25 − 16 = 9g
CuSO4. 5H2OCompound CuSO4 H2O Mass
R.F.M/R.M.M
Moles
Mole ratio16.0
160
16/160 = 0.1
0.1/0.1 = 19.0
18
9/18 = 0.5
0.5/0.1 = 5
n = 5 -
- III
- I and IV. Aluminium oxide is amphoteric.
- I and IV
- Sodium chloride has strong ionic bonds in a giant ionic structure that require more energy to break compared to the weak Van der Waals forces of attraction between hydrogen chloride molecules.
-
- K
- M
- L
- J
-
-
- Downward delivery or upward displacement method
- Upward delivery or downward displacement method
-
- the gas is denser/heavier than air
- the gas is less dense/lighter than air.
-
-
[4(414) + 244] − [3(414) + 326 + 431]
= 1900 − 1999
= − 99 kJmol− -
- There is no heating in the set up.
- The formation of aluminium oxide coating that prevents further reaction.
-
0.09 mol → 1000cm3
? → 24.5cm3
24.5 × 0.09 = 0.002205 moles
1000
M:R 1:1
∴ 0.002205 moles of CH3COOH
0.002205mol → 1cm3
? → 1000cm3
1000 × 0.002205 = 0.2205M
10 -
- carbon (IV) oxide / CO2
- C(s) + O2(g) → CO2(g) Carbon (charcoal) reacts with sufficient air to form carbon (IV) oxide.
- Add water to the mixture and stir. Sodium chloride dissolves but not copper (II) oxide. Filter the mixture to obtain sodium chloride solution as the filtrate. Heat the filtrate to evaporate the water and remain with sodium chloride.
-
-
- Gas N – sulphur (IV) oxide ; gas M – oxygen OR gas N – oxygen ; gas M – sulphur (IV) oxide.
- Platinum
- Sulphur (VI) oxide crystals are readily hydrolyzed by water.
- A brown solution is formed. Chlorine displaces iodide ions from the solution to form iodine.
-
- Al2Cl3
-
- 2Mg(s) + O2(g) → 2MgO(s) and 3Mg(s) + N2(g) → Mg3N2(s)
- Ecell = Ered − Eoxy
= +0.80 − (−0.13)
= + 0.80 + 0.13
= + 0.93V -
- 6HNO3 (aq) + S (s) H2SO4 (aq) + 6NO2 (g) + 2H2O (l)
- concentrated nitric (V) acid
-
- acid rain
- Eutrophication
-
-
- Add sodium hydroxide drop wise until in excess to separate test tubes with solutions containing copper (II) ions and iron (II) ions. A green precipitate is formed in the test tube with iron (II) ions while a pale blue precipitate is formed in test tube containing copper (II) ions.
-
- White sugar crystals are charred into a black mass. Concentrated sulphuric (VI) acid is a strong dehydrating agent and removed elements of water from sugar.
- Blue crystals turn white. . Concentrated sulphuric (VI) acid is a strong dehydrating agent and removed the water of crystalisation from the copper (II) sulphate crystals.
- mass = 200cm3 × 1g/cm3 = 200g
ΔH = mcΔT
= 200/1000 × 4.2 × 6.85 = 5.754kJ
Moles of NaOH used
1 mole → 1000cm3
? → 100cm3
100 × 1 = 0.1moles
1000
0.1 moles → 5.754kJ
1 mole → ?
1 × 5.754 = 57.54kJ/mol
0.1
= − 57.54kJ/mol -
- residue
- Immiscibility
-
- S + 3(−2) = −2
S – 6 = −2
S = + 4 - S + 3(−2) = 0
S – 6 = 0
S = + 6 - 2S + 3(−2) = −2
2S – 6 = −2
2S = +4
S = + 2
- S + 3(−2) = −2
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions and Answers - Pavement Mock Exams 2021/2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students