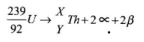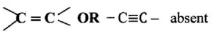INSTRUCTIONS TO CANDIDATES
- Answer all questions.
- All working MUST be clearly shown where necessary.
- Non-programmable silent electronic calculator and KNEC Mathematical tables may be used, except where stated otherwise
- Candidates should answer the questions in English
- Table 1 shows the atomic numbers and the first ionisation energies of three elements The letters are not actual symbols of the elements. Use it to answer the questions that follow
Table 1
Element Atomic Number First ionisation energy kjmol− A 3 519 B 11 494 C 19 418 - Explain the trend in first ionisation energy from A to C. (2 marks)
- Write the electronic configuration for the ion of C. (1 mark)
- Calculate the values of X and Y in the following nuclear equation. (2 marks)
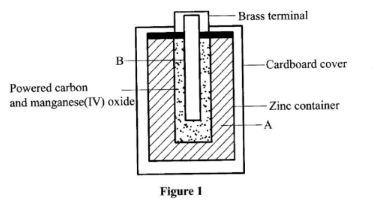
- The diagram in Figure 1 shows a section of a dry cell. Study it and answer the questions that follow.
- Name the part labelled B. (1 mark)
- The part labelled A is a paste. Give a reason why it is not used in dry form. (1 mark)
- What is the purpose of the zinc container? (1 mark)
- The empirical formula of lead(II) oxide was determined by passing excess dry hydrogen gas over 6.69 g of heated lead(II)oxide.
- What was the purpose of using excess dry hydrogen gas? (2 marks)
- The mass of lead was found to be 6.21g. Determine the empirical formula of the oxide. (Pb = 207,0:0 - 16.0)
(2 marks)
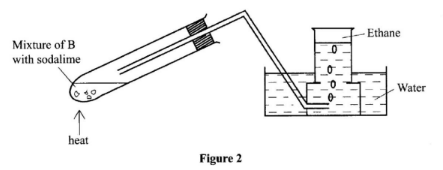
- The set-up in Figure 2 was used to prepare a sample of ethane gas. Study it and answer the questions that follow.
- Name B ................................. (1 mark)
- Write an equation for the complete combustion of ethane. (1 mark)
- State one use of ethane. (1 mark)
-
- State Charles' Law. (1 mark)
- Explain why the pressure of a fixed mass of a gas increases, when the volume of the gas is reduced at constant temperature. (2 marks)
- A sample of water is suspected to contain sulphate ions. Describe an experiment that can be carried out to determine the presence of sulphate ions. (3 marks)
-
- State one characteristic of a reaction where equilibrium has been attained. (1 mark)
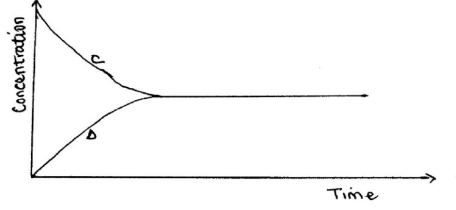
- The following equation is in a state of equilibrium:
CD
Use it to sketch a graphical representation of concentration against time in seconds for the equilibrium.
(2 marks)
- Copper(II)ions react with excess aqueous ammonia to form a complex ion.
-
- Write an equation for the reaction that forms the complex ion. (1 mark)
- Name the complex ion. (1 mark)
- Explain why CH4 is not acidic while HCl is acidic yet both compounds contain hydrogen. (1 mark)
-
- 20cm3 of ethanoic acid was diluted to 400cm3 of solution. Calculate the concentration of the solution in moles per litre. (C=12.0; H=10: 0=16.0) (Density of ethanoic acid =1.05 g/cm3) (3 marks)
- An oxide of element Khas the formula K2O5
- Determine the oxidation number of K. (1 mark)
- To which group of the periodic table does K belong? (1 mark)
- Potassium nitrate liberates oxygen gas when heated. Draw a diagram of a set-up that shows heating of potassium nitrate and collection of oxygen gas. (3 marks)
- Explain the observation made when chlorine gas is passed through a solution of potassium iodide. (3 marks)
- Using the elements chlorine, calcium and phosphorus:
- Select clements that will form an oxide whose aqueous solution has a pH less than 7. (1 mark)
- Write an equation for the reaction between calcium oxide and dilute hydrochloric acid. (1 mark)
- Give one use of calcium oxide. (1 mark)
- Starting with copper, describe how a pure sample of copper(IT) carbonate can be prepared (3 marks)
- In an experiment, concentrated nitric(V) acid was reacted with iron(II) sulphate. State and explain the observations made. (2 marks)
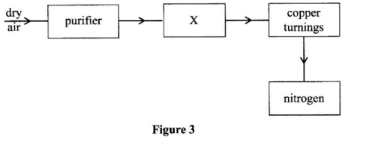
- The flow chart in Figure 3 shows the process of obtaining a sample of nitrogen gas. Study it and answer the questions that follow.
- Identify X (1 mark)
- Write an equation for the reaction with heated copper turnings. (1 mark)
- Name an impurity in the sample of nitrogen gas. (1 mark)
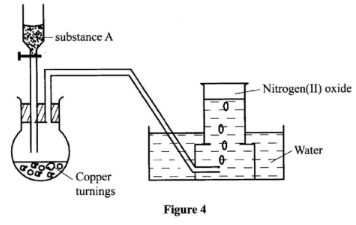
- The set-up in Figure 4 can be used to prepare nitrogen(II)oxide. Use it to answer the questions that follow.
- Name substance A. (1 mark)
- When the gas jar containing nitrogen(II)oxide is exposed to air, a brown colour is observed. Explain. (1 mark)
- Write an equation for the reaction which occurred in the flask. (1 mark)
- The following procedure was used to investigate the temperature changes that occur when sodium hydroxide solution is added to dilute hydrochloric acid.
- Place the acid in a glass beaker and record its temperature.
- Add a known volume of sodium hydroxide solution.
- Stir the mixture and record the highest temperature reached.
- Repeat steps (ii) and (iii) with different volumes of sodium hydroxide solution.
- State two factors that must be kept constant in this experiment (1 mark)
- Explain how the use of a polystyrene cup will affect the results. (1 mark)
- Study the flow chart in Figure 5 and answer the questions that follow.
- Identify substances K and L. (2 marks)
- Name one reagent that can be used to carry out process J. (1 mark)
- The atomic numbers of some elements P, Q, R and S are 6, 8, 12 and 17 respectively
- Draw the dot and cross (X) diagrams for the compounds formed when:
- R and Q react (1 mark)
- P and S react. (1 mark)
- Explain why the melting point of the compound formed by P and S is lower than that formed by R and Q.
(1 mark)
- Draw the dot and cross (X) diagrams for the compounds formed when:
-
- What is an inert electrode? (1 mark)
- State the products formed when brine is electrolysed using inert electrodes.
Anode: (1 mark)
Cathode: (1 mark)
- Explain how a student can establish whether a liquid sample extracted from a plant is pure. (2 marks)
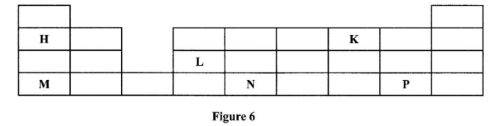
- Figure 6 shows part of the periodic table. The letters are not the actual symbols of the elements. Study it and answer the questions that follow.
- Write an equation for the reaction between M and K. (1 mark)
- Select the element which can form an ion with a charge of +3. (1 mark)
- An element J has atomic number 15. Indicate with a tick (V), on the part of the periodic table the position of J.
(1 mark)
- In terms of structure and bonding, explain why graphite is used as a lubricant in machines. (3 marks)
-
- What is meant by the term bleaching? (1 mark)
- Write the formula of the bleaching agent formed when chlorine gas reacts with aqueous sodium hydroxide.
(1 mark) - State the role of chlorine in water treatment. (1 mark)
-
- Name two ores in which sodium occurs. (1 mark)
- During extraction of sodium using the down's process, calcium chloride is added to the ore. Give a reason for the addition of calcium chloride. (1 mark)
- State two uses of sodium. (1 mark)
- When an aqueous solution of compound X was mixed with a few drops of bromine water, the colour of the mixture remained yellow. When another portion of solution X was reacted with acidified potassium dichromate(VI), the colour of the mixture changed from orange to green.
- What conclusion can be made from the use of:
- bromine water? (1 mark)
- acidified potassium dichromate(VT)? (1 mark)
- Solution X was reacted with a piece of a metal and a colourless gas was produced. Describe a simple experiment to identify the gas. (1 mark)
- What conclusion can be made from the use of:

MARKING SCHEME
-
- Ionisation energy decreases down the group 1 elements. ✓1
This is because atomic radii increases from A to C (down the group)/outermost clectron is far from nucleus hence requires less energy to be lost during reaction. ✓1 - Electron configuration of ion of C= 2.8.8 ✓1
- Ionisation energy decreases down the group 1 elements. ✓1
- x = 231 ✓1
y = 90 ✓1 -
- Carbon electrode (Anode)/Graphite electrode. ✓1
- To allow movement of ions / to have it as an electrolyte. When dry, the ions are immobile ✓1
- It is the cathode/negative electrode. ✓1
-
- To ensure all the oxide was reduced. ✓1
- Mass of oxygen 6,69 - 6.21 = 0.48g ✓½
Pb O
6.21 0.48 ✓½
207 16
0.03 0.03 ✓½
0.03 0.03
1 : 1 ✓½
E.F - PbO
-
- B is sodium propanoate ✓1
Accept formula : C2H5COONa/CH3CH2COONa - 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l) ✓1
-
- as Fuel
- production of ethene gas,
- production of hydrogen gas. ✓1
- B is sodium propanoate ✓1
- Charles' Law
- The volume of a fixed mass of a gas is directly proportional to the absolute temperature at constant pressure. ✓1
- As the volume decreases, there is increased bombardment/ collisions of the molecules against the walls of the container, hence increased pressure. ✓2
-
- Add aqueous barium nitrate / barium chloride to sample, ✓1 Followed by dilute nitric(V) acid or HCI; ✓1
- If white precipitate persists, then SO42− ions are present; ✓½
- If the precipitate dissolves then SO42− ions are absent. ✓½
OR - Add lead (II) nitrate solution
-
- The concentrations of reactants and products remain constant or Rate of forward reaction is equal to the rate of backward reaction. ✓1
-
✓2
(2 marks)
OR
-
-
- Cu(OH)2(s)+4NH3 (aq) → [CH(NH3)4]2+(aq) +2OH−(aq).
OR
Cu2+ (aq) + 4NH3 (aq) → [Cu(NH3)4]2+(aq) ✓1 - Tetraamine copper(II)ion ✓1
- Cu(OH)2(s)+4NH3 (aq) → [CH(NH3)4]2+(aq) +2OH−(aq).
- CH4 is a hydrocarbon, non-polar hence does not ionize in water.
HCl is polar hence ionizes in water.
-
- Molar mass of ethanoic acid (CH3COOH) = 60g ✓½
Mass of ethanoic acid = 20 x 1.05g/cm3
= 21g ✓½
Moles of ethanoic = 21/60
= 0.35 moles ✓½
Molarity = 0.35 ✓½
400/1000
=0.875M ✓1 -
- 2K + (5x-2)=0 ✓½
2K = +10
K=+5 ✓½ - Group 5 ✓1
- 2K + (5x-2)=0 ✓½
-
✓3
- Heating - 1 mark
- method of collection - 1 mark
- workability- 1 mark
- A dark grey/brown solid is deposited the solution turns black; ✓1
chlorine is more reactive/ a stronger oxidizing agent than iodine; ✓1 Therefore displaces it from a solution of its ions.✓1
OR
Cl2 (g) +2I− (aq) → 2 Cl−(aq) + I2 (S) -
- Phosphorus and chlorine ✓1
- CaO(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) ✓1
-
- used to neutralize acidic soil / liming; ✓1
- drying agent;
Any 1 correct @ 1 mark)
- To copper turnings, add 50% concentration H2SO4 or HNO3/Heat copper turnings to ✓1 form copper(II)oxide and add dilute H2SO4 or HNO3 or HCI; ✓1
To the resulting mixture, add excess sodium carbonate (soluble) ✓½
Filter mixture;
Rinse residue with water and dry between filter papers. ✓½ - The mixture changed from green to yellow / formation of a brown gas, ✓1 Iron(II) ions is oxidized by nitric) acid to Iron(III) ions/nitric(V) acid is reduced to nitrogen(II)oxide which is oxidized by oxygen to nitrogen(IV) oxide. ✓1
-
- Sodium hydroxide solution or Potassium hydroxide solution; ✓1
- 2Cu(s) + O2(g) → 2CuO(s) ✓1
- Argon, Neon, (Inert gases) ✓1
-
- Moderately concentration nitric(V) acid/50% concentrated nitric(V) acid. ✓1
- Colourless nitrogen(II)oxide (NO) is oxidized to brown nitrogen(IV) oxide (NO2). ✓1
- 3Cu(s) + 8HNO3 (aq) → 3Cu(NO3)2 (aq)+ 4H2O +2NO ✓1
-
-
- Concentration of acid and base; ✓½
- Volume of acid used. ✓½
-
- Improves accuracy; ✓½
- Polystyrene is a plastic and will not absorb heat /minimum heat loss, ✓½
-
-
- K-Ethanoic acid/(CH,COOH) ✓1
L-Ethene ✓1 - Acidified potassium dichromate(VI) OR acidified potassium manganate(VII) ✓1
- K-Ethanoic acid/(CH,COOH) ✓1
-
-
-
✓1
-
✓1
-
- R and Q form an ioninc compound with strong ionic bonds while ✓½ R and S from a covalent compound with weak Van der Waals forces. ✓½
-
-
- Inert electrode is one which does not participate in the reaction / does not affect the products of electrolysis / does not react; ✓1
- Anode - chlorine; ✓1
Cathode - Hydrogen; ✓1
-
- Measure the boiling point / freezing point; ✓1
- If the boiling point freezing point is sharp, then liquid is pure. ✓1
-
- 4M(s) + K2(g) → 2M2K(s) ✓1
OR
4K(s) + O2(g) → 2K2O(s) - L ✓1
- J should be placed in period 3. group 5 of the periodic table. ✓1
- 4M(s) + K2(g) → 2M2K(s) ✓1
-
- Graphite consists of layers of carbon atoms; ✓1
- The layers are held together by the weak Van der Waals forces of attraction; ✓1
- These layers therefore slide over each other thus preventing machine to machine contact. ✓1
-
- Removal of original colour from a substance and the remaining substance is white/ colourless : ✓1
- NaClO / NaOCl ✓1
- Kill germs / bacteria / microorganisms ✓1
-
-
- rock salt/NaCl /trona; ✓½
- salt petre/ NaNO3. ✓½
- To lower the melting point from 800°C to about 600°C; ✓1
-
- street lighting:
- coolant in nuclear reactors:
- extraction of titanium;
- extraction of gold;
- manufacture of sodium cyanide;
- manufacture of sodium peroxide. ✓1
(Any one correct @ 1mk)
-
-
-
-
Alkene, alkyne unsaturated hydrocarbon absent ✓1 - − OH/R − OH present ✓1
-
- Lower a burning splint to the gas, a 'pop' sound should be produced showing it is hydrogen. ✓1
-
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download KCSE 2017 Chemistry Paper 1 with Marking Scheme.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students