Instruction to the candidates
- Answer all the questions in the spaces in the spaces provided in this paper.
- KNEC Mathematical tables and silent electronic calculators may be used.
- All working MUST be clearly shown where necessary
-
- Define the term homologous series as used in the study of organic chemistry. (1mark)
- Draw the structural formula of the third member of the alkyne homologous series. (1mark)
- 92232Th emits a total of six alpha particles and four beta particles in its natural decay sequence to produce an isotope of lead as the final product.
- Write a nuclear equation to show the above decay process. (1mark)
- State one factor upon which the stability of isotopes depend. (1mark)
- Give one use of Lead applicable in Radioactivity. (1mark)
- The table below shows some properties of substances labeled A, B, C, D and E. Study it and answer the questions that follow.
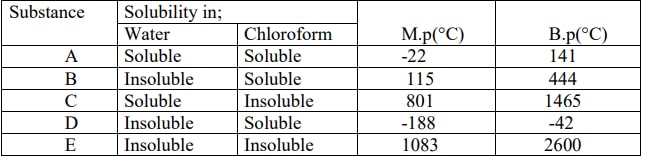
- What is the physical state of substance A at room temperature of 25°C? Explain. (1mark)
- Describe how you would separate a mixture of substances B, C and E. (2marks)
-
- State Boyle’s law. (1mark)
- Identify the substance responsible for the observation made when equal volume of hydrogen chloride gas and ammonia gas are introduced on opposite ends of a long glass tube. (1mark)
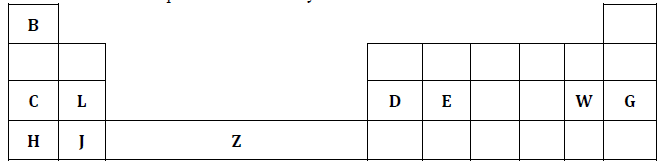
- The diagram below represents the periodic table. Study it and use it to answer the questions that follow.
- An element R has atomic number 35. Indicate by a tick (√) its position in the above grid of the periodic table. (1mark)
- Identify the most electropositive element in the above grid of the periodic table. (1mark)
- Explain the observation made when a compound of W and X is left exposed to moist air. (1mark)
-
- What is meant by the term standard enthalpy of solution? (1mark)
- A beaker contained 95cm3 of aqueous copper (II) sulphate at 43.7°C. When a scrap iron metal was added to the solution the temperature rose to 49.6°C.Given that the mass of copper deposited was 5.83g. Calculate the molar enthalpy change in kJmol-(specific heat capacity of solution= 4.2Jg-k-, d= 1g/cm3, Cu=63.5.) (2marks)
- The diagram below shows a set-up that was used for preparation and collection of nitrogen (IV)oxide.
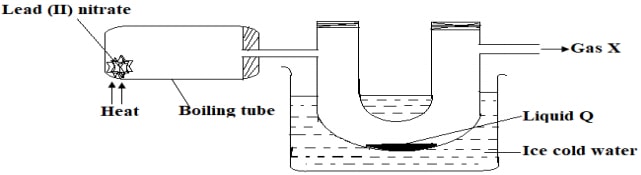
- State any one observation made in the boiling tube a part from brown fumes. (1mark)
- With the use of a chemical equation show how nitrogen (IV) oxide gas would be obtained from liquid Q. (1mark)
- Give one adverse environmental effect of nitrogen (IV) oxide. (1mark)
-
- What is meant by the strength of a base? (1mark)
- The curves below show how electrical conductivity of sulphuric (VI) acid and ethanoic acid vary with concentration.
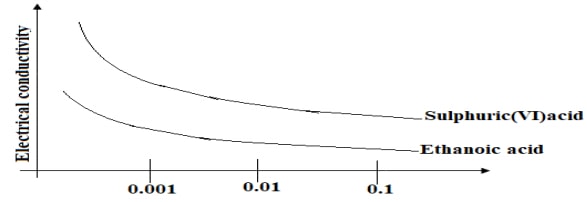
Explain why electrical conductivity of 0.01M sulphuric (VI) acid is higher than that of 0.01M ethanoic acid. (2marks)
- In an experiment a given volume of ammonia gas generated by the reaction of an ammonium salt with an alkali was completely reacted with 250cm3 of 2M phosphoric (v) acid.
- Determine the volume of ammonia gas that was used in the reaction (Molar gas volume at R.T.P = 24000cm3) (2marks)
- Calculate the maximum mass of ammonium phosphate that was obtained in the experiment.
(P=31.0, H=1.0, O=16.0) (1mark)
- The diagram below shows an incomplete set-up of apparatus that can be used in the synthesis of a salt in the laboratory.
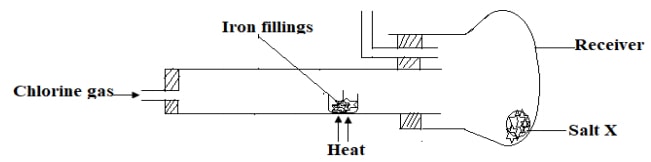
- Describe the appearance of salt X. (1mark)
- Complete the set-up above to show how salt X would be kept anhydrous. (1mark)
- What precaution should be taken to ensure the formation of salt X.? (1mark)
- The apparatus below was used to investigate the effect of carbon(II) oxide on lead(II)oxide.
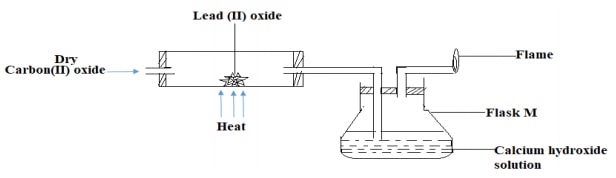
- State the observation that was made in the combustion tube at the end of the experiment. (1mark)
- Write an equation for the reaction that took place in flask M after a while. (1mark)
- State one use of carbon (II) oxide that is not related to its chemical properties illustrated in the set-up above. (1mark)
- The set –up below was used to electroplate a metallic spoon. Study it and answer the questions that follow.
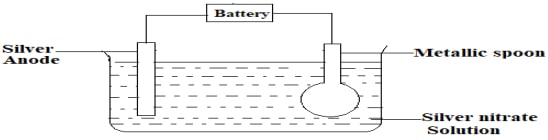
- Write an equation for the reaction that occurred at the cathode. (1mark)
- State and explain what happened at the anode. (1mark)
- Give the name of each of the processes that occur when the following substances are left in the open in petridishes.
- Concentrated sulphuric (VI) acid. (1mark)
- Sodium carbonate decahydrate. (1mark)
- Study a few chart below and use it to answer the questions that follow.
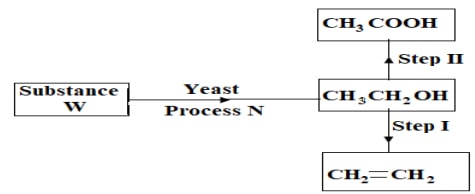
- Name process N. (1mark)
- Suggest a suitable reagent for use in step II. (1mark)
- Describe how step I is carried out. (1mark)
- In an experiment soap solution was added drop wise to a water sample until lather formed. It was found that 12cm3 of soap solution was used for lather to form. A similar volume of the water sample from the same source was subjected to heating and then treated with soap solution in a similar manner. This time 4cm3 of soap was used for lather to form.
- What conclusion can be drawn about the water sample following the two treatments with soap? (1mark)
- Give the systematic name of the compound formed when soap interacts with the water sample before lather forms. (1mark)
-
- State Le’ Chartelier’s principle. (1mark)
- The following reaction is in equilibrium in a closed container.
C(s) + H2O(g) ⇋ CO(g) + H2(g)
How will an increase in pressure affect the yield of hydrogen gas. (1mark)
- Hydrogen sulphide was bubbled into aqueous solution of iron(III) chloride.
- State and explain the observations made. (1mark)
- Write the equation for the reaction that took place. (1mark)
- Using the reaction of chlorine with hot concentrated potassium hydroxide solution and with the help of oxidation numbers, show that a redox reaction occurs. (2marks)
- A student weighed 1.0g of egg shells. He reacted this mass with 15cm3 of 2M hydrochloric acid. After the shells completely dissolved, the resulting solution was all transferred into a volumetric flask and the volume made up to 100cm3 mark by adding distilled water. The solution was labeled P. 20cm3 of this solution required 25cm3 of 0.1M sodium hydroxide for complete neutralization.
- Determine the quantity of excess hydrochloric acid in moles. (1mark)
- Calculate the percentage of calcium carbonate in the egg shell.
( Ca =40.0, C =12.0, O =16.0) (2marks)
- The following table gives the pH values of solution R, S and T.
Solution R S T pH Value 13.7 7.2 1.4 - Which solution will produce carbon (IV) oxide when reacted with sodium carbonate?(1mark)
- What would be the colour of solution R after adding a few drops of phenolphthalein indicator?(1mark)
-
- Name the synthetic allotrope of carbon. (1mark)
- In terms of structure and bonding explain why diamond is used in making drill bits.(1mark)
-
- Show the bonding in H4O++. (1mark)
- Explain the difference in the physical states of existence of hydrides of oxygen and sulphur. (2marks)
- The diagram below shows the energy changes that occur during the reaction;
A + B → C ∆H =?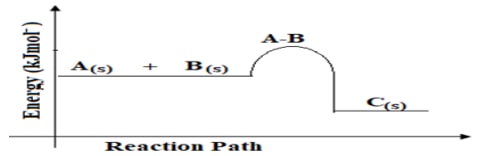
- What does A-B represent? (1mark)
- State the sign of ∆H. Give a reason for your answer. (1mark)
- On the same axes above draw an energy level diagram for the reaction when a suitable catalyst is used. (1mark)
-
- What is meant by a radical. (1mark)
- The table below contains atoms that form common radicals. Complete the table to show radicals formed from various atoms. (2marks)
Element Cl C O
- Ammonia gas is burnt in air enriched with oxygen according to the equation;
4NH3(g) + 3O2(g) 2N2(g) + 6H2O(l)- State one observation made when ammonia is burnt in absence of a catalyst. (1mark)
- If 80cm3 of ammonia are burnt in 100cm3 of oxygen, what will be the total volume of gas after combustion and what will be its composition?
(Assume that all volumes are measured at room temperature and pressure) (2marks)
- Study the set-up below and use it to answer the questions that follow.
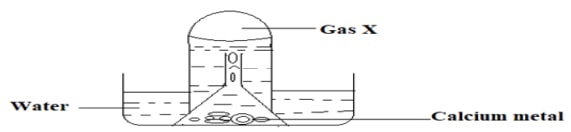
- What physical properties of the metal are demonstrated above. (1mark)
- The resultant solution at the end of the above experiment is found to have a pH value of 9.0.
Explain. (1mark) - State one industrial use of gas X. (1mark)
-
- Name the chief ore from which aluminium metal is extracted. (1mark)
- Give one observation made when aluminium metal is added to hot sodium hydroxide solution. (1mark)
- What property of aluminium makes it suitable for use in making aluminium foils used for food packaging. (1mark)
- When wood is burnt, a grey powder called ash remains. The ash is stirred with water and filtered, a colourless solution is obtained.
- What is the main component of the colourless solution? (1mark)
- Explain your answer in (a) above. (2marks)
- Sodium hydroxide solution and graphite are both capable of conducting an electric current.
What is the major difference between the two substances in their electrical conductivity?(1mark) - Chemical tests were carried out on separate samples of water drawn from the same source.
The observations made were recorded as shown in the table below.
State the inferences made in reactions. (3marks)Test Observation (i) Addition of aqueous calcium chloride No white precipitate (ii) Addition of sulphuric (VI) acid No effervescence, colourless solution (iii)Addition of a few drops of acidified barium nitrate No white precipitate (iv)Addition of excess aqueous ammonia White precipitate, insoluble in excess
Join our whatsapp group for latest updates
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Download Chemistry Paper 1 Questions No Answers - Maseno Mocks 2020/2021.
Tap Here to Download for 50/-
Get on WhatsApp for 50/-
Why download?
- ✔ To read offline at any time.
- ✔ To Print at your convenience
- ✔ Share Easily with Friends / Students