Kenya Certificate of Secondary Education
233/2- CHEMISTRY -Paper 2
(THEORY)
2 Hours
INSTRUCTIONS
- Answer all questions in the spaces provided.
- All working must be clearly shown where necessary.
- Mathematical tables and silent electronic calculators may be used.
QUESTION ONE
Study the flow chart below and answer the questions that follow
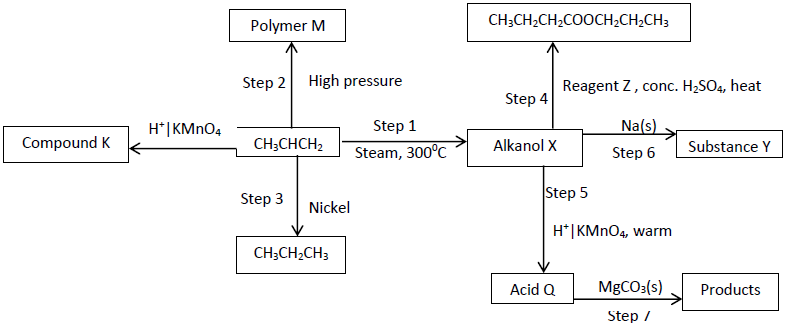
- Give the names of the following (3 marks)
- Compound K
- Substance Y
- Product obtained in step 4
- Identify the type of reaction that takes place in step 1 and give one other condition necessary for the reaction other than the temperature indicated. (1 mark)
Type of reaction
………………………………………………………………………………………………
Condition
……………………………………………………………………………………………... - Draw the structural formula of the following (2 marks)
- Polymer M
- Acid Q
- Give the industrial application of the reaction in step 3. (1 mark)
- Write a chemical equation for the reactions in steps 6 and 7. (2marks)
Step 6
………………………………………………………………………………………………
Step 7
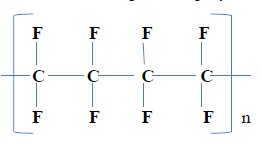
……………………………………………………………………………………………… - Below is the structure of part of a polymer.
- Give the name of the polymer. ( 1 mark)
- Draw the structure of the monomer. From which the above polymer is drawn. (1 mark)
- State one use of the polymer. (1mark)
QUESTION TWO
- Sodium hydroxide pellets were accidentally mixed with sodium chloride. 17.6g of the mixture were dissolved water to make one litre of solution. 100cm3 of the solution was neutralized by 40cm3 of 0.5M dilute sulphuric(VI) acid. (Na=23 , O=16 , H=1 , Cl=35.5)
- Write an equation for the reaction that took place. (1 marks)
-
- Calculate the number of moles of the substance that reacted with sulphuric (VI) acid . (2 marks)
- Calculate the number of moles of the substance that would react with sulphuric (VI) acid in one litre of solution. (1 mark)
- Determine the mass of the unreacted substance in one litre of solution and hence calculate its percentage in the mixture. (3marks)
- To a small portion of the solution mixture obtained, three drops of aluminium sulphate solution was added.
- State the observation that was made. (1 mark)
- Write an ionic equation for the reaction that took place. (1 mark)
- On complete combustion of a sample of a hydrocarbon, 0.352g of carbon (IV) oxide and 0.144g of water were formed. Given the relative molecular mass of the hydrocarbon is 56, determine the molecular formula of the hydrocarbon. C=12 , O=16 , H=1) (3 marks)
QUESTION THREE
- The following are standard reduction potentials for some half-cells.
Half cell Eθ (volts)
Mg2+(aq)|Mg(s) - 2.35
Cu2+(aq)|Cu(s) + 0.34
Zn2+(aq)|Zn(s) - 0.76
Ag+(aq)|Ag(s) + 0.80- Give the chemical symbol of the strongest oxidizing agent. (1mark)
- Arrange the given metals in order of reactivity starting with the least reactive. (1 mark)
- Which two metals, if used to construct an electrochemical cell would produce the lowest e.m.f. ( 1 mark)
- Can a solution of zinc (II) nitrate be stored in a container made of silver? Give a reason. (2 marks)
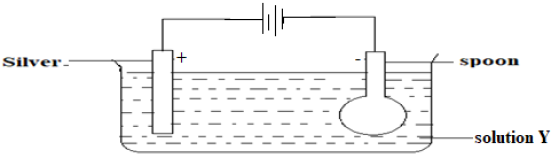
- One of the applications of electrolysis is the electroplating of objects. The set up below shows how we can electroplate a metallic spoon with silver. Study it and answer the questions that follow.
- Write an ionic equation for the reaction that occurs at the cathode. (1mark)
- Identify solution X ( 1 mark)
- State and explain what happens at the anode. (2 marks)
- During the electrolysis of acidified water, a current of 4 amperes was passed through the solution for 5 hours. Calculate the volume of the gas produced at the anode. ( 1F = 96500C , molar gas volume at room temperature = 25dm3) ( 3 marks)
QUESTION FOUR
- Sulphur is extracted by Frasch process in which three concentric pipes are sunk to the deposits which occur at a depth of about 200m. Superheated water and hot compressed air are blown down the outer and inner pipe respectively.
- State two properties of sulphur suitable for its extraction by the Frasch process. (1 mark)
- Give one use of sulphur in the paper industry (1mark)
- The diagram below shows the apparatus used to prepare hydrogen sulphide gas in the laboratory. Use it to answer the questions that follow.
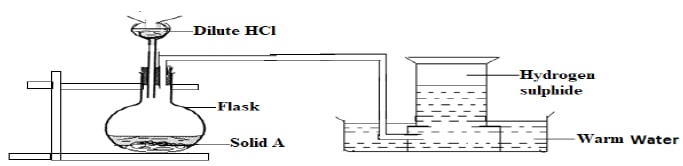
- Name solid A. (1 mark)
- Write an equation for the reaction taking place in flask Y. (1 mark)
- Briefly describe how the gas can be dried. (1 mark)
- Describe an experiment you would use to test for the presence of the gas. (1 mark)
- The diagram below shows some steps during contact process. Study it and answer the questions that follow.
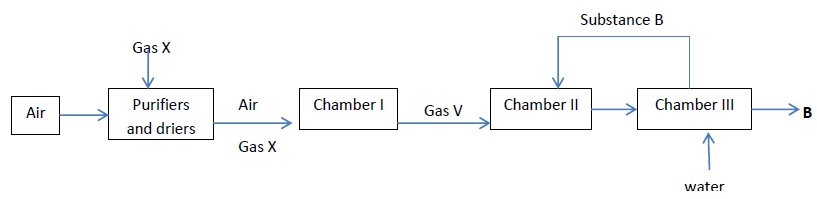
- One of the methods by which sulphur (IV) oxide is produced is by combustion of molten sulphur in air. Write a balanced chemical equation by means of which sulphur (IV) oxide can be produced by the combustion of sulphide ores in air. (1 mark)
- The gases are dried and purified before being passed into the catalytic chamber. Explain. (1 mark)
- Catalytic oxidation of gas X produces another gas V which is an acid anhydride. Explain how gas V can safely be converted to the corresponding acid. (2 marks)
- Name the suitable catalyst used in chamber I. ( 1 mark)
- Explain the major environmental risk associated with the contact process.(1 mark)
QUESTION FIVE
The flow chart below shows a sequence of reactions involving a mixture of two salts, mixture W. study it and answer the questions that follow.
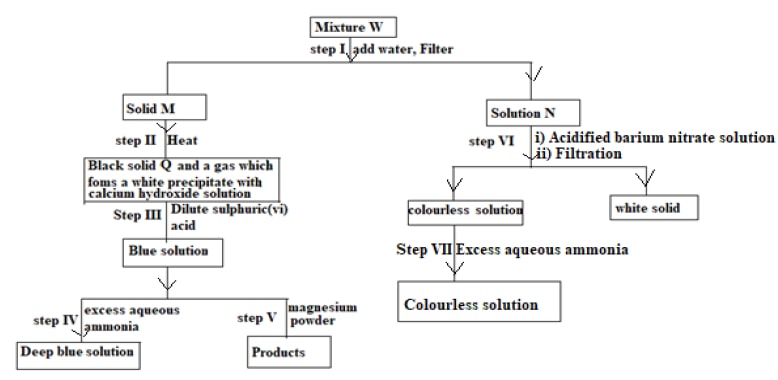
- What property of mixture W is shown in step I (1 mark)
- Name the following (1mark)
Solid M
……………………………………………………………………………………………
Solution N
……………………………………………………………………………………………… - Write an ionic equation for the reaction in step VI (1 mark)
- State and explain the observation made in step V (2marks)
- A solution of ammonia in water is an electrolyte whereas a solution of ammonia in methylbenzene is a non-electrolyte. Explain. (2 marks)
- A saturated solution of salt R at 25oC weighing 56g yielded 14g of anhydrous form of salt R when evaporated to dryness.
- Calculate the solubility of salt R at 25oC. (2marks)
- Give one precaution you would take if you performed the above experiment. (1 mark)
QUESTION SIX
- The table below gives some bond energies of some bonds.
Bond Bond Energy N≡N 945 N – N 278 F – F 159
Calculate the enthalpy change for the reaction; (2marks)
N2(g) + 3F2(g) → 2NF3(g) -
- state Hess’s law (1 mark)
- Define enthalpy of formation of a substance ( 1mark)
- Use the following information to answer the questions that follow
ΔHθf (C2H5OH)(l) = - 276 kJmol-1
ΔHθf (CO2)(g) = - 393 kJmol-1
ΔHθf (H2O)(l) = - 286 kJmol-1- Draw an energy cycle diagram linking heats of formation and combustion of ethanol with its constituent elements and label the various heat changes. (2 marks)
- Calculate the molar heat of combustion of ethanol using the above data. (2marks)
- Using your answer in (ii), calculate the heating value of ethanol. ( C= 12, O= 16, H= 1 ) (1mark)
- Hydration energy of the magnesium gaseous ion is -1931 kJmol-1 while the lattice energy and the heat of solution of magnesium chloride are +2489 kJmol-1 and -170 kJmol-1 respectively. Using an energy cycle diagram, work out the hydration energy of the chloride gaseous ion. (2 marks)
QUESTION SEVEN
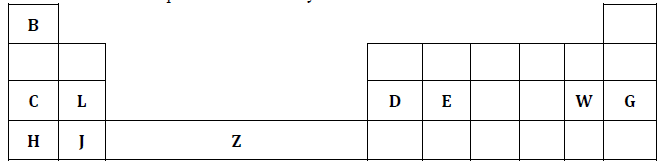
The figure below represents a section of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements.
- What chemical family does element J belong to. (1 mark)
- Compare the reactivity of elements C and H. explain your answer. (2 marks)
- Write the chemical formula of the chloride of D. (1 mark)
- Name the type of structure of the chloride in (c) above. (1 mark)
- Name the bonds that would exist when the compound in (c) above combines with ammonia molecule. (1 mark)
- Using dots(.) and crosses(x) to represent the valence electrons show the bonding in the addition compound formed in (e) above. (1mark)
- State and explain the difference in atomic and ionic radius of element W. (2 marks)
- When a piece of burning element L is lowered into a gas jar containing nitrogen (IV) oxide, it continues to burn. Explain this observation. (2 marks)
Download Chemistry Paper 2 Questions with No Answers - Maseno Mock Exams 2020/2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students