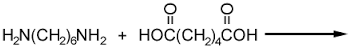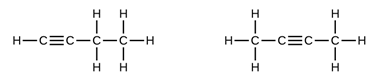INSTRUCTIONS TO CANDIDATES
- Answer all questions
- Electronic calculators may be used
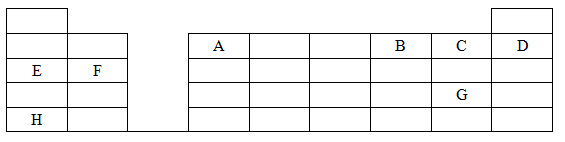
- The grid below shows part of the periodic table. Study it and answer the questions that follow. The letters are not the actual symbols of the elements.
- Give the name of the family to which element F belongs. (1mark)
- Identify an element which forms a stable divalent anion (1mark)
- Give the formula of:
- The compound formed between A and B (½mark)
- The sulphate of H (½mark)
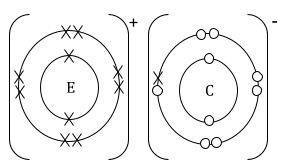
- Using dot (.) and cross(x) diagram, show the bonding in the compound formed between E and C. (2marks)
- Compare the atomic radii of elements C and D. Explain. (2marks)
- Select the element that has the lowest ionization energy. Explain. (1mark)
- 0.081g of element A reacts with 20cm3 of dilute hydrochloric acid. Calculate the molarity of the hydrochloric acid. (R.A.M = 27) (3marks)
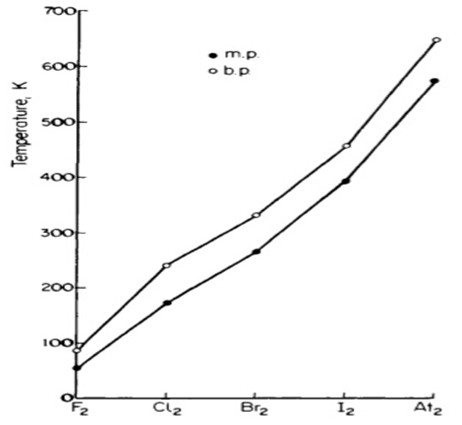
- The graph below shows the trend of the melting points and the boiling points of the family to which element G belongs. Explain the trend in the melting points. (2marks)
-
-
- What is a fuel? (1 mark)
- Calculate the heating value of propane, C3H8, given that its molar enthalpy of combustion is 2200 kJ mol-1. . (2 marks) (C=12, H=1)
-
- Define molar enthalpy of combustion. (1 mark)
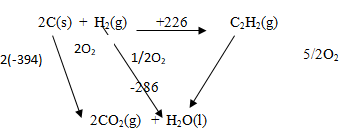
- Use the information provided by the thermochemical equations below to calculate the molar enthalpy of combustion of ethyne. (3 marks)
C(s) + O2(g) → CO2(g) ∆H= -394 kJ mol-1
H2(g) + ½O2(g) → H2O(g) ∆H= -286 kJ mol-1
2C(s) + H2(g) → C2H2(g) ∆H= +226 kJ mol-1
- Study the data given below and answer the questions that follow.
Substance/ion Enthalpy change CaCl2(s) Lattice energy = −2237 kJ mol-1 Ca2+(g) Hydration energy = −1650 kJ mol-1 Cl−(g) Hydration energy = −364 kJ mol-1 - Determine the molar enthalpy of solution of calcium chloride in water. (2 marks)
- Draw an energy level diagram for the dissolution of calcium chloride in water. (3 marks)
-
-
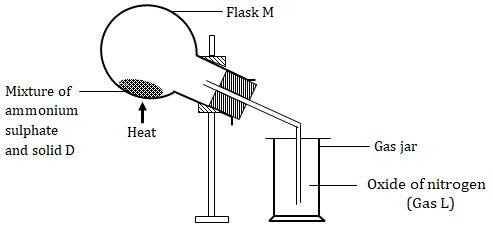
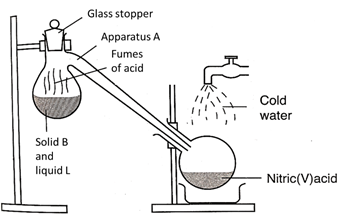
- The diagram below shows the preparation of an oxide of nitrogen. Study the diagram and answer the questions that follow.
- Name the solid D. (1mark)
- Write the equation for the reaction in the flask M (1mark)
- Burning magnesium ribbon was lowered into the gas jar containing gas L. State and explain the observations made. (2marks)
- Write the two equations for the reactions taking place in (iii) above. (2marks)
- A drop of water was added to the product formed in (iii) above. A colourless gas K with a choking smell was produced. Write the chemical equation for the production of gas K (1mark)
-
- Give two uses for nitric (V) acid. (2marks)
- A factory uses nitric (V) acid and ammonia gas as the only reactants for the preparation of ammonium nitrate. If the daily production of the ammonium nitrate is 4800kg, calculate the mass of ammonia gas used daily in kg. (N=14.0, O = 16.0 H= 1.0) (3 marks)
- The diagram below shows the preparation of an oxide of nitrogen. Study the diagram and answer the questions that follow.
- A group of form four students of Cockelbet Secondary School carried out an experiment to determine the solubility of potassium chlorate. The table below shows the results obtained.
Total volume of water added(cm3) 10.0 .20.0 30.0 40.0 50.0 Mass of KClO3(g) 5.0 5.0 5.0 5.0 5.0 Temperature at which crystals appear(°C) 80.0 65.0 55.0 45.0 30.0 Solubility of KClO3 (g/100gH2O) - Complete the table to show the solubility of KClO3 at different temperatures. (3marks)
- Plot a graph of mass of KClO3 per 100g water against temperature at which crystals form. (3marks)
- From the graph, determine ;
- The solubility of KClO3 at 40°C. (1mark
- The temperature at which the solubility of KClO3 is 35g/100g water. (1mark)
- Explain the shape of the graph. (1mark)
- State one application of solubility and solubility curves. (1mark)
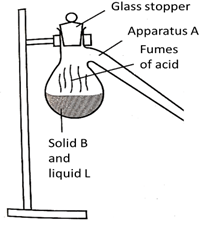
- The set up below is used to prepare Nitric (V) acid in the laboratory. Study it and answer the questions that follow.
- Complete the set-up. (3 marks)
- Name substances
- B………………………
- L ………………………(2 marks)
- Name apparatus A……………………………………………………………… (1 mark)
- Write an equation between substances B and L (1 mark)
- During preparation of hydrogen gas Nitric (V) acid is not used. Explain (2 marks)
- Give two commercial use of Nitrogen (2 marks)
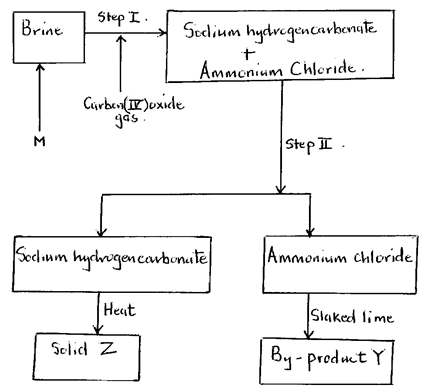
- The flow chart below is a simplified version of the steps in the manufacture of Sodium carbonate.
- Name three main raw materials of the above process. (3 marks)
- Name substance M. (1 mark)
- Name the process taking place in step II. (1 mark)
- Write a balanced chemical equation to show how product Y is formed. (1 mark)
- 98.123 Kg of Sodium hydrogen carbonate was manufactured in this process.
- Write a chemical equation for the formation of solid Z. (1 mark)
- Determine in Kilograms, how much of solid Z was produced in this process.
(Na = 23, H = 1, O = 16, C = 12) (3 marks)
-
- Draw and name all the isomers of C4H6 (2marks)
- The following monomers react to form a polymer:
Draw and name the structure of the polymer (2marks) - Chlorofluorocarbons have a wide range of uses. However they have been linked to depletion of ozone.
- Explain the problem caused by depletion of ozone layer. (1mark)
- Give one other environmental problem caused by chlorofluorocarbons. (1mark)
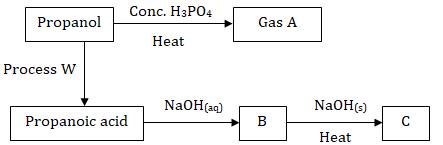
- Study the flow chart below and use it to answer the questions that follow.
- Name:
- Process W (½mark)
- Substances A, B and C (1½marks)
- Write an equation for the combustion of substance C (1mark)
- Give one use of gas A (1mark)
- Name:
- Explain how a sample of propan-1-ol could be distinguished from a sample of propanoic acid by means of a chemical reaction (2marks)

MARKING SCHEME
-
- Alkaline-earth metals (1mark)
- B (1mark)
-
- A2B3 (½mk)
- H2SO4 (½mark)
-
Distribution of electrons (1mk) and charges (1mk)
No label (0mk) -
- D has a smaller atomic size than C//C has larger atomic size than D (1mark)
- D has more protons than C//D has stronger nuclear charge than C (1mark)
- H (½mk). Loses electrons most readily/largest atomic radius/weakest nuclear charge (explanation ½mk)
- 2A(s) + 6HCl(aq) → 2ACl3(aq) + 3H2(g) 1mark
Moles of A = 0.081 = 0.003moles ½mark
27
Mole ratio 1:3
Moles of HCl = 0.003 x 3 = 0.009moles ½mark
0.009×1000 ½mk = 0.45M ½mark
20 - The melting point increase with increase in atomic number. 1mark
This is due to increase in the strength of van der waal’s forces (inter-molecular forces) 1mark
-
-
- A fuel is a substance that can be used as a source of energy. (1 mark)
OR A substance that produces useful energy when it undergoes a chemical or nuclear reaction. - Molar mass of C3H8 = 12 x 3 + 1 x 8= 44 g/mol-1 ½mark
Heating value = 2200 1 mark
44
= 50 kJ/g ½mark
- A fuel is a substance that can be used as a source of energy. (1 mark)
-
- molar enthalpy of combustion is the heat change when one mole of a substance is burnt completely in oxygen. (1mark)
-
∆HC(ethyne)= −(+226) + 2(−394)+ (−286) (1 mark)
= −226−788−286
= −1300 kJ/mol-1 (1 mark)
-
- ∆Hsoln = −(−2237) + −1650)+ (2 x −364) (1 mark)
= −141 kJ mol-1 (1 mark) -
(3 marks)
- ∆Hsoln = −(−2237) + −1650)+ (2 x −364) (1 mark)
-
-
-
- Potassium nitrate (Reject KNO3) (1mark)
- (NH4)2SO4 (S) + 2KNO3(s) → K2SO4(s) +2H2O(l) +N2O(g) (1mk)
- The magnesium ribbon continues to burn with a dazzling flame forming white solid .(1mark)
This is because burning magnesium produces sufficient heat to dissociate nitrogen (I) oxide into oxygen and nitrogen gases (½mk). The oxygen supports combustion of magnesium to form white magnesium oxide while nitrogen combines with hot magnesium metal to form white magnesium nitride. (½mark) -
- 2Mg(s) + O2(s) → 2MgO(s) (1 mark)
- 3Mg(s) + N2(g) → Mg3N2(s) (1 mark)
- Mg3N2(s) + 6H2O(l) → 3Mg(OH)2(s) + 2NH3(g) (1mark)
-
-
- Manufacture of nitrogenous fertilizers such as ammonium nitrate. (any two correct uses)
- Manufacture of textile dyes
- Manufacture of explosives
- HNO3(l) + NH3(g) → NH4NO3(s) √ 1mk
Mole ratio: 1:1:1
Molar mass of ammonia = 17g
Molar mass of ammonium nitrate = 80g √½mk
80g NH4NO3 → 17g
4800000g → 17x4800000 = 1020000g
80 √1mk
= 1020kg of ammonia gas. √½mk
-
-
-
- Total volume of water added(cm3)
NB: √½mk for expressing solubility values to the same number of decimal pointsTotal volume of water added(cm3) 10.0 .20.0 30.0 40.0 50.0 Mass of KClO3(g) 5.0 5.0 5.0 5.0 5.0 Temperature at which crystals appear(°C) 80.0 65.0 55.0 45.0 30.0 Solubility of KClO3 (g/100gH2O) 50.0✓½ 25.0✓½ 16.7✓½ 12.5✓½ 10.0✓½ - Graph marking points:
- scale (graph to cover ˃= ½ grid)√½
- Labeling of axes√½
- Plotting of points √1
- Curve (smooth) √1
-
- 11g/100gH2O √1
- 72°C (± 0.5)√1
- Solubility of KClO3 increases with increase in temperature/more KClO3 dissolves as temperature rises√1
-
- Extraction of soda ash(Sodium carbonate) from Trona√1
- Extraction of common salt(sodium chloride). Etc.
- Total volume of water added(cm3)
-
-
-
- B – Sodium Nitrate/KNO3✔1
- L – Concentrated H2SO4✔1
- A – Retort Flask✔1
- NaNO3(s) + H2SO4(l) → HNO3(aq) + NaHSO4(aq) ✔1
- HNO3 is a powerful✔1 oxidizing agent. It oxides H2 ✔1 to water.
-
- Semen storage for artificial insemination.✔1
- Manufacture of ammonia in the Haber process✔1
-
-
-
- Ammonia gas
- Limestone / calcium carbonate
- Brine / concentrated sodium chloride
- Ammonia gas
- Filtration
- 2NH4Cl(aq) + Ca(OH)2aq → CaCl2 + 2H2O(l) + 2NH3
-
- 2NaHCO3(s) → Na2CO3(s) + H2O(l) + CO2(g)
- From equation: 2 moles NaHCO3 produces 1 mole Na2CO3
R.F.M of NaHCO3 is (1 x 23) + (1x1) + (1 x 12) + (3 x16) = 84
(84g/1000g x 1) kg is 0.084 kg
2 mole NaHCO3 is 0.084 x 0.168 kg
R.F.M of Na2CO3 is (2 x 23) + (1 x 12) + (3 x 16) = 106
(106g/1000g x 1 kg) is 0.106 kg
If, 0.168 kg NaHCO3 produces 0.106 kg
98.128 produces (98.128 x 0.106 )
0.168
= 61.9109Kg Solid Z
-
-
-
But – 1 – yne But – 2 – yne (structure ½mk, name ½mk total 2mks) - Nylon 6,6. 1mk
1mk
-
- Harmful rays from the sun reaches the earth’s surface causing harm to humans 1mk
- Contribute to global warming 1mk
-
-
- Oxidation (½mk)
-
- A: Propene
- B: Sodium propanoate
- C: Ethane (1½mk)
- 2C2H6 + 7O2 → 4CO2 + 6H2O (1mk)
-
- Used in manufacture of polypropene used in making ropes, plastic crates, buckets, basins, plastic tables and chairs etc
- Manufacture of other chemicals e.g. acetone, propan-1-ol etc
- Used as an alternative to ethyne(acetylene) in welding flame
-
- Add sodium carbonate/ sodium hydrogen carbonate to separate samples of each 1mk. Propanoic acid produces bubbles of a gas/effervescence/fizzling while propan-1-ol does not 1mk
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 2 Questions and Answers - Pavement Mock Exams 2021/2022.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students